1. Background
Postoperative cognitive dysfunction (POCD) is a common problem in adult patients after surgical procedures. POCD refers to a decline in a variety of neuropsychological domains, such as verbal or visual memory, executive functioning, language comprehension, attention, and concentration (1). Cataract surgery is one of the most common ophthalmic interventions. It can be performed under monitored anesthesia care with or without local anesthesia (2). Certain drugs, including propofol, benzodiazepines (BZDs), and opioids, are used either alone or in combination in cataract surgery (3-5). BZDs may lead to excess sedation and dizziness, especially in older patients (6). Propofol can cause a decreased level of consciousness and excessive sedation (7). Because BZDs and propofol have no analgesic effect, they are used in combination with opioids, but such combinations may increase the risk of dizziness, excessive sedation, and consciousness problems (8). Cataract surgery is typically performed in older patients; hence, the adverse effects of the aforementioned drugs on cognitive functions may be particularly challenging.
Dexmedetomidine is a new α2 agonist with sedative and analgesic effects and no negative effects on respiration, such as respiratory depression (9-12). Furthermore, this drug can induce sedation without increasing delirium (13). Dexmedetomidine has a short half-life (about two hours) and is currently approved for short-term sedation in intensive care units (ICUs) because it has minimal effects on ventilation. Previous pharmacological studies have shown that this drug is a suitable sedative agent for procedures involving mild pain (14).
Working memory is necessary for the memorizing and managing of information. It saves information on a temporary basis and is required for cognitive activities (15).
2. Objectives
In view of the adverse effects of some sedative drugs commonly used in outpatient surgeries, this study used MMSE and N-back tests to compare the efficacy and safety of dexmedetomidine and remifentanil for patients’ cognitive function.
3. Patients and Methods
A double-blind randomized controlled trial was designed and carried out in patients who were candidates for cataract surgery under local anesthesia in Labbafinejad Hospital between 2012 and 2013. Inclusion criteria were as follows: age between 40 and 70 years, ASA class I or II, and surgery duration less than one hour. Patients with an initial mini mental state examination (MMSE) score less than 23, nausea and vomiting more than three times per day, operation duration over 120 minutes, heart rate less than 45, previous history of psychotic disorders, head trauma, or drug abuse were excluded from the study.
Patients were simply randomized into two groups (50 patients per group): remifentanil (R) and dexmedetomidine (D). Demographic data such as age, sex, weight, and operative time were recorded. Likewise, baseline cardiovascular parameters, including blood pressure (BP), heart rate (HR), mean arterial pressure (MAP) and oxygen saturation (SpO2), were recorded.
The MMSE is a so-point questionnaire used extensively in clinical and research settings to measure cognitive impairment (16). Scores under 23 represent serious mental problems and call for psychological treatment. Preoperative MMSE scores were taken for all patients. In the case of POCD, postoperative MMSE scores decreased two or more points from the preoperative scores.
Patients in the D group received 0.5 µg/kg dexmedetomidine (for 10 minutes), 5 minutes before local anesthesia and the maintenance dose then with 0.2 µg/kg/hour was started. During the procedure, the bispectral index (BIS) was maintained between 70 and 80. If it dropped below this range, the maintenance dose was decreased to 0.1 µg/kg/hour and, with increasing BIS, was increased to 0.4 µg/kg/hour.
The loading dose of remifentanil was given at 0.1 µg/kg (for 10 minutes); 5 minutes before local anesthesia in the R group and maintenance dose by 0.05 µg/kg/minutes was started. During the procedure, the BIS was maintained between 70 and 80. With decreasing BIS, the drug dose was decreased to 0.025 µg/kg/minutes, and with increasing BIS, the dose was increased to 0.1 µg/kg/minutes.
Vital signs, such as respiratory rate, SpO2, BP, and HR, were controlled before anesthesia and every 5 minutes during the procedure. All patients received 5 L/minutes oxygen with oxygen mask. Some situations, including hypoxia, bradycardia, tachycardia, pain, vomiting, and anxiety, were managed with appropriate medications.
Bradycardia (HR < 40 for 1 minute) and tachycardia (HR > 100 for 1 minute) were managed with 0.5 mg atropine and 0.1 mg propranolol (up to a maximum dose of 1 mg), respectively. Hypotension (MAP < 60 mmHg for 1 minute) and hypertension (MAP > 120 mmHg for 1 minute) were treated with 10 mg ephedrine and 50 µg TNG, respectively. When SpO2 fell below 92%, oxygen with mask and bag was delivered to the patient until SpO2 reached 92% or higher.
When BIS reached 70 - 80, surgery was begun. HR, MAP, and SpO2 were recorded every 5 minutes. After surgery, drug administration was discontinued and patients were transferred to the recovery room. The MMSE test was done for each patient 120 minutes after surgery.
Data were uploaded into SPSS version 21. Student’s t-test and the Mann-Whitney U test were performed in order to analyze parametric and nonparametric variables. A sample size of 100 patients was estimated for 85% power, and a P < 0.05 was considered significant.
4. Results
There was no significant difference between the two groups with respect to demographic data, including age, male-to-female ratio, ASA class, and operative time (Table 1).
| Demographic Data | Dex | Remi | P Value |
|---|---|---|---|
| Age | 50.26 ± 9.44 | 51.9 ± 10.02 | 0.14 |
| Weight | 69.00 ± 5.63 | 70.46 ± 6.64 | 0.531 |
| Sex | 0.37 | ||
| Male | 30 | 26 | |
| Female | 20 | 24 | |
| Duration of surgery | 35.03 ± 7.62 | 37.73 ± 6.29 | 0.246 |
Comparison of Demographic Data Between the Two Groups
Preoperative assessment revealed no significant difference between the two groups with respect to HR, MAP, and SpO2 (Table 2).
| Dex | Remi | P Value | |
|---|---|---|---|
| Pulse rate | 73.3 ±5.2 | 73.1 ± 4.5 | 0.61 |
| MAP | 96.3 ± 5.5 | 95.2 ± 6.6 | 0.37 |
| O2 Saturation | 98.4 ± 0.5 | 98.5 ± 0.5 | 0.38 |
Comparison of Preoperative Pulse Rate, MAP, and SpO2 Between the Two Groups
Mean MAP and HR were significantly lower in the D group than in the R group both during and after surgery (P = 0.01 and 0.009, respectively), but there was no significant difference in SpO2 between the two groups (Table 3).
| Dex | Remi | P Value | |
|---|---|---|---|
| Pulse rate | 65.4 ± 7.6 | 72.1 ± 4.5 | 0.009 |
| MAP | 76.3 ± 5.4 | 85.2 ± 8.6 | 0.01 |
| SpO2 | 98.8 ± 0.6 | 98.9 ± 0.9 | 0.7 |
Comparison of the Effects of Dexmedetomidine and Remifentanil on Pulse Rate, MAP, and SpO2 After Surgery
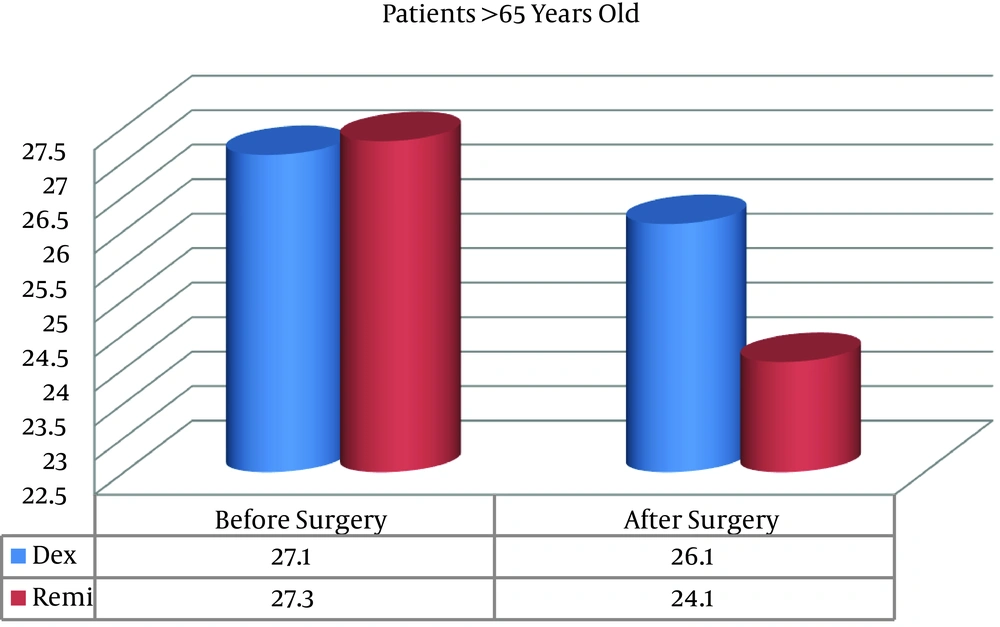
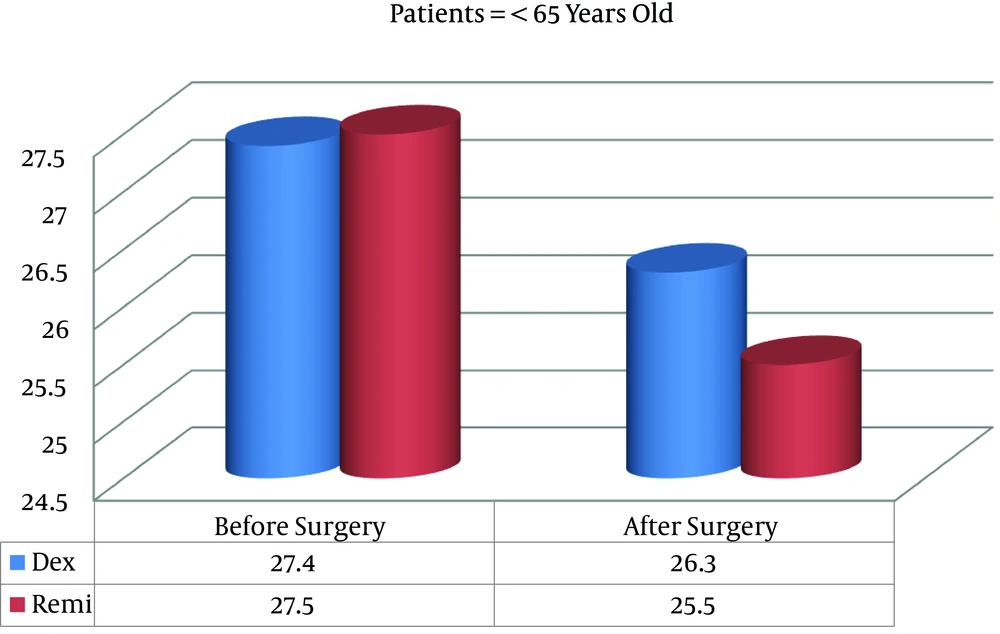
Preoperative MMSE scores were similar between the two groups, but postoperative MMSE scores were significantly better in the D group than in the R group in patients both older and younger than 65 years (Figures 1 and 2).
5. Discussion
Dexemedetomidine is widely used in ICU wards as a sedative and analgesic agent (17). This double-blind randomized clinical trial showed that sedation with dexmedetomidine results in better cognitive function than sedation with remifentanil. HR and MAP in the D group were lower than in the R group, and these differences could be due to decreased sympathetic response and circulatory catecholamine release in the D group. These findings are similar to those of a study by Arain and Ebert that compared the hemodynamic change between dexmedetomidine and propofol for sedation in surgery under local anesthesia (18).
POCD is a defect in cognitive function due to anesthetic drugs. It is a common neurological complication that can last for a few days to several months after surgery and typically leads to decreased concentration and memorial function. Although the pathogenesis of POCD remains unknown, it can affect quality of life and temporarily weaken brain function after anesthesia. POCD related to noncardiac surgery may occur in more than 25% of cases in 7 days after surgery and in more than 9% one month after surgery (16).
Previous studies have confirmed that anesthesia plays a significant role in POCD. Li et al. (19) showed that anesthesia, especially with inhalatory drugs, results in cholinergic disturbance and damage to cognitive structures. Other reports demonstrated that cognitive function may be influenced by the duration of anesthesia and the dose of anesthetic drugs (20-22), and Pan et al. reported that different anesthetic methods exert different effects on cognitive function (23).
In our study, the MMSE test was used to assess cognitive function at different periods. MMSE is a valid, easy-to-take, and brief test that can be administered at bedside. In the current study, the preoperative MMSE score was nearly identical between the two groups, whereas after surgery, the scores of the R group decreased and those of the D group remained relatively unaffected. These results are compatible with the findings of Williams-Russo et al. (24), who showed that with dexmedetomidine, there was no POCD after epidural anesthesia under controlled hypotension.
Maghawry et al. (17) compared the effects of dexmedetomidine and esmolol on brain oxygen saturation in patients that underwent controlled hypoventilation for shoulder arthroscopy. Cognitive disorder may be occasioned by decreased brain-blood flow during hypotension. Brain-blood oxygen monitoring is not routinely performed during anesthesia. Maghawry et al. reported that there was no significant difference between the MMSE scores of the dexmedetomidine and esmolol groups either before or after surgery because of saving brain blood oxygening during surgery even with controlled hypotension (55 mmHg < MAP < 65 mmHg). They concluded that dexmedetomidine is a safer drug than esmolol. Likewise, Townes et al. (25) reported no decrease in the MMSE scores of young patients after induced hypotension.
Confounding factors, such as demographic differences, comorbidities, and history of drug abuse, that may have affected the final results were omitted from this study. A key limitation of this study was its inability to complete long-term patient follow-up.
5.1. Conclusion
This study showed that dexmedetomidine may be a suitable agent for sedation in cataract surgery because it results in a more favorable postoperative cognitive status than remifentanil. Likewise, dexmedetomidine had no significant adverse effects on cardiovascular or respiratory systems.