Abstract
Background:
Candida species have an unstable resistance to common antifungal drugs. The treatment of oral candidiasis requires the identification of new anti-Candida agents with no side effects like medicinal plants.Objectives:
The present study aims to investigate the antifungal effects of caspofungin and oregano essential oil on oral Candida species isolated from cancer patients.Methods:
Seventy-three Candida species were identified and isolated by conventional and microbiological tests from cancer patients (n = 100) suspected of oral candidiasis. The minimum inhibitory concentrations (MICs) of the oregano essential oil and caspofungin were determined by microdilution assay and evaluated according to the Clinical and Laboratory Standards Institute (CLSI-2017). The gene regions of samples were studied using the polymerase chain reaction (PCR) method.Results:
Candida glabrata (35, 47.9%) was the predominant species. Most of the Candida strains were isolated from patients with stomach cancer (35, 47.9%). The highest resistance to caspofungin was reported for C. albicans (5.5%). Also, FKS mutant isolates were associated with resistance to caspofungin. MIC90 value for oregano essential oil against C. albicans was 4096 µL/mL, which was equal to MIC90 value against C. glabrata. There was a significant difference between the MICs of caspofungin and oregano essential oil, that inhibits the growth of Candida isolates.Conclusions:
The results of this study showed that the caspofungin has high antifungal effect on Candida species, specially non-albicans Candida. As the findings indicated, the oregano had good anti-Candida potent. Therefore, we could hope to treat fungal diseases by producing an appropriate herbal medicine.Keywords
Caspofungin Oregano Candida fks1 Gene Traditional Iranian Medicine
1. Background
The increased occurrence of oropharyngeal candidiasis in immunocompromised patients needs more studies. Candida albicans is often associated with oral infections in patients with cancer and drug addicts. Among all Candida species, Candida glabrata, is a significant pathogenic yeast in immune-compromised persons, such as cancer patients (1-3). Patients on chemotherapy or radiotherapy have been shown to have increased susceptibility to fungal infections. Studies have reported C. glabrata as the common non-albicans Candida species among cancer patients (4, 5). Due to the high mortality rate and reduced susceptibility of this species to azole drugs, echinocandins like caspofungin (CAS) are used in treating severe infections (6, 7).
Caspofungin is a class of antifungal drugs preventing the formation of 1,3-β-D-glucan polymer in the fungal cell wall by inhibiting 1,3-β-D-glucan synthase enzyme. In most fungi, 1,3-β-D-glucan synthase is formed of FKS subunit, which is induced by fks1 and fks2 genes. The fks1 gene encodes a target of these inhibitors, so mutations in two specific regions of the fks1 subunit of 1,3-β-D-glucan synthase decreased caspofungin susceptibility on Candida spp (8-10).
Due to the increasing rates of oral candidiasis caused by albicans and non-albicans Candida species, studying the drug-resistance frequency and related causes seems necessary (11). Nowadays, scientists pay particular attention to using herbal and traditional medicine too. Medicinal plants have been effectively used for treatment and prevention of infectious diseases. Oregano (Origanum vulgare) is a native plant in Iran cultivated in the northern and western provinces of the country. Oregano essential oil has antimicrobial and antioxidant properties due to its essential oil components, such as alpha-thujene, alpha-pinene, octanone, and thymol (12). Researchers studied the antibacterial effects of oregano in 2020. This research demonstrated that the essential oil of oregano had antibacterial and anti-biofilm forming effects on the Streptococcus mutans isolates (13).
2. Objectives
The present study aimed to evaluate and compare the inhibitory effects of caspofungin and oregano essential oil on oral Candida isolates taken from cancer patients, as well as to investigate the molecular pattern in caspofungin-resistant isolates.
3. Methods
3.1. Isolation of Candida Species
In this study, a total of 100 saliva samples were taken from cancer patients with symptoms of oral candidiasis, such as erythematous lesion, ulcers, and pain, from December 2019 to June 2021. The cancer patients were on chemotherapy and/or radiotherapy and had been admitted to three hospitals in Golestan province, Iran. The saliva samples were collected without stimulation. The study procedures were performed according to medical ethics standards. A written informed consent was obtained from all patients. The demographic data, including age, gender, and clinical history of cancer were documented for each patient. Candida isolates were identified by conventional morphological and biochemical methods (14, 15). Finally, Candida species was confirmed by polymerase chain reaction (PCR) according to company protocol of Candida spp. detection kit (Iranian Gene Fanavar Institute, Iran) using 18S rRNA universal primers (14).
3.2. Evaluation of Minimum Fungistatic Concentration of Oregano Essential Oil
After collecting and identifying the oregano (Origanum vulgare) leaves around Gorgan in northern Iran, the aerial parts of the plant were cleaned and dried in the shade in a ventilated greenhouse at 30°C. After drying, the plant limbs were crushed into small pieces by a Moulinex model mill made in Spain. After sieving by Pars laboratory sieve (Testsieve-Mesh No.), one gram of each was weighed with a digital scale of Sartorius model made in Germany with an accuracy of 0.001 g. After extraction of oregano leaves, it was saturated with 70% ethanol at a ratio of 1 to 10. Using the rotary evaporator and maceration assay, ethanol was evaporated, and the essence was prepared. Finally, the mixture was filtered using a Whatman filter paper No. 2, USA and stored at 4°C until use.
To determin minimum inhibitory concentration (MIC) and obtain a concentration of 8 - 4,096 µL/mL, the oregano essential oil was dissolved in dimethyl sulfoxide (DMSO). The antifungal effect of different concentrations of essence against Candida isolates was determined by broth microdilution method. After incubation at 35°C for 48 hours, the fungal MIC raised to 50% or 90% compared to positive controls, considered as MIC50 and MIC90, respectively. The negative control well contained the stock with RPMI, and the positive control well contained RPMI with yeast suspensions (13). Candida albicans ATCC90028 was used as the reference strain.
3.3. Caspofugin Sensitivity Test of the Clinical Candida Isolates
In microdilution broth test, a yeast suspension (103 CFU/mL) was prepared in RPMI 1640 spectrophotometrically (with glutamines, without bicarbonate, and with PH indicator, Sigma-Aldrich, USA). The concentration range of caspofungin stock (Basel, Switzerland) was 0.03 - 8 µg/mL (16). After preparing a serial dilution from caspofungin and inoculating yeast suspension to wells, the microplates were incubated at 35°C for 48 hours. The wells with yeast suspension and drug stock were used as the positive and negative controls, respectively. According to the standard CLSI M27 (17), strains with MIC > 2 µg/mL and MIC ≤ 2 µg/mL were considered as non-sensitive and sensitive to caspofungin, respectively. C. parapsilosis (ATCC22019) and C. krusei (ATCC6258) were used for quality control.
3.4. PCR Assay
DNA of caspofungin-resistant isolates was extracted by glass bead/phenol–chloroform method. DNA was used as a template for fks1 gene amplification using FKSI-F1719 and FKSI-R2212 (18) (Table 1). Amplification was programmed for an initial denaturation at 94°C for 5 min, followed by 35 cycles of denaturation at 95°C for 30 seconds, annealing at 57°C for 30 seconds, extension at 72°C for 1 min, and a final extension at 72°C for 3 min. The PCR products were electrophoresed on 1% agarose gel, and the bands were visualized under UV light after staining with ethidium bromide.
| Primer Sequence | Sequence (5'-3') | Product (bp) |
|---|---|---|
| FKS1-F1719 | 5'- CATTGCTGTGGCCACTTTAG -3' | 514 |
| FKS1-R2212 | 5'- GATTTCCATTTCCGTGGTAGC -3' | 514 |
3.5. Data Analysis and Statistics
All data were analyzed using chi-square test (SPSS 16.0 version). Either chi‑square or Fisher’s exact test was utilized to analyze the relations between variables. A P-value less than 0.05 was considered as statistically significant.
4. Results
4.1. Patient Characteristics
A total of 73 Candida species were isolated from 100 clinical specimens. The species distribution of Candida isolates was C. glabrata (35, 47.9%), C. albicans (31, 42.5%), C. parapsilosis (5, 6.85%), and C. kefir (2, 2.74%). Out of 73 Candida isolates, 27 (37%) and 46 (63%) isolates were taken from males and females, respectively. The highest incidence rate of Candida isolates (28.9%) was in the cancer patients aged 45 - 60 years. Twenty-nine (39.7%) patients were in an intensive care unit (ICU) at the start of caspofungin therapy. Most of the Candida strains were isolated from patients with stomach cancer (35 out of 73). There was a statistically significant relationship between frequency of Candida species and age groups and type of cancer (Table 2). Also, 23 (65.7%) patients were under chemotherapy.
Abundance of Candida Species and Distribution of Demographic Characteristics in Cancer Patients
| Variables | Candida albicans (n = 31) a | Candida glabrata (n = 35) a | Candida parapsiolosis (n = 5) a | Candida kefir (n = 2)a | Chi‑square Statistics | P-Value |
|---|---|---|---|---|---|---|
| Gender | 0.74 b | 0.054 | ||||
| Male | 8 (10.9) | 18 (24.7) | 1 (1.4) | 0 (0) | ||
| Female | 23 (31.5) | 17 (23.3) | 4 (5.5) | 2 (2.7) | ||
| Age range | 3.54 | 0.023 | ||||
| 0 - 15 | 7 (9.6) | 0 (0) | 0 (0) | 0 (0) | ||
| 15 - 30 | 8 (10.9) | 2 (2.7) | 1 (1.4) | 0 (0) | ||
| 30 - 45 | 6 (8.2) | 12 (16.4) | 1 (1.4) | 0 (0) | ||
| 45 - 60 | 4 (5.5) | 15 (20.6) | 1 (1.4) | 1 (1.4) | ||
| Older than 60 | 6 (8.2) | 6 (8.2) | 2 (2.7) | 1 (1.4) | ||
| Types of cancer | 11.30 | 0.01 | ||||
| Bladder cancer | 5 (6.8) | 4 (5.5) | 0 (0) | 0 (0) | ||
| Kidney cancer | 6 (8.2) | 5 (6.8) | 1 (1.4) | 0 (0) | ||
| Stomach cancer | 11 (15.1) | 22 (30.1) | 0 (0) | 2 (2.7) | ||
| Non-hodgkin cancer | 9 (12.3) | 4 (5.5) | 4 (5.5) | 0 (0) |
4.2. Susceptibility to Antifungal Agents
The comparative in vitro susceptibilities of the Candida isolates to the antifungal agents showed that the MIC50 value for caspofungin against C. glabrata was 2 µg/mL in comparison with the range of reference strain (MIC = 0.25 µg/mL) and was 2-fold higher than MIC50 against C. albicans. Six (8.2%) isolates included C. albicans, and C. glabrata were non-sensitive to caspofungin (MIC > 2 μg/mL). MIC90 value for oregano essential oil against C. albicans was 4096 μL/mL, which was equal to MIC90 value against C. glabrata (Table 3). Comparison of the efficacy of the caspofungin and oregano essential oil against Candida isolates showed a significant difference between these agents.
The changes to the MIC of oregano essential oil against C. albicans showed that the difference between growth and non-growth of caspofungin-resistant isolates was not significant (Table 4). The average MIC of oregano essential oil in comparison with caspofungin against C. albicans and C. glabrata was reported at 3015 µg/mL, in which the most growth fluctuations were seen in densities of 256 and 512 µg/mL.
Comparison of the Growth and Non-growth of Candida Species in the Presence of Oregano Essential Oil
4.3. The fks1 PCR in Caspofungin-Resistance Isolates
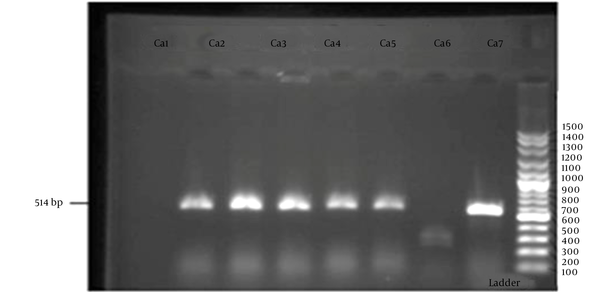
In the present study, 8.2% of isolates showed resistance to caspofungin, out of which four (5.5%) cases belonged to C. albicans and two (2.7%) cases belonged to C. glabrata. In PCR, the presence of 514-bp fragments compared with DNA marker indicated that the test was positive (Figure 1). In this study, all the caspofungin-resistant isolates contained fks1 gene.
Agarose gel electrophoresis of fks1 gene profile for Candida albicans and Candida glabrata isolates; lanes: 1, 2, 3, 4, 5 ,7.
5. Discussion
Studies show that opportunistic infections, like oral candidiasis caused by Candida species, have increased significantly. Although C. albicans is the predominant yeast, there have been reports of candidiasis caused by other Candida species such as C. glabrata. The decreased immune system leads to host susceptibility to pathogens. Cancer can affect the immune system directly through chemotaxis (19). One of the antifungal agents with the high antifungal activity range belongs to the group of echinocondins like caspofungin (20).
The present study was conducted on 100 cancer patients with suspected oral candidiasis. The results showed that from 73 Candida isolates, about 47.9% belonged to C. glabrata. Thus, C. glabrata was the predominant species of oral candidiasis in patients with cancer, and 22 (30.1%) cases were taken from patients with stomach cancer. Of these patients, 15 individuals had reoccurrence of the infection despite receiving antifungal treatments. Researchers identified seven caspofungin-non-susceptible Candida strains (three C. tropicalis, two C. glabrata, and two C. albicans) from 650 Candida isolates from hospitalized cancer patients (21). In present study, six (8.2%) cases showed resistance to caspofungin, out of which two (2.7%) cases belonged to C. glabrata. A study proved that caspofungin had more activity against oropharyngeal non-albicans Candida species (5). According to studies, aminocandin has the potential for extended interval dosing in the treatment of infections caused by C. glabrata (22). Based on the results of the present study, the prevalence of oral candidiasis by non-albicans Candida species is increasing and these species have a potential to reduce susceptibility to antifungal agents. Although caspofungin has a good potent, unfortunately resistance to it among fungi is increasing due to mutation in their structural and regulatory genes, such as fks1 gene (23-25).
In a study by Balashov et al., the reason for caspofungin-resistant among 85 C. albicans isolates studied was reported to be mutation in their fks1 gene (18). In research by Desnos-Ollivier et al. (26) from a total of twenty-five C. albicans isolates, the mutation was reported in 8 isolates. In the present study, all of caspofungin-resistant isolates contained fks1 gene.
The high prevalence of drug-resistant Candida species has emerged in recent years, so it requires the identification of new anti-Candida agents with no side effects or toxicity like medicinal plants. Due to their low toxicity and ease of absorption, medicinal plants have been used all around the world for numerous diseases since a long time. In this regard, there is a long history of applying herbal medicine for treating diseases in Iran (27). In current study, we diagnosed the antifungal virtues of oregano essence as an important medicinal plant. A study indicated that oregano essential oil exerted relatively favorable antimicrobial effects against S. mutans in vitro and in vivo conditions (13).
5.1. Conclusions
According to our results, caspofungin had more antifungal effects on C. glabrata than C. albicans. The detection of mutations in fks1 by DNA sequencing can predict drug-resistance of Candida strains with high sensitivity. However, further research on the approved caspofungin-resistance seems necessary in the future. As the findings indicated, the oregano had good anti-Candida potent. In our study in identical concentration, oregano essential oil had better antifungal activity on C. albicans compared to C. glabrata isolates, but there was a significant difference between the MIC of caspofungin and plant essence. Hence, fungal diseases might be treated by producing an appropriate herbal medicine.
References
-
1.
Yenisehirli G, Bulut N, Yenisehirli A, Bulut Y. In Vitro Susceptibilities of Candida albicans Isolates to Antifungal Agents in Tokat, Turkey. Jundishapur J Microbiol. 2015;8(9). e28057. [PubMed ID: 26495115]. [PubMed Central ID: PMC4609313]. https://doi.org/10.5812/jjm.28057.
-
2.
Duvenage L, Walker LA, Bojarczuk A, Johnston SA, MacCallum DM, Munro CA, et al. Inhibition of Classical and Alternative Modes of Respiration in Candida albicans Leads to Cell Wall Remodeling and Increased Macrophage Recognition. mBio. 2019;10(1). [PubMed ID: 30696734]. [PubMed Central ID: PMC6355986]. https://doi.org/10.1128/mBio.02535-18.
-
3.
Forche A, Solis NV, Swidergall M, Thomas R, Guyer A, Beach A, et al. Selection of Candida albicans trisomy during oropharyngeal infection results in a commensal-like phenotype. PLoS Genet. 2019;15(5). e1008137. [PubMed ID: 31091232]. [PubMed Central ID: PMC6538192]. https://doi.org/10.1371/journal.pgen.1008137.
-
4.
Bagg J, Sweeney MP, Lewis MA, Jackson MS, Coleman D, Al MA, et al. High prevalence of non-albicans yeasts and detection of anti-fungal resistance in the oral flora of patients with advanced cancer. Palliat Med. 2003;17(6):477-81. [PubMed ID: 14526879]. https://doi.org/10.1191/0269216303pm793oa.
-
5.
Shokouhi T, Bandalizadeh Z, Hedayati MT, Mayahi S. In vitro antifungal susceptibility of Candida species isolated from oropharyngeal lesions of patients with cancer to some antifungal agents. Jundishapur J Microbiol. 2011;4(12):19-26.
-
6.
Sipsas NV, Lewis RE, Raad ,I, Kontoyiannis DP. Monotherapy with caspofungin for candidaemia in adult patients with cancer: a retrospective, single institution study. Int J Antimicrob Agents. 2009;34(1):95-8. [PubMed ID: 19251404]. https://doi.org/10.1016/j.ijantimicag.2009.01.006.
-
7.
Pfaller MA, Messer SA, Boyken L, Rice C, Tendolkar S, Hollis RJ, et al. Caspofungin activity against clinical isolates of fluconazole-resistant Candida. J Clin Microbiol. 2003;41(12):5729-31. [PubMed ID: 14662968]. [PubMed Central ID: PMC309007]. https://doi.org/10.1128/JCM.41.12.5729-5731.2003.
-
8.
Chowdhary A, Prakash A, Sharma C, Kordalewska M, Kumar A, Sarma S, et al. A multicentre study of antifungal susceptibility patterns among 350 Candida auris isolates (2009-17) in India: role of the ERG11 and FKS1 genes in azole and echinocandin resistance. J Antimicrob Chemother. 2018;73(4):891-9. [PubMed ID: 29325167]. https://doi.org/10.1093/jac/dkx480.
-
9.
Carrano G, Paulone S, Lainz L, Sevilla MJ, Blasi E, Moragues MD. Anti-Candidaalbicans germ tube antibodies reduce in vitro growth and biofilm formation of C. albicans. Rev Iberoam Micol. 2019;36(1):9-16. [PubMed ID: 30686747]. https://doi.org/10.1016/j.riam.2018.07.005.
-
10.
Sasso M, Roger C, Lachaud L. Rapid emergence of FKS mutations in Candida glabrata isolates in a peritoneal candidiasis. Med Mycol Case Rep. 2017;16:28-30. [PubMed ID: 28491490]. [PubMed Central ID: PMC5413194]. https://doi.org/10.1016/j.mmcr.2017.04.004.
-
11.
Larkin EL, Dharmaiah S, Ghannoum MA. Biofilms and beyond: expanding echinocandin utility. J Antimicrob Chemother. 2018;73(suppl_1):i73-81. [PubMed ID: 29304214]. https://doi.org/10.1093/jac/dkx451.
-
12.
Namvar Aghdash S, Mokhtari M. [Study of anticonvulsive effects of equeous extract of origanumvulgare on chemical kindling in male mice]. J Shahid Sadoughi Univ Med Sci. 2016;24(7):538-46. Persian.
-
13.
Hejazinia F, Fozouni L, Azami NS, Mousavi S. The Anti-Biofilm Activity of Oregano Essential Oil Against Dental Plaque-Forming Streptococcus mutans In Vitro and In Vivo. J Kermanshah Univ Med Sci. 2020;24(3). e107680. https://doi.org/10.5812/jkums.107680.
-
14.
Golirad N, Fozouni L, Pordeli H. Evaluation of Susceptibility to Fluconazole and Voriconazole in Oral Candida glabrata Isolates from Drug Addicts. Int J High Risk Behav Addict. 2019;8(4). e92807. https://doi.org/10.5812/ijhrba.92807.
-
15.
Pappas PG, Kauffman CA, Andes D, Benjamin DK, Calandra TF, Edwards JE, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;48(5):503-35. [PubMed ID: 19191635]. [PubMed Central ID: PMC7294538]. https://doi.org/10.1086/596757.
-
16.
Wanjare S, Gupta R, Mehta P. Caspofungin MIC Distribution amongst Commonly Isolated Candida Species in a Tertiary Care Centre - An Indian Experience. J Clin Diagn Res. 2016;10(11):DC11-3. [PubMed ID: 28050365]. [PubMed Central ID: PMC5198318]. https://doi.org/10.7860/JCDR/2016/23731.8883.
-
17.
Wayne P. Clinical and Laboratory Standards Institute: Reference method for broth dilution antifungal susceptibility testing of yeasts; approved standard. Clinical and Laboratory Standards Institute. 2008;3(document M27-A3 and Supplement S):6-12.
-
18.
Balashov SV, Park S, Perlin DS. Assessing resistance to the echinocandin antifungal drug caspofungin in Candida albicans by profiling mutations in FKS1. Antimicrob Agents Chemother. 2006;50(6):2058-63. [PubMed ID: 16723566]. [PubMed Central ID: PMC1479158]. https://doi.org/10.1128/AAC.01653-05.
-
19.
Kahn JN, Garcia-Effron G, Hsu MJ, Park S, Marr KA, Perlin DS. Acquired echinocandin resistance in a Candida krusei isolate due to modification of glucan synthase. Antimicrob Agents Chemother. 2007;51(5):1876-8. [PubMed ID: 17325225]. [PubMed Central ID: PMC1855530]. https://doi.org/10.1128/AAC.00067-07.
-
20.
Al-Abeid HM, Abu-Elteen K, Elkarmi AZ, Hamad MA. Isolation and characterization of Candida spp. in Jordanian cancer patients: prevalence, pathogenic determinants, and antifungal sensitivity. Jpn J Infect Dis. 2004;57(6):279-84.
-
21.
Kofteridis DP, Lewis RE, Kontoyiannis DP. Caspofungin-non-susceptible Candida isolates in cancer patients. J Antimicrob Chemother. 2010;65(2):293-5. [PubMed ID: 20016020]. https://doi.org/10.1093/jac/dkp444.
-
22.
Brzankalski GE, Najvar LK, Wiederhold NP, Bocanegra R, Fothergill AW, Rinaldi MG, et al. Evaluation of aminocandin and caspofungin against Candida glabrata including isolates with reduced caspofungin susceptibility. J Antimicrob Chemother. 2008;62(5):1094-100. [PubMed ID: 18658194]. [PubMed Central ID: PMC2721694]. https://doi.org/10.1093/jac/dkn304.
-
23.
Denning DW. Echinocandins: a new class of antifungal. J Antimicrob Chemother. 2002;49(6):889-91. [PubMed ID: 12039879]. https://doi.org/10.1093/jac/dkf045.
-
24.
Bachmann SP, Patterson TF, Lopez-Ribot JL. In vitro activity of caspofungin (MK-0991) against Candida albicans clinical isolates displaying different mechanisms of azole resistance. J Clin Microbiol. 2002;40(6):2228-30. [PubMed ID: 12037093]. [PubMed Central ID: PMC130826]. https://doi.org/10.1128/JCM.40.6.2228-2230.2002.
-
25.
Wiederhold NP, Grabinski JL, Garcia-Effron G, Perlin DS, Lee SA. Pyrosequencing to detect mutations in FKS1 that confer reduced echinocandin susceptibility in Candida albicans. Antimicrob Agents Chemother. 2008;52(11):4145-8. [PubMed ID: 18794385]. [PubMed Central ID: PMC2573102]. https://doi.org/10.1128/AAC.00959-08.
-
26.
Desnos-Ollivier M, Bretagne S, Raoux D, Hoinard D, Dromer F, Dannaoui E, et al. Mutations in the fks1 gene in Candida albicans, C. tropicalis, and C. krusei correlate with elevated caspofungin MICs uncovered in AM3 medium using the method of the European Committee on Antibiotic Susceptibility Testing. Antimicrob Agents Chemother. 2008;52(9):3092-8. [PubMed ID: 18591282]. [PubMed Central ID: PMC2533459]. https://doi.org/10.1128/AAC.00088-08.
-
27.
Moradi M, Hassani A, Ehsani A, Hashemi M, Raeisi M, Naghibi SS. Phytochemical and antibacterial properties of Origanum vulgare ssp. gracile growing wild in Kurdistan province of Iran. J Food Qual Hazards Control. 2014;1(4):120-4.