1. Background
Pain is the most common chief complaint of patients in Emergency Departments (EDs) (1). Therefore, one of the most important responsibilities of clinicians and healthcare providers is to eliminate pain (2). Pain caused by urolithiasis is a common clinical problem in EDs affecting 5% to 15% of the world population (3). The prevalence of urolithiasis tends to grow, and its incidence is reported to be 7% and 10% in males and females in developed countries, respectively (4, 5).
Currently, Nonsteroidal Anti-inflammatory Drugs (NSAIDs) and narcotics are the most commonly used medications to relieve pain generated by renal colic. According to the evidence, which compared the efficacy of narcotics and NSAIDs and their adverse effects, the effectiveness of NSAIDs is more or at least equal to narcotics (4).
Given the adverse effects of NSAIDs and narcotics, finding new medications with fewer side effects and good analgesic effects, such as tamsulosin, is essential (4). Renal colic is caused by hyperperistalsis due to ureteral obstruction; therefore, α-blockers, including tamsulosin, dilate the ureter by blocking the α-receptors in distal ureter, increasing the rate of spontaneous stone passage to 65% (6).
As a competitive α1-antagonist with a structure different from other α-blockers, tamsulosin has high bioavailability and a long half-life (9 to 15 hours). This medication has hepatic metabolism and affects the α1A and α1D receptors more than the α1B ones. Furthermore, compared to other α–blockers, tamsulosin causes less orthostatic hypotension (7). To the best of our knowledge, a limited number of studies have been conducted on this topic.
While α1-blockers are routinely used to treat lower urinary tract symptoms, several studies determined the effect of these medicines on decreasing pain, basal tone, frequency of peristalsis, and ureteral contractions (8).
2. Objectives
This study aimed to determine the best way to alleviate pain in patients with renal colic. For this purpose, the analgesic effect of intravenously administered morphine along with control and intravenously administrated morphine along with oral tamsulosin are compared in this study.
3. Methods
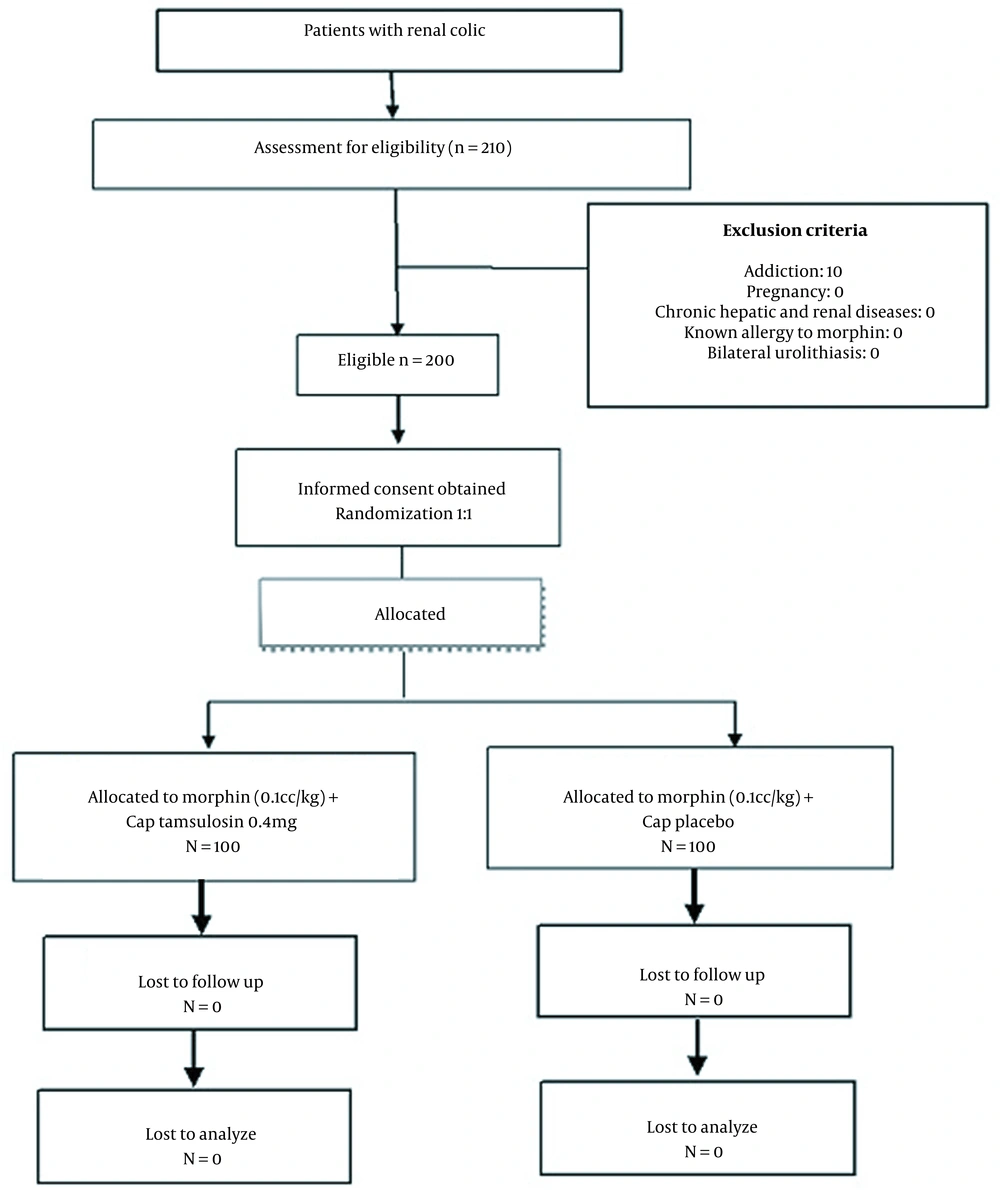
This double-blind clinical trial was conducted in Edalatian ED, a referral emergency ward in Mashhad, Iran, from October to March 2016. Patients referred to the ED with ultrasound-confirmed renal colic aged 18 to 55 were included. They were enrolled in the study based on inclusion criteria and their agreement to participate in the study. The exclusion criteria were drug addiction, pregnancy, chronic hepatic and renal diseases, known allergy to morphine, and bilateral urolithiasis. Through randomized block design, the patients were randomly assigned to two groups, A and B (100 individuals per group). Before prescribing any analgesic, the emergency medicine assistant asked the patients about their pain intensity using a Visual Analog Scale (VAS). The patients’ pain intensity was assessed before, 30 minutes, two, four, and six hours after the treatment using the VAS, while the patients could not see their previous marked values. The patients in group A were treated with morphine (0.1 mg/kg) diluted with normal saline to 10 cc administered over 45 seconds intravenously, along with an oral tamsulosin capsule (0.4 mg) by a nurse researcher colleague. At the same time, the patients in group B received intravenously administered morphine (with the same dose) along with the control. At the end of the study, the methodologist’s colleague opened the coding box, and then the researcher found out which drug each patient had taken.
3.1. Sample Size
The sample size was calculated based on the sample size in clinical trial studies (4). Hence, 87 people in each group were considered, which increased to 100 people with a 15% loss. Also, μ was determined based on the average pain intensity. The methodologist calculated this sample size, which was not implied in any study.
3.2. Data Collection and Processing
Herein, we evaluated the patients with renal colic who were referred to the ED. After performing sonography and confirming renal colic, they were included in this study. Two-hundred patients were divided into two groups based on a random table; a nurse colleague, based on the coding, administered the drug (Figure 1). The emergency medicine assistant reevaluated the patient after 30 minutes, two, four, and six hours after the medication administration. Ultimately, the methodologist opened the coding box, and then both groups were compared.
3.3. Statistical Analysis
Data analysis was performed in SPSS version 16 using the t-test and Mann-Whitney U test to compare the mean pain intensity score of the groups in proportion to the distribution of variables. Moreover, chi-squared and Fisher’s exact test were applied to compare the qualitative variables, and the Friedman test was used to evaluate the patients’ pain intensity change. In all the measurements, P-values less than 0.05 were considered statistically significant.
3.4. Ethical Considerations
Before the study, medicines’ benefits and adverse effects were explained to the participants, and written informed consent was obtained from them.
4. Results
In this clinical trial study of 200 patients, the mean age of the patients was 35.34 ± 8.32 years (range 20 to 54 years) (P = 0.061). Additionally, the patients’ mean weight was 78.77 ± 14.73 kg (range 52 to 115 kg) (P = 0.347).
In this study, 144 (72%) patients were males, and 56 (28%) were females (P = 0.431). Further, a family history of urolithiasis in the patients’ first-degree relatives was observed in 68 (34%) (P = 0.179). None of the patients had a history of analgesic use, and no pregnant subject participated in this study.
The size of calculi was less than 5 mm in 80 (40%) patients, between 5 and 8 mm in 72 (36%) patients, and more than 8 mm in 28 (14%) patients, with a mean of 6.2 ± 4.54 mm (range 1.2 to 30 mm) (P < 0.001). The renal calculi were located in the renal pelvis and lower pole of the kidney, middle calyx, upper calyx, lower calyx, all three calyces, and ureter in 12 (6%), 20 (10%), 16 (8%), 32 (16%), 24 (12%), and 96 (48%) patients, respectively (P = 0.334). Hydronephrosis was observed in 128 (64%) patients (P = 0.105); out of 128 patients, 64, 24 (14%), 8 (4%), 20 (10%), and 8 (4%) patients had mild, mild to moderate, moderate, moderate to severe, and severe hydronephrosis, respectively (Table 1).
| Characteristics | Total (N = 200) | Tamsulosin Group (N = 100) | Placebo Group (N = 100) | P-Value |
|---|---|---|---|---|
| 1. Demographic Variables | ||||
| Age (y) | 35.34 ± 8.32 | 36.44 ± 7.95 | 34.24 ± 8.57 | 0.061 |
| Weight (kg) | 78.77 ± 14.73 | 77.09 ± 13.28 | 80.39 ± 15.91 | 0.347 |
| Sex | 0.431 | |||
| Male | 144 | 69 | 75 | |
| Female | 56 | 31 | 25 | |
| 2. Family History | ||||
| Family history urolithiasis | 68 | 39 (39%) | 29 (29%) | 0.179* |
| 3. Sonographic Findings | ||||
| Stone size* | 6.2 ± 4.54 | 5.46 ± 2.97 | 7.1±5.83 | < 0.001 |
| Location | 0.334 | |||
| Renal pelvis and lower pole of kidney | 12 | 4 (4%) | 8 (8%) | |
| Calyces | 92 | 50 (50%) | 42 (42%) | |
| Ureter | 96 | 46 (46%) | 50 (50%) | |
| Hydronephrosis severity | 0.105 | |||
| Mild | 64 | 35 | 29 | |
| Mild to moderate | 28 | 8 | 20 | |
| Moderate | 8 | 8 | 0 | |
| Moderate to severe | 20 | 15 | 5 | |
| Severe | 8 | 4 | 4 | |
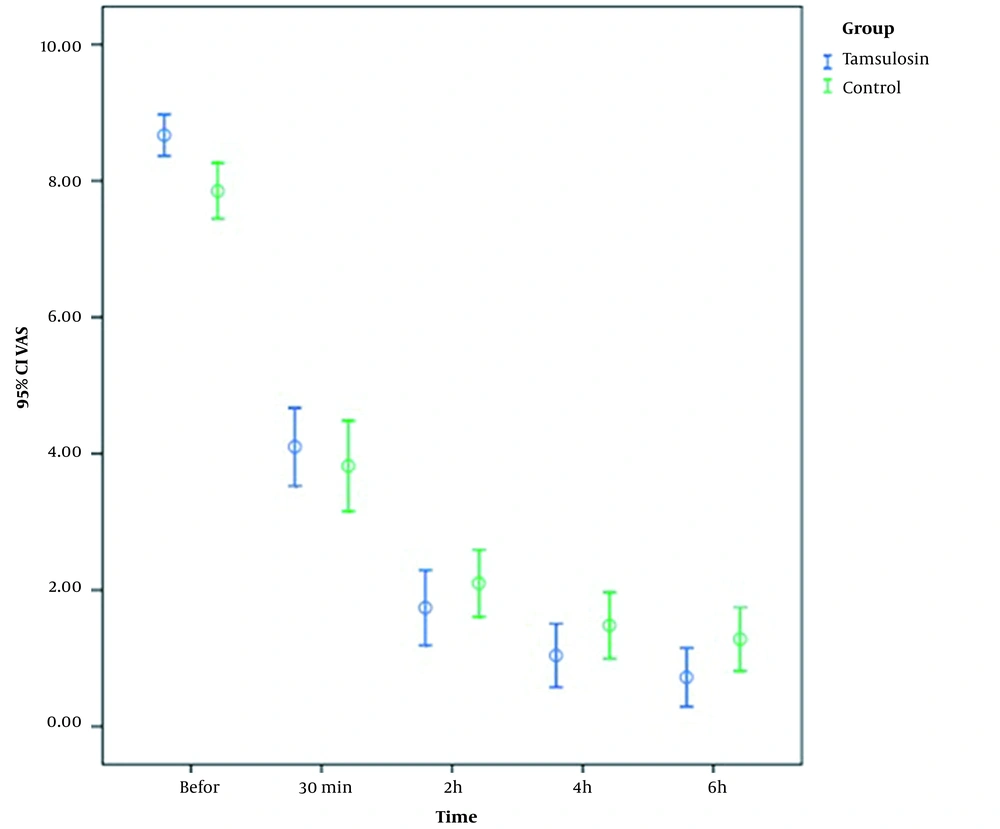
The patients’ pain intensity was assessed before, 30 minutes, two, four, and six hours after the treatment using the VAS, and the results were compared using the Mann-Whitney U test. According to the results, before the intervention, the mean pain intensity was 8.67 ± 1.53 and 7.85 ± 2.05 in the tamsulosin and control groups, respectively (P = 0.003). Additionally, 30 minutes after the treatment, the patients’ pain intensity in the tamsulosin and control groups reduced to 4.1 ± 2.88 and 3.82 ± 3.34, respectively (P = 0.498). Moreover, the pain intensity in the mentioned groups was 1.74 ± 2.77 and 2.1 ± 2.47 two hours after the treatment, respectively (P = 0.137) (Table 2). After four hours, the pain intensity was 1.04 ± 2.35 and 1.48 ± 2.44 in the tamsulosin and control groups, respectively. In addition, the mean pain intensity after six hours was 0.72 ± 2.17 and 1.28 ± 2.35 in the mentioned groups, respectively (P = 0.008) (Figure 2).
| Characteristic | Pain Intensity (N=100) | ||
|---|---|---|---|
| Group A | Group B | P-Value | |
| Before | 8.67 ± 1.53 | 7.85 ± 2.05 | 0.003 |
| 30 min | 4.1 ± 2.88 | 3.82 ± 3.34 | 0.498 |
| 2-hours | 1.74 ± 2.77 | 2.1 ± 2.47 | 0.137 |
| 4-hours | 1.04 ± 2.35 | 1.48 ± 2.44 | 0.028 |
| 6-hours | 0.72 ± 2.17 | 1.28 ± 2.35 | 0.008 |
The adverse effects of the medicines were observed in eight patients, including four (2%) cases of headache and four (2%) cases of diplopia, both in the tamsulosin group (Table 3).
| Characteristic | Side Effect (N=100) | |
|---|---|---|
| Group A | Group B | |
| Headache | 4 (2%) | 0 |
| Diplopia | 4 (2%) | 0 |
5. Discussion
Regarding the results of this study, tamsulosin can control acute pain in patients with renal colic and decrease the use of narcotics. Given that renal colic is caused by ureteral hyperperistalsis, α-blockers such as tamsulosin increase the possibility of spontaneous stone passage due to dilatation of the distal ureter (6). Most former studies were conducted to evaluate the effects of tamsulosin on the speed of stone passage. While most reported decreased pain intensity and episodes in patients, little attention has been paid to this medicine in managing acute pain in patients with renal colic.
Resim et al., in 2005, conducted a study in Turkey on 60 patients in two groups to evaluate tamsulosin’s efficacy in treating distal ureteral calculi (9). The first group received routine treatment, including hydration and NSAIDs, while the second group was prescribed tamsulosin (0.4 mg) in addition to the common treatment. According to the results, spontaneous stone passage was observed in 73.3% and 86.6% of the patients in the first and second groups, respectively (P = 0.196). There were more episodes of ureteric colic in the first group than in the second one during the stone passage (P = 0.038). Further, the mean pain intensity score of the patients in the first and second groups was reported to be 8.3 and 5.7 using the VAS, respectively (P < 0.001). Regarding the study’s results, α1-blockers decreased the number of ureteric colic episodes and pain intensity caused by spontaneous stone passage. These results are in congruence with our findings, especially because the pain intensity measured by the VAS was significantly decreased during each episode.
Furthermore, Cakiroglu et al. conducted a study in Turkey in 2013 on 123 patients with urinary calculus sized between 6 and 15 mm in the lower, middle, or upper ureter, who were candidates for wave lithotripsy (10). The mean size of the stones was 10.7 and 11.4 mm in the control and tamsulosin groups, respectively (P = 0.24). The mean pain intensity in these patients during stone passage was 3.87 and 2.73 in the control and tamsulosin groups using the VAS, respectively (P < 0.001). However, the mentioned study was inconsistent with the current study. This study evaluated acute pain, while Cakiroglu et al. assessed the severity and number of pain episodes during the stone passage. Additionally, the mentioned medication was not used simultaneously with narcotics, and the sizes of stones were greater in the aforementioned study than in the present study. Nevertheless, the analgesic effects of tamsulosin on pain management in the patients were confirmed.
In 2012, Lu et al. conducted a systematic review and meta-analysis to evaluate the effect of tamsulosin on the stone passage (11). Totally, 29 studies were assessed with 2,763 patients to examine the efficacy of tamsulosin and constructive treatment of ureteral calculi. The mentioned analysis determined that tamsulosin therapy led to 19% improvement in renal stone removal. It is consistent with the current study, which demonstrated tamsulosin’s safety and efficacy in treating ureteral stones, and it should be prescribed for most patients with distal ureteral calculi sized less than 10 mm.
Pedram et al., in 2009, conducted a study of 240 patients with renal stones sized between 5 and 20 mm in three groups of A, B, and C (12). The patients in group A (control group) received the routine treatment, including a diclofenac suppository (100 mg) at night, three diclofenac tablets (25 mg) daily, and hydration, the subjects in group B received tamsulosin (0.4 mg) in addition to the routine therapy, and the patients in group C were treated with terazosin (2 mg) along with the routine therapy. After that, the patients were followed-up for three months over three visits. The lowest pain intensity score (4.7) was detected in the tamsulosin group using the VAS, whereas the mentioned score was 5.2 and 5.5 in groups C and A, respectively. In addition, the intravenously administered analgesics decreased in groups B and C (four and two individuals, respectively) compared to the control group (eight subjects). It was confirmed that while α-blockers decreased pain-related symptoms in patients after extracorporeal shock wave lithotripsy, they did not affect stone passage.
Several studies have yielded contradictory results. For instance, in Vincendeau et al. study in 2010, 120 patients (63 and 66 in placebo and tamsulosin groups, respectively) with ureteral calculi sized 2 to 7 mm were assessed (13). The rate of spontaneous stone passage was 70.5% and 77% in the placebo and tamsulosin groups, respectively (P = 0.41). Although tamsulosin was well tolerated, its administration did not increase the stone passage rate.
Issapour et al., in 2009, conducted a study to compare the effect of tamsulosin and indomethacin on the rate and speed of distal ureteral stone passage and revealed no significant difference in this regard (14). In this study, the prescription of tamsulosin was not recommended as a complementary treatment for increasing the stone passage rate without considering the size of the stone and the patient’s age. They found adverse effects of the medicine in eight patients, four patients experienced headaches, and four patients had diplopia. It is worth mentioning that these side effects might be due to the use of narcotics. Subsequently, tamsulosin is a safe medication with low adverse effects and is preferred to common medications used for treating renal colic, including narcotics and NSAIDs.
In addition to the common use and confirmed effects of tamsulosin on stone passage, the current study demonstrated a significant impact of this medicine on decreasing pain intensity in patients with renal colic. Therefore, tamsulosin can be used as a complementary treatment to control the pain in these patients and decrease narcotics use.
5.1. Study Limitations
One of the major limitations of this study was the probable effect of not controlling some of the evaluated criteria, including the size and location of renal stones, the patient’s diet and sleep habits, and the difference between their pain thresholds. On the other hand, using the VAS to evaluate the patient’s pain intensity was patient-based; therefore, this limitation might have affected the results.