1. Background
Patients undergoing total knee arthroplasty (TKA) can experience significant postoperative pain. Adequate analgesia facilitates early rehabilitation and improves patient satisfaction (1). Multimodal protocols that incorporate medications with different mechanisms of action can shorten hospital length of stay and improve perioperative outcomes (2). Such protocols typically utilize peripheral nerve blocks (3) or periarticular local anesthetics (4) in addition to oral medications.
Adductor canal blocks (ACBs), which target the saphenous nerve and provide comparable analgesia to femoral nerve blocks with a lesser degree of quadriceps weakness, have been the focus of multiple recent investigations (5, 6). A limitation of most of the existing literature focusing on ACBs is the exclusion of total knee revision (TKR) surgery. Consequently, there are very few data to guide analgesic management specific to TKR, which can result in more severe postoperative and persistent pain than primary TKA (7). One randomized, controlled trial in TKR patients that compared continuous ACBs to placebo in a multimodal analgesia regimen found that although ACBs improved analgesia during flexion, there were no differences in all secondary endpoints, including opioid consumption and pain at rest (8); however, the study was underpowered. Whether an ACB provides adequate analgesia for TKR in the context of a multimodal analgesia regimen is still not known. Thus, the purpose of this study was to compare postoperative analgesia outcomes between TKA and TKR with the same multimodal protocol including continuous ACBs over a three-year period. We hypothesized that patients who underwent TKR with a continuous ACB in a multimodal protocol would use more opioids than patients who received the same regimen but underwent primary TKA.
2. Methods
2.1. Patient Selection and Measurements
This retrospective review was approved by the Thomas Jefferson University institutional review board (IRB Control #16D.090) with a waiver of requirement for patient consent. Patients were excluded if they were taking ≥ 20 mg of intravenous (IV) morphine equivalents daily at the time of preoperative evaluation, consistent with previous investigations (9). All patients who underwent primary TKA (current procedural terminology (CPT) code 27447 - primary total knee replacement) between 1/1/2014 and 3/29/2016 that was performed by one of four arthroplasty-trained orthopaedic surgeons whose protocols included a continuous ACB (n = 383) were eligible for the study. Of these 383 patients, 30 patients were randomly selected and one patient was excluded for taking ≥ 20 mg of IV morphine equivalents at the time of surgery, yielding a total of 29 patients for analysis. For TKR procedures, eligible patients included those who underwent surgery during the same time period by one of five arthroplasty-trained orthopaedic surgeons whose protocols included continuous ACBs (n = 383). Specifically, patients who underwent TKR and had one of the following CPT codes were eligible: 27486 (revision of total knee arthroplasty, with or without allograft; one component); 27487 (revision of total knee arthroplasty, with or without allograft; femoral and entire tibial component); or 27488 (removal of prosthesis, including total knee prosthesis, methylmethacrylate, with or without insertion of spacer, knee - if a periprosthetic joint infection was present). The records of 33 consecutive patients who underwent TKR were screened, with four patients taking ≥ 20 mg of IV morphine equivalents, yielding 29 patients for analysis. Therefore, 58 patients were ultimately included in the study.
Study variables extracted included: age, gender, body mass index (BMI), surgeon, procedure, estimated blood loss, surgery duration (defined as time from incision to completion of skin closure), presence and duration of tourniquet, number of previous procedures on that knee, preoperative medications, postoperative opioids, numerical pain ratings (0 to 10 scale with 0 indicating no pain and 10 indicating the worst pain imaginable), and postoperative ACB catheter boluses and infusion rates. Diabetic neuropathy was not an exclusion criterion.
2.2. Perioperative Patient Management
All but two patients received spinal anesthesia and a multimodal analgesia regimen of acetaminophen, pregabalin, and celecoxib, which began preoperatively and was continued for 48 hours, unless the patient was allergic to one of the medications. Two patients, both in the TKR group, received general anesthesia. Patients with a sulfa allergy received naproxen instead of celecoxib. Continuous ACBs were placed postoperatively by a fellowship-trained regional anesthesiologist or supervised regional anesthesia fellow using continuous ultrasound guidance and 15 - 25 mL of 0.5% ropivacaine. Intravenous opioids were available to patients in the post-anesthesia care unit (PACU) as intravenous “on-demand” boluses of fentanyl, hydromorphone, or morphine without restriction and administered by the PACU nursing staff for moderate or severe pain (verbal pain score of 5 or greater out of 10). Oxycodone 5 - 10 mg was available to patients postoperatively on the general ward every four hours as needed. Intravenous opioids were available as “on-demand” intravenous boluses to patients on the general ward for breakthrough pain not controlled by the multimodal regimen.
In our regimen, ACBs are routinely placed with continuous ultrasound guidance at the mid-thigh position. The femoral artery is identified deep to the sartorius muscle. Using an out-of-plane technique, the primary block consisting of 15 - 25 mL of 0.5% ropivacaine is placed through a needle with ultrasound confirmation of proper spread lateral to the femoral artery and deep to the sartorius muscle. The catheter is then inserted to a depth of approximately 8 cm at the skin and is secured. A continuous infusion of ropivacaine 0.2% is started in the recovery room at 8 - 10 mL/hr for primary TKA patients and 10 - 14 mL/hr for TKR patients. The infusion of ropivacaine 0.2% can be increased by 2 mL/hr at the discretion of the acute pain management service (APMS), generally for pain ratings of 5 or greater, to a maximum rate of 14 mL/hr. Catheter boluses of ropivacaine 0.2% were given when pain ratings were 5 out of 10 or greater at the discretion of the APMS. Catheters remained in place until discharge for all TKR patients. For primary TKA, catheter removal was either at discharge or at home, depending on surgeon preference. No data relating to analgesia were collected for time periods after discharge.
TKA and TKR were performed using standard techniques with tourniquets applied at the discretion of the orthopaedic surgeon. A medial parapatellar approach was performed in all cases, without any additional extensile approaches for any cases. In primary TKA, posterior stabilized and cruciate-retaining components were utilized, and the patella was resurfaced in all cases. In TKR, extensive synovectomies were performed in every case, utilizing semi-constrained and hinged components. For TKR cases where components were explanted for periprosthetic joint infection, no esmarch was used to exsanguinate the leg, and dynamic and static antibiotic cement spacers were utilized at the discretion of the operating surgeon. No lateral releases were performed in any cases. No additional medications were injected intra-articularly into the knee joint.
All time periods for analgesic consumption calculations began with arrival to the recovery room. Tramadol was not counted in the calculation of total opioid consumption. Opioids, catheter boluses and infusion rates, and pain ratings were extracted from the electronic medical record. Anesthetic and surgical details were retrieved from the hospital’s anesthesia information management system (Innovian, Drager; Lubeck, Germany), orthopedic databases, and patient medical charts. Opioids were converted to IV morphine equivalents using a standard conversion chart (10).
2.3. Sample Size Determination and Data Analysis
It was observed that a mean of 45 mg of IV morphine was consumed during the first 48 hours after TKA in this protocol and a clinically useful difference between groups was determined to be 45% (approximately 20 mg of IV morphine) in opioid consumption between the groups, which was similar to that used in a previous study (5). With power = 0.90 and α = 0.01, it was determined that 29 patients per group were needed.
Continuous parametric data were analyzed using Student’s t-test for independent groups and the Kruskal-Wallis test was used for non-parametric data. Pain ratings were analyzed using ANOVA with repeated measures. Changes in catheter infusion rates were calculated using the Mann-Whitney U test. Least squares regression analysis was used to test for an association between the number of previous knee surgeries and postoperative pain levels. Statistical analyses were performed using Systat, v.13 (San Jose, CA) with P < 0.05 set for statistical significance.
3. Results
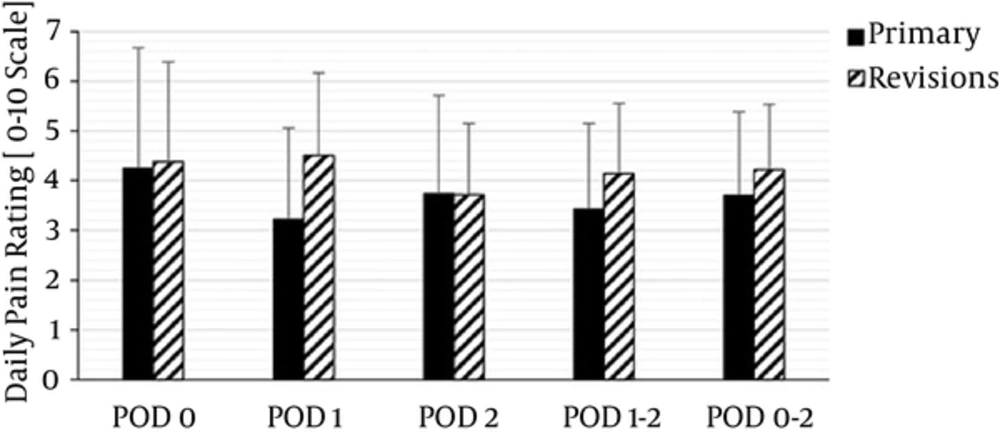
There were no significant differences between the groups with regard to gender, age, or BMI (Table 1); however, surgical duration (P < 0.001) and tourniquet duration (P < 0.001) were both longer for the TKR group. There were no significant differences in the proportion of patients in each group that had a tourniquet applied for surgery. For the primary outcome of 48-hr opioid consumption, there was no significant difference (P = 0.80; Table 2) between primary TKA and TKR groups. Postoperative pain ratings were not significantly different on any postoperative day or overall (Table 2 and Figure 1). A trend toward higher pain ratings in the TKR group was noted for postoperative day 1 only. There were also no significant differences in the secondary outcome variables: number of catheter boluses and catheter infusion rate changes between the two groups (P = 0.67 and 0.29, respectively; Table 2).
| Variable | Primary Knee Arthroplasty (n = 29) | Revision Knee Arthroplasty (n = 29) | P Value |
|---|---|---|---|
| Age, y | 66.5 (8.7) | 67.0 (11.43) | 0.842 |
| Male/Female (% male) | 8/21 (27.6) | 10/19 (34.5) | 0.574 |
| BMI, kg/m2 | 33.3 (6.1) | 33.5 (7.2) | 0.909 |
| ASA Physical Status | 0.255 | ||
| 1 | 1 (3.5) | 0 | |
| 2 | 14 (48.3) | 11 (37.9) | |
| 3 | 14 (48.3) | 18 (62.1) | |
| Surgery Duration, min | 78.5 (24.5) | 119.8 (34.0) | < 0.001b |
| Tourniquet Use, % | 28/29 (96.6) | 26/29 (89.7) | 0.300 |
| Tourniquet Duration, min | 72.9 (24.6) | 106.5 (19.7) | < 0.001b |
Demographics and Surgical Dataa
| Outcome | Primary Total Knee Arthroplasty (n = 29) | Revision Total Knee Arthroplasty (n = 29) | P Value |
|---|---|---|---|
| 48-hour Opioid Consumptionb | 53.0 (27 - 69) | 55.0 (17 - 75) | 0.803 |
| POD 0 Pain Ratings | 3.7 (2.8 - 5.3) | 4.3 (3.2 - 5.8) | 0.135 |
| POD 1 Pain Ratings | 2.8 (1.8 - 5.0) | 4.8 (3.5 - 5.7) | 0.06 |
| POD 2 Pain Ratings | 4.0 (2.5 - 5.2) | 3.8 (3.2 - 4.7) | 0.432 |
| Overall Postoperative Pain Ratings | 3.6 (2.6 - 5.2) | 4.4 (3.0 to 5.2) | 0.323 |
| Total Catheter Boluses | 0.0 (0.0 to 2.0) | 1.0 (0.0 to 2.0) | 0.668 |
| Proportion of Patients with Catheter Infusion Rate Increase, % | 2/29 (6.9) | 7/29 (24.1) | 0.291 |
Postoperative Analgesia Outcomesa
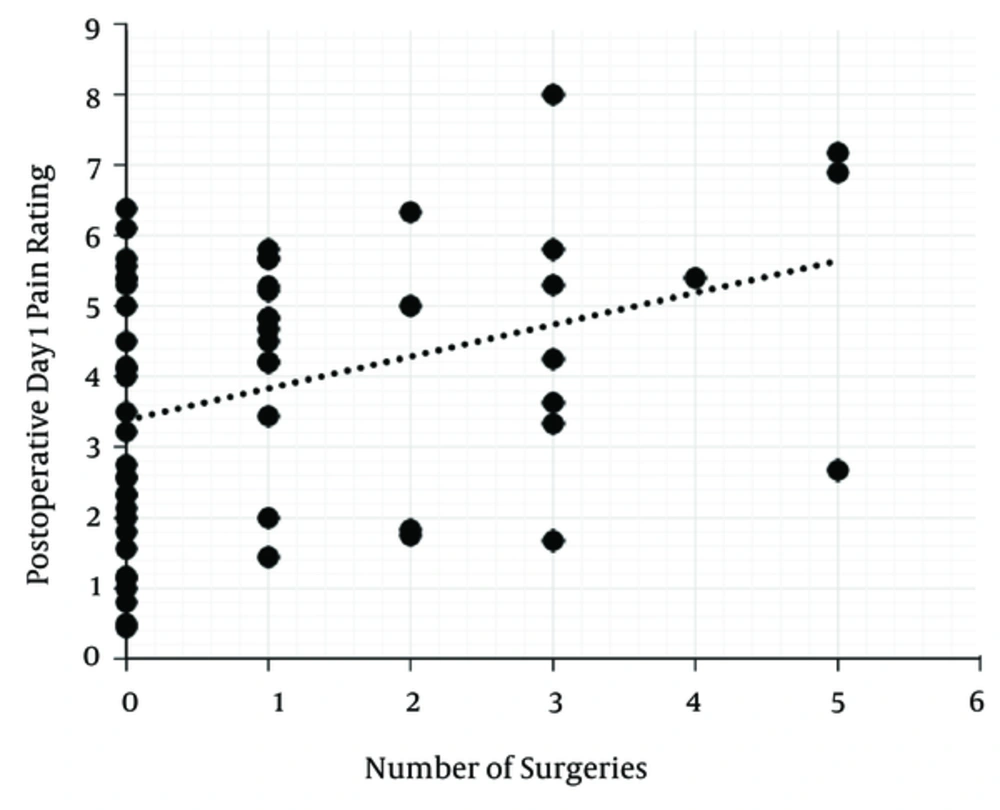
Least squares regression analysis revealed a significant positive correlation (R = 0.35, P = 0.008; Figure 2) between the number of previous knee surgeries and pain ratings on postoperative day 1. There was no significant association between the number of previous knee surgeries and pain ratings on postoperative days 0 and 2. There was also no significant association between the number of previous knee surgeries and opioid consumption, as well as the number of catheter boluses on any postoperative days.
4. Discussion
In this retrospective study, we included patients undergoing both TKA and TKR, a notably important surgical population considering that the number of TKRs may see a cumulative increase of 306% in the United States between 2012 and 2030 (11). As more TKRs are performed, the focus on effective postoperative analgesia should be a priority as it has been recognized as a critical part of successful recovery (12). This is especially important in a healthcare environment that places high value on patient outcomes and satisfaction. We disproved our hypothesis and found that there were no differences in pain levels between these two groups when a continuous ACB was utilized as part of a multimodal analgesic regimen.
This result is somewhat surprising, given the expected increase in pain for TKR patients (7). However, because revisions are often excluded from studies investigating analgesic interventions for knee arthroplasty, little is known about postoperative pain management in this increasingly common patient population. The indication for TKR plays a role in postoperative pain and outcomes; stiffness and infection appear to result in greater levels of postoperative pain and poorer function (13). Although we did not have data on preoperative stiffness, we did observe a trend toward greater pain scores in the revision cohort, particularly on postoperative day 1. This trend during postoperative day 1 could potentially be explained by the resolution of the primary ACB. In one study, breakthrough pain did not occur after ACB for TKA until a median time of 10.5 hours (14), which means many patients would experience block resolution on postoperative day 1. Primary block resolution, therefore, may play a role in the timing of postoperative pain increases. However, these differences were not statistically significant and did not translate into any differences in opioid consumption. It is possible that the median TKR pain rating on postoperative day 1 (4.8 out of 10) did not affect patients enough to request additional opioid analgesics. There was also a trend in the TKR group toward more catheter infusion rate increases, but this also did not reach statistical significance. The ability of the APMS to titrate the continuous ACB may have addressed this difference in pain and decreased the need for additional opioids on postoperative day 1.
One additional factor that could have contributed to the trend toward greater pain levels on postoperative day 1 was the longer tourniquet duration in the TKR group. Several studies have found that longer tourniquet times can worsen postoperative pain after TKA (15, 16) and that could certainly be a contributor toward our observed trend.
The finding that the number of previous surgeries was positively correlated with postoperative pain levels warrants further study. Although the theory that repeated surgery may lead to more tissue trauma and therefore greater risk of persistent postsurgical pain has been suggested for other procedures (17), it has not been well studied in the TKR setting. Although our results suggest that TKR patients as a whole have adequate postoperative analgesia in our multimodal protocol, there were clearly individuals within the cohort that had more severe postoperative pain and tended to be patients who underwent multiple prior knee surgeries. This finding suggests that our multimodal protocol with ACBs may not be adequate for some patients and that alternative analgesic strategies may be needed.
The overall median pain ratings, opioid consumption, and number of ACB catheter boluses were slightly higher in the TKR group, but this difference did not achieve statistical significance. The presence of multimodal analgesia that included acetaminophen, pregabalin, and celecoxib may have affected these results as well. As Hebl et al. (2) reported, a multimodal analgesic pathway that includes peripheral nerve blocks has many benefits, including improved postoperative analgesia. There is also evidence for the use of celecoxib alone in improving postoperative analgesia after TKA (18). Although it is clear that the indication for TKR affects the degree of postoperative pain (13), we did not have adequate numbers to separate patients by indication and could not determine to what degree indication for surgery affected pain.
While the strength of this study is that it reflects clinical practice, this study has several limitations. In addition to limitations inherent to any retrospective study, the timing of pain ratings was not identical for all patients. To account for this, we compared mean pain ratings over 24-hr periods. A second limitation is that all revision surgeries were grouped together. The indication for TKR can have a significant impact on pain, but the number of patients in our cohort was too small to analyze this. Third, preoperative pain ratings were not recorded, which can affect the degree of postoperative pain (19). However, we excluded patients who took ≥ 20 mg of morphine equivalents daily, which should have minimized the chance of including patients with severe preoperative pain. Finally, the starting infusion rates for the catheters were not consistent across patients and TKR patients had higher mean starting infusion rates, reflecting clinical practice. Thus, changes in rates of infusion were analyzed to minimize the differences between starting infusion rates.
In conclusion, patients who underwent TKR had a similar postoperative pain as those who underwent primary TKA while using the same multimodal protocol that included a continuous adductor canal block. Prospective studies that include TKR patients should be undertaken to confirm these findings.