1. Background
Use of propofol often results in pain upon injection, which can be distressing for patients (1-5). Many patients have reported some degree of pain or discomfort on propofol injection and several interventions have investigated using different drugs and methods to prevent this pain (6-10). Uses of lidocaine, alfentanil, cold saline, or cold propofol are among these methods. In addition, using a larger dorsal hand vein can reduce the pain; however, it might not be available in some patients (11).
Although several methods have been reported to nearly abolish the pain upon injection, further studies are needed to investigate effective and safe methods to reduce pain upon propofol injection. The nitrovasodilators are a various group of drugs that results in vascular relaxation by releasing nitric oxide (NO). These drugs imitate endothelium-derived NO; on the other hand, nitrates and sodium nitroprusside produce NO directly, independent of vascular endothelium (12). A few studies have been conducted to evaluate the use of nitroglycerine, as a single drug, for pain reduction in the patients undergoing propofol injection (13-16). This effect might be due to the pain modulating and anti-inflammatory characteristics of NO, which is a metabolite of nitroglycerine in smooth muscle cells of vessels (17-20).
2. Objectives
This study was performed to evaluate the effects of nitroglycerine on pain severity in patients undergoing propofol injection.
3. Patients and Methods
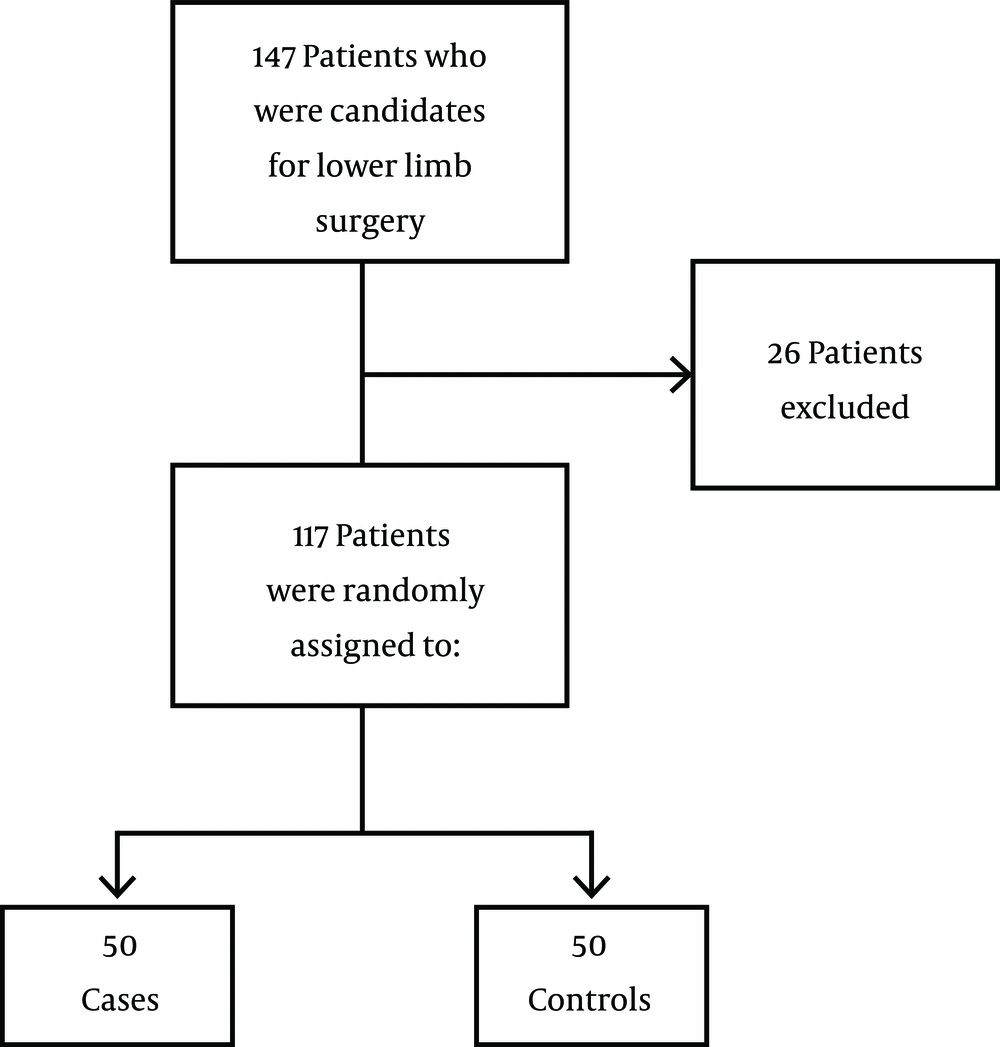
This double-blind randomized clinical trial was conducted in Imam Reza Hospital, Birjand University of Medical Sciences, Birjand City, Iran, 2011 - 2012. A total of 143 patients who were categorized as American Society of Anesthesiologist (ASA) class I and II, aged between 18 to 50 years, and were candidates for an open leg wound surgery were admitted to the hospital; 26 of these patients were excluded according to exclusion criteria and from the remaining 117, 100 patients were randomly selected. The sample size calculation formula was as follows (Equation 1):

where n stands for sample size required in each group, P1 for expected proportion of subject to show no pain in case group, P2 for expected proportion of subject to show no pain in control group, (P1 - P2) for clinically significant difference, Zα /2 for 5% level of significance (1.96), and Zβ for 95% power (21). Based on the study of Nathanson, et al. (4), P1 and P2 were expected to be 0.7 and 0.03, respectively. Thus, n1 and n2 were calculated as 35 that gave us a total sample size of 70. To be more precise, the final sample size was decided to be 100 (50 in each group) (Figure 1).
None of the participants had thin dorsal veins and any history of receiving analgesia or sedation within last 24 hours. In addition, none of the selected patients had contraindications for nitroglycerine use. Informed written consent was obtained from all participants and the Ethical Committee of Imam Reza hospital approved this study. Patients were randomly allocated to case and control groups using the random number table. Without any premedication after the institution of electrocardiogram, arterial blood pressure (systolic, diastolic, and mean arterial pressure) and pulse oximetry for monitoring (Saadat Alborz B7), two 20-G cannula was inserted into the dorsal vein of both hands. The right one was used for infusion of intravenous fluids and the left one was used for drug administration. Then, infusion of 3 mL/kg of 0.9% saline was started from right arm. Furthermore, patients in the case group received infusion of 20 μg of nitroglycerine, diluted in 5 mL of 0.9% saline from the left arm. We decided to choose 20 μg of nitroglycerine based on our goal, i.e., achieving the local effects of nitroglycerine without any sever hemodynamic effect such as sever tachycardia and hypotension. In many previous studies on nitroglycerine dosage, using 20 μg of nitroglycerine intravenously did not cause any severe hemodynamic effects. In addition, in previous studies on analgesic effects of nitroglycerine, the transdermal nitroglycerine patch was used, which was also used in congestive heart failure without any adverse effects and with a low bioavailability (22, 23). Patients in the control group received 5 mL of 0.9% saline from left arm. Administration time for both groups was ten seconds. Both the patient and the anesthesiologist were blinded to the type of administered fluid in the 5-mL syringes. The 5-mL syringes containing nitroglycerine or normal saline were prepared in the same appearance and color by a nurse who was also blinded to the purpose of the study. Twenty seconds later, 100-mg bolus dose of propofol (Lipuro, 10 mg/mL, B. Braun, Melsungen, Germany), which was kept at room temperature, was injected. Propofol was administered over a period of five seconds. Any behavioural signs such as facial grimacing, arm withdrawal, or tears were noted and recorded by the anesthesiologist. Then pain on injection was assessed using a four-point scale: “zero” for no pain, “one” for mild pain (pain only in response to questioning and without any behavioral signs), “two” for moderate pain (pain reported spontaneously without questioning), and “three” for severe pain (strong vocal response or response accompanied by facial grimacing, arm withdrawal, or tears). The induction of anesthesia was completed with the 1.5 mg/kg of propofol for each patient minus the initial 100-mg bolus dose; in addition, atracurium (0.5 mg/kg) and fentanyl (3 μg/kg) were administered and systolic and diastolic blood pressures and heart rate were recorded 30 seconds later. Oral intubation of the trachea was done three minutes after administration of atracurium. Anesthesia was continued with 100% oxygen and isoflurane. Within recovery period, the left hand was examined for pain, edema, or other reactions by the anesthesiologist who was blinded to the administered drug.
The age, sex, weight, homodynamic status (systolic and diastolic blood pressures and heart rate), drug adverse effects and pain severity were the study variables. Data was analyzed using SPSS 18.0 (SPSS Inc, Chicago, Illinois, the United States). Differences were tested by independent-samples t test, Fisher exact test, and Chi square test and were considered statistically significant at P values < 0.05.
4. Results
Table 1 illustrates the basic characteristics of the participants. The pain severity in nitroglycerine group was significantly less in comparison with the control group (P < 0.001) (Table 2). The local adverse reactions were only seen in three patients in control group while no patient in the case group showed any adverse reaction (P = 0.24). The systolic blood pressure showed no significant difference between two groups before and after the induction of anesthesia but the diastolic blood pressure and the heart rate were significantly different between the case and control groups (Table 3).
| Variables | Case | Control | P Valueb |
|---|---|---|---|
| Age, y | 34.1 ± 12.3 | 36.8 ± 11.9 | 0.16 |
| Weight, kg | 68.1 ± 10.8 | 68.5 ± 10.2 | 0.84 |
| Gender | |||
| Male | 30 (60) | 24 (48) | 0.12 |
| Female | 20 (40) | 26 (52) | - |
a Data are presented as mean ± SD or frequency (%).
b P values were calculated using Students’ t test or Chi square as appropriate.
| Pain Severity | Case | Control | P Value b |
|---|---|---|---|
| None | 32 (64) | 9 (18) | < 0.001 |
| Mild | 13 (26) | 16 (32) | - |
| Moderate | 4 (8) | 12 (24) | - |
| Severe | 1 (2) | 13 (26) | - |
a Data are presented as No. (%).
b P value for the difference between case and control groups was calculated using Chi square test.
| Homodynamic Variables | Case | Control | P Value c |
|---|---|---|---|
| SBP before induction, mm Hg | 127.0 ± 15.1 | 123.8 ± 15.7 | 0.08 |
| SBP after induction, mm Hg | 100.5 ± 14.0 | 103.4 ± 13.7 | 0.07 |
| DBP before induction, mm Hg | 80.0 ± 8.46 | 77.9 ± 9.55 | 0.06 |
| DBP after induction, mm Hg | 57.5 ± 12.6 | 64.1 ± 12.3 | < 0.001 |
| Heart rate before induction, beats/min | 88.5 ± 17.2 | 77.2 ± 13.1 | < 0.001 |
| Heart rate after induction, beats/min | 84.3 ± 16.0 | 75.3 ± 9.69 | < 0.001 |
a Data are presented as mean ± SD.
b Abbreviations: DBP, diastolic blood pressure; and SBP, systolic blood pressure.
c P values were calculated using independent-Samples t test.
5. Discussion
To the best of our knowledge, this is the first study to investigate the effects of using intravenous nitroglycerine for reducing the severity of pain caused by propofol injection. This pain is a common problem with a higher frequency in young patients, patients with a thin peripheral vein, and female patients (11). In this study, the effect of applying nitroglycerin before injection of propofol was investigated in 100 patients of ASA classes I and II. The case group developed less pain during the propofol injection.
Nitroglycerin releases NO (24), which acts the same as endothelial NO, resulting in vasodilatation, decreased vascular resistance, lower blood pressure, inhibition of platelet aggregation and adhesion, inhibition of leukocyte adhesion and transmigration, and reduced vascular smooth muscle proliferation. On the other hand, nitrates and sodium nitroprusside directly produce NO, independent of vascular endothelium (24). Administration of nitrovasodilators results in the release of NO, which activates soluble guanylyl cyclase and produces cyclic GMP from guanosine triphosphate in smooth muscle cells (17, 18, 25, 26). Accumulation of cyclic GMP activates cyclic GMP-dependent protein kinase, which is involved in the opening of adenosine triphosphate (ATP)-sensitive K+ channel, to produce spinal or peripheral antinociception and in Na+/K+-ATPase activation (25, 27-29). Nonadrenergic noncholinergic inhibitory reactions to autonomic nerve stimulation are mostly mediated through NO (30). The sensory information processing is partly controlled by afferent nitrergic nerves (31). It has been shown that NO, which can be released from primary sensory nerves, can affect mesenteric vasodilatation (32). NO has an important role in afferent signaling of pain through the dorsal horn of the spinal cord and in autonomic control through nitrergic innervation. Release of NO from the peripheral endings of spinal afferents can stimulate many of their homeostatic actions (31, 33, 34). Some studies have stated that NO inhibitors attenuate the antinociceptive effects of morphine; on the other hand, others have concluded that NO inhibition promotes morphine-induced analgesia (24, 35). The NO formed by N-methyl-D-aspartate (NMDA)-receptor activation diffuses to adjacent nerve terminals to modulate neurotransmitter release (36). Moreover, NO generators have anti-inflammatory effects by blocking the neurogenic component of inflammatory edema when used topically (19, 37). We believe that our results were likely due to a relative dilution of the drug, resulting in a higher venous flow secondary to vasodilation.
In a study on 100 adults of ASA class I and II, scheduled for various elective surgical procedures under general anesthesia, Singh et al. concluded that granisetron, nitroglycerine, and magnesium sulfate were consecutively the most effective drugs in attenuating pain of intravenously injected propofol (38). Likewise, Turan et al. (39) suggested the application of transdermal nitroglycerin for reduction of pain severity of propofol injection. However, O’hara et al. (40) demonstrated that in comparison to nitroglycerine, lidocaine is associated with a decreased incidence of propofol-induced pain. In the current study, instead of topical nitroglycerine, intravenous nitroglycerine was used as a pain reduction agent; thus, the significant pain reduction effect of nitroglycerine can be due to its administration rout.
As a limitation, atracurium was used because we did not have access to cisatracurium. The hemodynamic effects of atracurium might have affected the hemodynamic results of our participants. However, use of a routine drug such as atracurium can be considered as an advantage because of its widespread use in Iran.
In conclusion, the use of nitroglycerine injection may improve analgesic effects without any sever hemodynamic consequences and additional adverse effects. Hence, its use to reduce the propofol injection-induced pain is recommended.