1. Background
Neuropathic pain originates from pathophysiological defects of the peripheral or central nervous system (1, 2). Several animal models were introduced to investigate the pathological and physiological mechanisms of neuropathic pain. One such model is chronic sciatic nerve constriction injury (CCI) for neuropathic pain. Although several behavior measures have been developed to evaluate pain in rodent models rated by responses of the paw to heat or mechanical stimuli, their fidgety activity and reactivity to human contact make it hard to stimulate rodents consistently and gain uniform and trustworthy responses (3).
Sedation increases from minimal to deep in a dose-response manner. Sedation is categorized into minimal, moderate, and deep levels (4). Under deep sedation, subjects show purposeful reaction after painful stimulation. In fact, our previous study showed that when investigating mechanical threshold test by using arterial clips with incremental pressure applied to the base of bilateral toes, being deeply sedated by isoflurane lowered rodents’ fidgety activity and reactivity to human contact, which helped us stimulate the rodents consistently and gain trustworthy responses (3).
2. Objectives
The aim of the present study was to see if deep sedation induced by inhalational anesthesia could help us stimulate the rodents consistently and get trustworthy reactions when using three different pushers for nociceptive mechanical pressure thresholds of the hind paw.
3. Methods
3.1. Animals
All the experiments were administered based on the Guidelines on Ethical Standards for Investigation of Experimental Pain in Animals and approved by the University Ethics Board (Approved No. 2016-8). The experiments were performed on male prairie voles (aged 8 weeks).
3.2. Intervention
Both sham (7 male prairie voles) and CCI rodents (8 male prairie voles) were operated on as described previously. Briefly, the rodents were anesthetized with isoflurane (3%) for the exposure of the common sciatic nerve at the right mid-thigh. In contrast to the sham rodents, in which no ligatures were applied, three ligatures (catgut 6.0) were loosely placed around the sciatic nerve until they elicited a slight twitch in the related lower extremity in CCI rodents.
3.3. Score Methods of Behavioral and Threshold Test
Sham and CCI rodents were evaluated before surgery and four and seven days later. First, for scoring of nociceptive behavior, each of the animals was placed in a chamber for 5 min. Each rodent was assessed with a modified rating method (stance score) as shown before. Briefly, 0 = No identifiably disturbed gait or stance; 1 = Moderately disturbed stance, the foot touching on the ground but with toes contracted; and 2 = Severely disturbed gait and stance, the foot either completely raised from the ground or only the lateral part of the foot lightly touching the ground and toes pulled together (3). Intercalary scores (0.5 and 1.5) were adopted for rodents that showed a behavior between the aforementioned definitions.
Afterwards, the threshold for response to pressure and mechanical stimuli was evaluated by using three different pushers (round, flat, and tip-fine) (Figures 1A-C) with a digital force gauge DSV-50N (Force measurement IMADA, Japan) and arterial clips with incremental pressure (30, 60, 120, 225, 350g/mm2).
Anesthesia induction was performed with 4.0% isoflurane in 100% oxygen in a plastic chamber. After losing the righting reflex, the head of the rodent was placed in a chamber in which 1.3% isoflurane was given to the rodent that was spontaneously breathed, in the meantime, isoflurane concentration at the chamber gas outlet was monitored by using an agent analyzer (Datex-Ohmeda, Helsinki, Finland). After 10 min of equilibration with 1.3% isoflurane, pressure with each pusher was attached to the center of bilateral hind paws at a linearly increasing rate of 0.3 N/s three times at five-minute intervals, until a whole movement of the head, trunk, or extremities (judged as a positive response) was observed. We began with a round pusher, a flat pusher, and then a fine-tip pusher. To avoid tissue damage, the pressure cutoffs of the round, flat, and fine-tip pushers were set at 7.0 N, 5.0 N, and 3.0 N, respectively. The pressure threshold with each pusher was calculated as the average of the three readings. After 10-minute intervals, each clip was attached to the base of bilateral toes for 5 sec. This was performed three times, starting with a 30-g clip in a step-by-step increasing manner after negative reactions until a positive reaction was shown as described previously (3). The threshold was determined as the pressure when a positive reaction was noted.
3.4. Statistics
Data are shown as the mean (SD). All the analyses were performed with GraphPad prism 5. For statistical analyses of the data among limbs of CCI and sham rodents, the analysis of variance test was performed, and then Tukey’s test was conducted for post hoc tests. To analyze the data of each group, repeated measured analysis of variance was run, and then Tukey’s test was performed for post hoc test. We regarded a P value of less than 0.05 as significant.
4. Results and Discussion
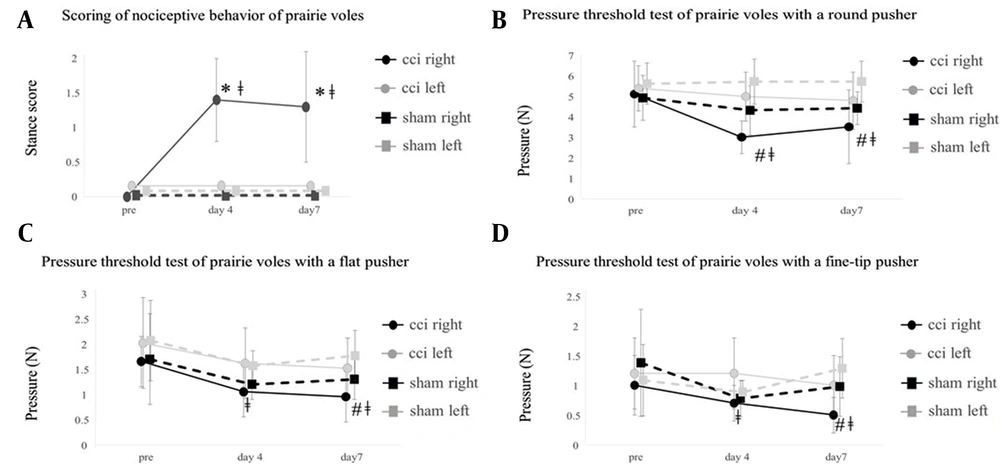
The CCI affected right hind paw prominently increased the scoring of nociceptive behavior (stance score) from the baseline level four and seven days after the experiment, which was prominently different from both the non-CCI affected left hind paw and the hind paws of sham prairie voles (Figure 2A). The CCI affected right hind paw prominently decreased the pressure threshold test of the round pusher from the baseline level four and seven days after the experiment, which was prominently different from the left hind paw of sham prairie voles four and seven days after the experiment (Figure 2B). The CCI affected right hind paw prominently decreased the pressure threshold test of the flat pusher from the baseline level four and seven days after the experiment, which was prominently different from the left hind paw of sham prairie voles seven days after the experiment (Figure 2C).
A: Change in the scoring of nociceptive behavior of prairie voles. *, different from the other hind paws (P < 0.05). ǂ, different from the previous value (P < 0.05). B: Change in the pressure threshold test of prairie voles with a round pusher. #, different from the sham left hind paw (P < 0.05). ǂ, different from the previous value (P < 0.05). C: Change in the pressure threshold test of prairie voles with a flat pusher. #, different from the sham left hind paw (P < 0.05). ǂ, different from the previous value (P < 0.05). D: Change in the pressure threshold test of prairie voles with a fine-tip pusher. #, different from the sham left hind paw (P < 0.05). ǂ, different from the previous value (P < 0.05).
The CCI affected right hind paw prominently decreased the pressure threshold test of the fine-tip pusher from the baseline level four and seven days after the experiment, which was prominently different from the left hind paw of sham prairie voles seven days after the experiment (Figure 2D).
The CCI affected right hind paw prominently decreased the mechanical threshold test of clipping from the baseline level four and seven days after the experiment, which was prominently different from the left hind paw of sham prairie voles at seven days after the experiment.
The present study demonstrated that the CCI affected right hind paw prominently increased the scoring of nociceptive behavior four and seven days after the experiment, and the CCI affected right hind paw prominently decreased pressure and mechanical threshold tests four and seven days after the experiment, which means that the pressure and mechanical thresholds were relevant to the scoring of nociceptive behavior in CCI prairie voles.
Although behavioral measures have been developed to evaluate pain in rodent models rated by responses of the paw to physical stimuli (2, 3), researchers often struggle to get consistent and reliable reactions because of variable laboratory, experimenter, and animal conditions (5). In contrast, our results showed that under deep sedation, behavioral responses were nearly consistent between the three types of mechanical stimuli and constant force clips. These results imply that being deeply sedated by inhalational anesthesia might lower rodents’ fidgety activity and reactivity, which helped us stimulate the rodents consistently and gain trustworthy responses. In addition, since we defined 1.3% isoflurane anesthesia as an opposite deep sedation in our previous study (3), we decided to perform the analyses of the threshold for response to pressure and mechanical stimuli with 1.3 isoflurane anesthesia in the present study and, in fact, neither rodents’ fidgety activity nor reactivity to human contact was observed.
There is a limitation to the present study. Painful stimulation could increase the arousal level; thus, the chronic pain induced by CCI might have influenced the isoflurane induced sedation. Therefore, we should have monitored sedation levels by using electroencephalography.
In sum, the CCI affected right hind paw prominently increased the scoring of nociceptive behavior from the baseline level four and seven days after the experiment, and the CCI affected right hind paw prominently decreased pressure and mechanical threshold tests from the baseline level four and seven days after the experiment, which means the measure applied using pushers and arterial clips while sedated with isoflurane was helpful for mechanical threshold test.