1. Introduction
Knee osteoarthritis, as a progressive disease is one of the most common causes of pain, motor disorder and disability in the elderly (1-3). Non-surgical interventions for pain control of knee osteoarthritis include weight loss, exercise, changes in daily activities, physiotherapy, nonsteroidal anti-inflammatory drugs (NSAIDs), analgesics and intra-articular injection such as corticosteroids, hyaluronic acid, Growth hormone, dextrose and performing pulsed radiofrequency (4-9). Corticosteroids and NSAIDs are common medical treatments. However, the higher risk of gastrointestinal bleeding is a common systemic side effect of NSAIDs. Furthermore, intra-articular corticosteroid injection causes damage to the articular cartilage, higher incidence of secondary infection and other side effects which limit its consumption (10).
By increasing age, the cartilage is eroded and endures degenerative changes due to physiological and biomechanical changes as well as metabolic effects and trauma (11). The ability of cartilage in regenerating itself is limited due to restriction of vessel perfusion and innervation and less systemic adjustment after degeneration. Therefore, intra-articular injection of drugs or drug combination with the restructuring abilities in cartilage tissue repair has always been the focus of this field (12). Various compounds such as hyaluronic acid, and irritants such as dextrose and erythropoietin have been used so far with various effectiveness levels and mechanisms (8, 13). Growth hormone has generative and constructive effect on cartilage and chondrocyte (9). The concentration of platelets in platelet-rich plasma (PRP) is above the baseline value (14). Platelets are sources with high concentrations of cytokines and a group of growth factors regulating the repair process including the regeneration of tissues (15). It seems that certain growth factors in PRP such as β-Transforming growth factor are associated with chondrogenesis in cartilage repair (16).
In addition, growth hormone leaves regulatory effects in skeletal growth and bone mineral density. This hormone also stimulates the growth of cartilage through the local and systemic production of Insulin like Growth factor-1(IGF-1) and possibly by direct stimulation of chondrocyte proliferation (9, 17).
According to these facts, this study intended to examine the knee joint function by adding growth hormone to PRP in the intra-articular injectate solution.
2. Materials and Methods
In this clinical, randomized, double-blind trial, a total of 54 patients with knee osteoarthritis were enrolled who were candidate for the intra-articular injection. The subjects were included in the study after submitting their written consent. The inclusion criteria were age 40 - 70 years old, stage I or II of knee osteoarthritis (by Kellgren and Lawrence scale, Table 1). The exclusion criteria were rheumatoid arthritis, hemophilia, history of previous knee surgery, drug addiction, alcohol and use of anticoagulants and NSAIDs in the past seven days. This trial was registered with IRCT2015070210599N4.
| Degree | Definition |
|---|---|
| 0 | No radiographic features of osteoarthritis |
| 1 | Possible joint space narrowing and osteophytes formation |
| 2 | Definite osteophyte formation with possible joint space narrowing |
| 3 | Multiple osteophytes, definite joint space narrowing, sclerosis and possible bony deformity |
| 4 | Large osteophytes, marked joint space narrowing, severe sclerosis and definite bony deformity |
The preliminary information of the patients including gender, weight, height and age were recorded.
According to the study by Gobbi et al., the activity scores of patients with knee osteoarthritis before and 6 months after injection of PRP were 77.8 ± 5.7 and 86.3 ± 4.7, respectively. Given the confidence level of 0.05 and power of 95%, the sample size was estimated to be 21 (18). However, the sample size in the present study was expanded to 27 subjects. Then, the patients were divided randomly into two equal groups of 27. Initially, the patient’s knee involvement was measured based on WOMAC scale. WOMAC is composed of the following three components: 1) Pain (5 items), 2) Joint stiffness (2 items), and 3) Knee function (17 items). Scoring in each case ranged from 0 to 4, and the total score of the scale cover 0 to 96 (19). After preparing routine cardiac monitoring (blood pressure, heart rate, and ECG) and blood oxygen saturation, 20ml of patient’s blood was taken intravenously under sterile conditions and placed in the centrifuge kits (Becton Dickinson, UK) for PRP preparation. This solution was centrifuged at 3200 rpm for 20 minutes. The plasma at the top of the solution was isolated and again centrifuged for 5 minutes more at 1500 rpm. Then, 5 mL of plasma at the top of this solution was prepared for intra-articular injection in group P. In group PS, 4 units of growth hormone (Somatropin, Bio Sidus, Argentina) were added to the solution of PRP.
The intra-articular injection was performed as follows: under sterile conditions and, after local anesthesia and putting the multi-frequency linear probe (6 - 13 MHz with 6 cm penetration) placed at the top of the patella horizontally. Sono-visible needle 22 gauge with 50 mm length (Visioplex Needle, Vygon, France) from superolateral quadrant of the patella entered and directed with in-plane technique under ultrasound guidance (SonoSite, S-Nerve) into the knee joint space. Then, the prepared solution was injected into the knee joints without the patients and the physician being aware of the difference between the contents of the syringes. For an hour, the patients were monitored and then discharged in case there were not any adverse effects. In addition, the same procedure was repeated one month later in both groups.
The condition of patient’s knee at baseline (immediately before the first injection), one month later (immediately before the second injection), and two months later (one month after the second injection) were assessed through WOMAC questionnaire, as well as the incidence of any side effects. The data were analyzed through SPSS 20. Chi-square test was used for the qualitative data. The independent t-test was used to compare the quantitative data. To assess the quantitative data in multiple times, the analysis of variance with repeated observations was used. The P values less than 0.05 were considered significant. The Lambda Wilks and Mauchly tests were used to compare differences between groups at different times.
3. Results
In this study, 54 patients with knee osteoarthritis were evaluated. Table 2 displays the demographic variables.
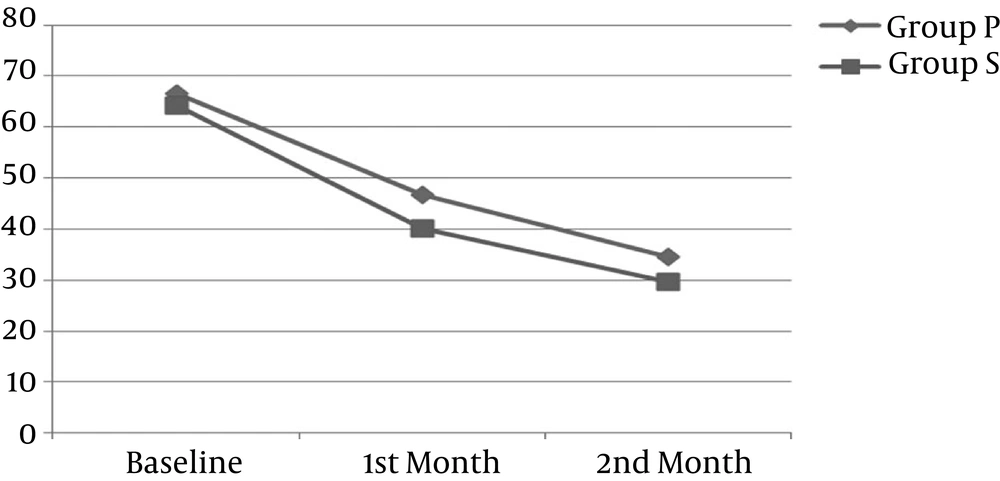
The mean of WOMAC at baseline (immediately before the first injection) was 66.67 ± 9.23 in P group and 64.33 ± 10.69 in PS group (P = 0.460).
The mean of WOMAC one month later (just before the second injection), was 46.67 ± 11.53 in group P and 40.00 ± 17.29 in group PS. Data were analyzed through the independent t-test, indicating that WOMAC in group PS was significantly lower than in group P (P = 0.030).
The mean of WOMAC two months later (one month after the second injection) was 34.62 ± 13.79 in group P and 29.60 ± 18.58 in group PS. The difference between the two groups was not significant (P = 0.235).
| Group P, Mean ± SD | Group PS, Mean ± SD | P Value | |
|---|---|---|---|
| Baseline (immediately before the first injection) | 66.67 ± 9.23 | 64.33 ± 10.69 | 0.460 |
| 1st month (one month after the first injection) | 46.67 ± 11.53 | 40.00 ± 17.29 | 0.030a |
| 2nd month (one month after the second injection) | 34.62 ± 13.79 | 29.60 ± 18.58 | 0.235 |
aThe difference is significant.
The variance test with repeated measure analysis was used to assess the WOMAC scores in the two groups at different times. Moreover, the Mauchly and Wilks Lambda tests were used to compare differences between the groups. The intergroup analysis using the Wilks Lambda test demonstrated that the mean variations of WOMAC decreased significantly in both groups (F = 128, O = 0.0001). Due to the significance of Mauchly test (P = 0.008), the Greenhouse-Geisser test was employed to assess the difference between the two groups. The test showed that the two groups were not significantly different despite a further decline in the WOMAC for group PS at the end of the second month (F = 0.703, P = 0.476).
| Groups | Baseline | First Month | Second Month | P Value |
|---|---|---|---|---|
| Group P | ||||
| Pain | 14.33 ± 2.23 | 9.80 ± 1.96 | 7.61 ± 2.1 | 0.0001 |
| Stiffness | 3.66 ± 1.98 | 2.80 ± 1.11 | 1.62 ± 0.84 | 0.0001 |
| Physical function | 48.68 ± 7.54 | 34.07 ± 8.92 | 20.37 ± 9.11 | 0.0001 |
| Total | 66.67 ± 9.23 | 46.67 ± 11.2 | 34.62 ± 13.79 | |
| Group PS | ||||
| Pain | 14.7 ± 2.45 | 8.4 ± 1.84 | 5.92 ±1.78 | 0.0001 |
| Stiffness | 3.85 ± 1.74 | 2.28 ± 1.56 | 1.56 ± 0.84 | 0.0001 |
| Physical function | 48.35 ± 8.41 | 29.32 ± 9.76 | 22.12 ± 11.55 | 0.0001 |
| Total | 64.33 ± 10.69 | 40.00 ± 17.29 | 29.60 ± 18.58 |
4. Discussion
The results of this study showed that the addition of growth hormone to PRP through intra-articular injection in the knee joint of patients with knee osteoarthritis caused lower WOMAC scores in the first month. In addition, the WOMAC score at the end of the second month declined, even though it was not significantly different.
There have been a variety of non-invasive and semi-invasive methods used in the treatment of knee osteoarthritis. These treatments include physical therapy, nonsteroidal anti-inflammatory drugs, glucosamine, corticosteroid injections, hyaluronic acid, PRP, prolotherapy, radiofrequency, and the use of growth hormone (12). Intra-articular opioids have been used more for acute pain setting (20). There are several studies that evaluated the effects of PRP to alleviate the symptoms and the pain caused by osteoarthritis (16, 17, 19). The activated platelets release mediators such as growth factors and cytokines. In in-vivo studies, the PRP may increase chondrocyte proliferation and differentiation. The PRP is likely to have anti-inflammatory effects, which may exert this effect by inhibiting the NF-KB pathway (21).
In a study on animal model (pig), Lippross found that the intra-articular injection of PRP could significantly reduce joint inflammation (22).
In a study by Kanchanatawan et al., the short-term outcomes of intra-articular injection of PRP in the treatment of knee osteoarthritis were evaluated (23). The results of that study showed that the short-term outcomes(less than one year) from the injection of PRP improved the performance of patients (including the WOMAC) compared with hyaluronic acid and placebo. The researcher argued that intra-articular injection of PRP was generally more effective than hyaluronic acid and placebo in reducing the symptoms and improving the quality of life.
In a study released by Angoorani et al., the intra-articular injection of PRP was compared by stimulating intradermal nerves (24). The results showed that intra-articular injection of PRP provided an effective and safe technique for short-term treatment of patients with knee osteoarthritis. In another study, Forogh et al. examined the effect of a single dose injection of PRP and corticosteroid on knee osteoarthritis (25). The results of this study showed that a single dose of PRP injection reduced the joint pain to a greater extent and longer than corticosteroids. Moreover, it relieved the symptoms and enhanced the daily activity and quality of life in the short term. In this study, the patients’ pain and osteoarthritis outcomes were measured through the visual analogue scale (VAS). The patients were evaluated in the second and sixth months after injection. However, our study rather involved WOMAC which is a more comprehensive scale and the patients received two injections, each time evaluated one month after injection.
In another systematic review conducted by Campbell et al., the intra-articular PRP was compared against corticosteroids, hyaluronic acid, oral NSAIDs and placebo (26). The results showed that intra-articular injection of PRP could provide a therapy for knee osteoarthritis with the potential to relieve symptoms even for 12 months. However, the frequent use of PRP injection increases the risk of adverse reactions (25). In one study, a single dose injection of PRP and its dual injection led to a significant difference in comparison with saline injection (19). In this study, both pain and physical activities similar to the current study were assessed through WOMAC at 1.5, 3 and 6 months after injection. Vaquerizo et al. found that there was a significant difference between the PRP and hyaluronic acid groups, considering 50% reduction in WOMAC scores and the PRP group experienced higher improvement in joint function (27).
Growth hormone is known as an important regulator of bone growth and bone mineral density. This factor stimulates the cartilage growth probably by producing local and systemic IGF-1 as well as by direct stimulation of cartilage cell proliferation. Circulating growth hormone or one of its mediators may be responsible for osteochondral defect repair (28).
This study explored the addition of growth hormone to PRP combined in order to reduce the symptoms of knee osteoarthritis. Compared with only PRP, this combination managed to significantly relieve pain and joint stiffness, thus improving the performance of the patients at the end of the first month after the injection. Although this difference was not significant at the end of the second month, the average pain, joint stiffness and impaired function was to a large extent lower in Group PS than the other group.
Fortier et al. examined the role of growth hormone in cartilage repair as a review study (29). This study showed that the use of growth factors was promising in the treatment of localized cartilage defects such as osteoarthritis. Nonetheless, there is still a need for further studies in this area. The results of a study by Ekenstedt et al. suggested that the chronic lack of growth hormone causes further destruction in the articular cartilage in osteoarthritis (30). In an animal study by Kim et al., it was found that the simultaneous injection of hyaluronic acid and growth hormone improves the osteoarthritis more effectively than hyaluronic acid alone (9).
There were a few limitations in the current study, including the limited follow-up time. It is recommended that future studies should evaluate patients for a longer period.
4.1. Conclusions
The use of platelet-rich plasma along with growth hormone may improve knee function in patients with osteoarthritis such as pain, joint stiffness and effective performance of activities.