1. Introduction
The erector spinae plane block (ESPB), first described by Forero et al., is an innovative regional anesthetic technique that delivers local anesthetics into the interfascial plane between the erector spinae muscles and transverse processes (1). This approach provides a multi-dermatomal sensory block of the anterior, posterior, and lateral thoracic and abdominal walls (1). The ESPB has been widely utilized across various surgeries, including spine surgery (2-5), due to its technical simplicity, safety profile, and effective pain control (6-8). Multiple systematic reviews have demonstrated the benefits of ESPB in spine surgery, such as reducing opioid consumption, decreasing postoperative nausea and vomiting, and shortening hospital stays (8-10). While ESPB is commonly used as an adjunct to general anesthesia (GA) (11-13) or as a postoperative analgesic technique (14), its potential as part of a multimodal regional anesthesia in the context of open spine surgery remains largely unexplored, with only one case documented in the literature (15). This is particularly important when trying to avoid GA-associated complications in high-risk patients (16).
In this report, we present our experience using a multimodal regional anesthetic approach — spinal anesthesia (SA) and ESPB with magnesium sulfate infusion — in a patient with systemic sclerosis and pulmonary fibrosis undergoing laminectomy. This case highlights the potential advantages of regional anesthesia techniques in managing a high-risk patient while minimizing perioperative risks and optimizing patient outcomes.
2. Case Presentation
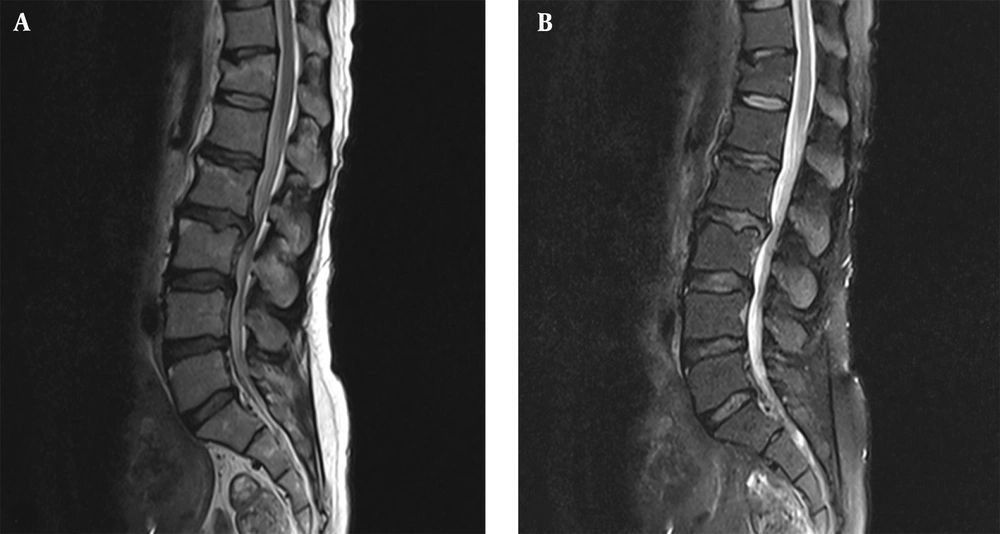
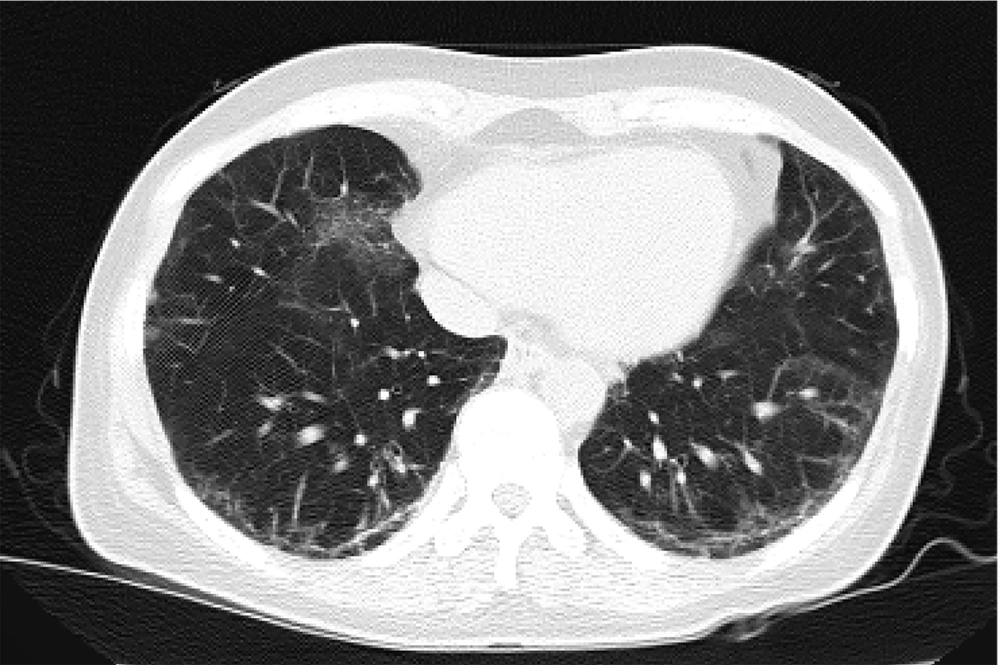
A 46-year-old woman (ASA III) presented with progressive back pain radiating to her left foot, which did not respond to medical management and necessitated surgical intervention. Her medical history was significant for systemic sclerosis with pulmonary fibrosis and pulmonary arterial hypertension, as well as hypothyroidism. The patient had limited mouth opening due to dermal thickening. On neurological examination, she had normal muscle strength and intact reflexes. MRI of the spine revealed spinal stenosis at levels L2-L4 (Figure 1). A chest CT scan showed pulmonary fibrosis (Figure 2), and pulmonary function tests demonstrated a restrictive pattern. Additionally, her systolic pulmonary artery pressure was measured at 40 mmHg, necessitating minimal sedation due to cardiopulmonary concerns.
Given the patient's medical history, which placed her at high risk for respiratory complications associated with general anesthesia, we decided to adopt a regional anesthetic approach. Standard monitoring was implemented, including heart rate, non-invasive blood pressure, pulse oximetry, side stream capnography, electrocardiography, and temperature to ensure hemodynamic stability throughout the surgery.
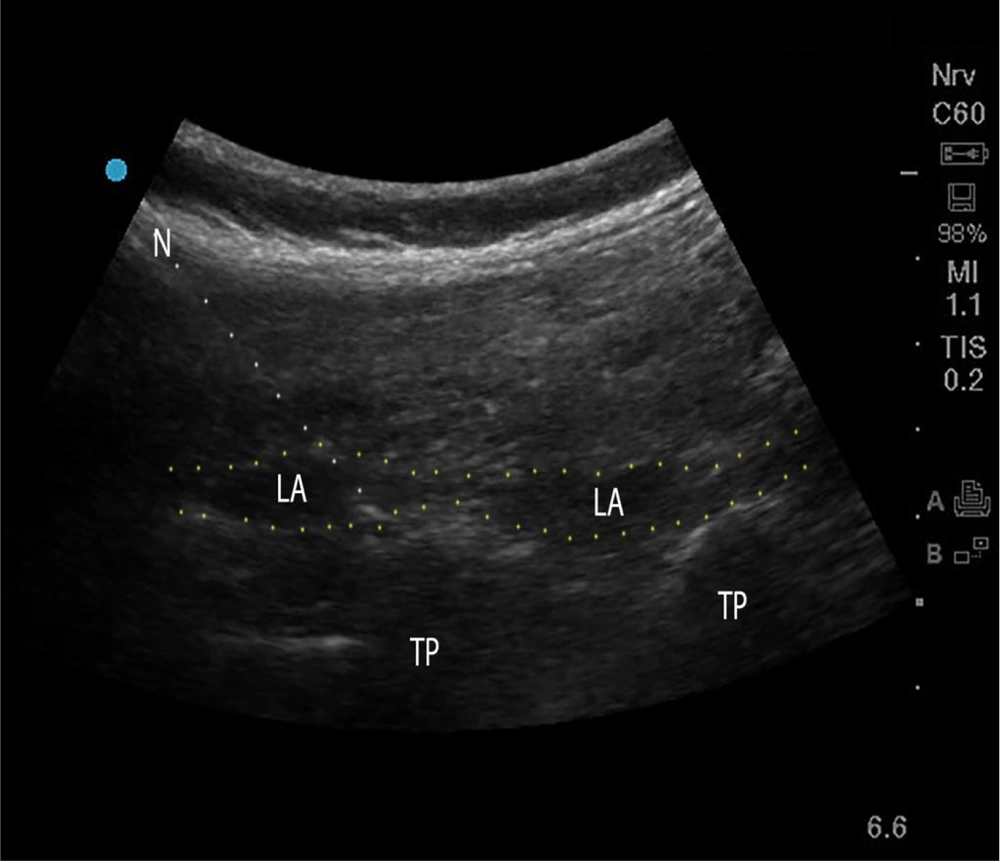
The ESPB was performed under ultrasound guidance using a curvilinear transducer (3 - 8 MHz, Sonosite Edge II; Sonosite, Inc., FUJIFILM). The transducer was positioned to visualize the L3 transverse process. Under sterile conditions, a 22-gauge, 80 mm needle (B. Braun Stimuplex Ultra 360) was advanced craniocaudally using the in-plane approach until the needle tip was in the fascial plane between the erector spinae muscle and the transverse process of the L3 vertebra. A total of 40 mL of 0.25% ropivacaine was injected bilaterally (Figure 3).
Following ESPB, spinal anesthesia was administered at the L3 level. Using a 25-gauge spinal needle, 3 mL of 0.5% bupivacaine was injected into the subarachnoid space after confirming clear cerebrospinal fluid flow. As an adjunct to the regional anesthesia, intravenous magnesium sulfate was loaded and maintained at a rate of 30 mg/kg/h throughout the surgery.
The patient underwent L2-L4 laminectomy to alleviate the spinal stenosis. The surgery took 2.5 hours and was completed without perioperative complications. In the post-anesthesia care unit (PACU), the patient reported no pain (visual analog scale score of 0). The patient was transferred to the neurosurgical ward, where analgesia was first required 12 hours after surgery. Following the ward's standard analgesic protocol, the patient received acetaminophen, gabapentin, and oxycodone at fixed intervals, with diclofenac available as rescue medication. Diclofenac was only needed on postoperative days 2 and 3. The patient was discharged on postoperative day 4.
3. Discussion
We present the anesthetic management of a high-risk patient with systemic sclerosis who underwent lumbar laminectomy using regional anesthesia — SA and ESPB — plus magnesium sulfate infusion. The ESPB in open spine surgery is typically added to general anesthesia or used as postoperative analgesia, making our case the second report of its kind in the literature to use ESPB with spinal anesthesia. This multimodal approach provided both intraoperative and postoperative advantages. Intraoperatively, adequate anesthesia was achieved while avoiding general anesthesia (GA)-associated complications and maintaining hemodynamic stability. Postoperatively, it resulted in sufficient pain control, reduced opioid consumption, and no cardiopulmonary complications or postoperative nausea and vomiting. Spine surgery patients typically experience severe pain in the first 12 hours postoperatively, which intensifies with movement due to surgical trauma and tissue damage (13). With this approach, our patient remained pain-free during this period post-surgery. This is especially important for systemic sclerosis patients as they are more sensitive to respiratory depression associated with traditional opioid-based pain management (13, 17).
Systemic sclerosis is an autoimmune disease characterized by progressive fibrosis of the skin and internal organs such as the heart, lungs, kidneys, and gastrointestinal system (18). The anesthesia approach for these patients must be customized based on the specific organs affected by this condition. Our patient had interstitial pulmonary fibrosis and pulmonary artery hypertension. Additionally, she had dermal thickening and limited mouth opening, which made tracheal intubation challenging. Despite general anesthesia being the standard approach for open spine surgery (19, 20), given our patient’s condition, we opted against it to minimize the risks of potential respiratory complications (16) and adopted a regional anesthesia (RA) approach.
In spine surgery, RA is primarily achieved through either epidural or spinal anesthesia (21). Both techniques, while effective, require careful consideration. Key challenges include the lack of a secure airway, technical difficulties, and potential interference with neuromonitoring (21). In our case, we utilized spinal anesthesia as our primary anesthetic technique. Spinal anesthesia has demonstrated comparable safety and efficacy to GA, with the added benefit of prolonged analgesic effects leading to faster recovery and discharge (22). Although spinal anesthesia alone is considered sufficient for laminectomy (23), we incorporated ESPB and magnesium sulfate into our anesthetic regimen. This decision was primarily influenced by our center's extended operative time of 2.5 hours, compared to typical durations of 79 and 97 minutes for SA and GA, respectively (24). This extended duration was due to our setting as a teaching hospital involving resident training. Managing adequate anesthesia for this prolonged period using SA alone would have required higher doses of local anesthetics, potentially causing hemodynamic instability — a particular concern in our patient.
In a similar approach, Karthik et al. demonstrated the efficacy of combined thoracic segmental SA and ESPB as a safe and effective alternative to general anesthesia in high-risk patients with multiple comorbidities (hepatic, renal, and cardiac), providing hemodynamic stability, superior analgesia, quicker recovery, and early ambulation (25). The ESPB is well-established in spine surgery and has consistently demonstrated improved pain control, lower opioid requirement, enhanced recovery, and patient satisfaction (8, 14). It also enhances surgical conditions via muscle relaxation and provides longer postoperative analgesia, lasting up to 72 hours, ensuring a smoother transition from the intraoperative to postoperative period (22). However, in the context of open spine surgery, it is either used in combination with general anesthesia or in postoperative pain management (8, 14, 26). To our knowledge, there is only one previous record describing the combination of ESPB with spinal anesthesia used for a dual minimally invasive transforaminal lumbar interbody fusion and lumbar decompression in an 87-year-old patient, who tolerated the procedure without any postoperative complications (15). This makes our case particularly valuable in advancing regional anesthesia approaches for open spine surgery. It is noteworthy that while ESPB has been used as the sole anesthetic approach in some cases (27-29), it has not been reported for open spine surgery.
The ESPB typically involves large volumes of local anesthetic, with optimal amounts in adults ranging from 20 - 30 mL (30). Our use of 20 mL of 0.25% ropivacaine bilaterally (40 mL total) aligns with these recommendations. This technique allows the anesthetic to spread within the fascial plane over 3 - 6 vertebral levels in a craniocaudal direction, providing effective multi-level analgesia while ensuring doses remain within safe limits to avoid systemic toxicity (30). In a recent meta-analysis, ESPB was compared to thoracolumbar interfascial plane (TLIP) block and midtransverse process to pleura (MTP) block in lumbar spine surgery, and there were no significant differences in postoperative opioid consumption, pain scores at 24 hours, or incidence of postoperative nausea and vomiting between ESPB and TLIP block (9). However, ESPB has a simpler execution and fewer complications due to its target and needle trajectory being distant from critical structures such as pleura and major vessels (7).
We added intravenous magnesium sulfate infusion into our anesthetic approach due to its well-documented advantage in reducing post-surgery pain, required doses of anesthetic medication, opioid consumption, and postoperative nausea and vomiting (31-34). Magnesium achieves its analgesic effect by blocking NMDA receptors, thereby reducing pain transmission and central nervous system excitability (35). Also, it acts as a calcium channel blocker, leading to bronchodilation and vasodilation in arterioles and coronary arteries (34). Its antiarrhythmic properties help prevent potential bupivacaine-associated tachyarrhythmias, enhancing overall patient safety and outcomes (34). In our study, intraoperative hemodynamics remained stable, with mean arterial pressure decreases limited to less than 20% of initial values without requiring vasopressors, consistent with a similar study in patients undergoing posterior lumbar spinal fusion surgery that measured heart rate and mean arterial pressure without significant differences between magnesium and control groups (36).
In conclusion, our case demonstrated the successful implementation of multimodal regional anesthesia, combining SA, ESPB, and magnesium sulfate infusion for lumbar laminectomy in a high-risk patient with systemic sclerosis. Our experience suggests that this multimodal strategy could be an alternative to general anesthesia for similar high-risk patients undergoing spine surgery, especially when prolonged operative times are anticipated. Our findings represent a single case, and previous studies on this combination have been limited; therefore, future larger prospective studies are needed to establish its role in routine clinical practice.