1. Introduction
Interstitial nephritis is a common extraglandular manifestation of Sjogren’s syndrome (1). The renal manifestation in these patients include hypokalemia and metabolic acidosis. Hypokalemia leading to flaccid hypotonic muscular paralysis is a rare but reported presentation in patients with distal renal tubular acidosis associated with Sjogren’s syndrome (2). Hypomagnesemia is rare but has been described in sporadic case reports (3, 4). Normocalcemic carpopedal spasm is rarely seen in these patients. There is scant literature on normocalcemic tetany, and a causal mechanism is attributed to severe hypokalemia and or hypomagnesemia. We report unusual hypokalemic periodic paralysis (negative neurological manifestation) and persistent tetany (neuro-excitatory manifestation) in a woman with Sjogren’s syndrome.
2. Case Presentation
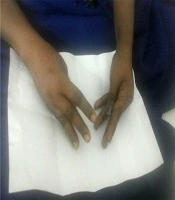
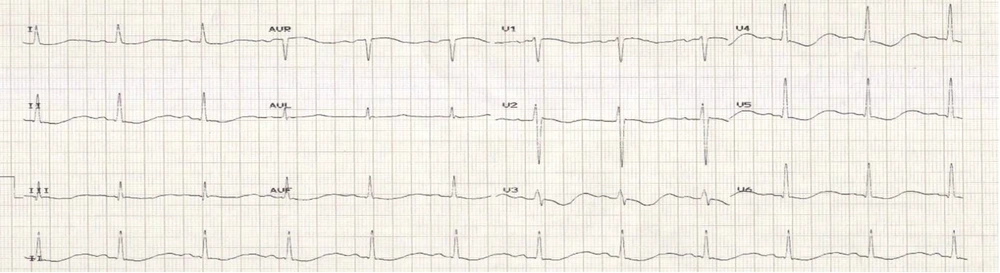
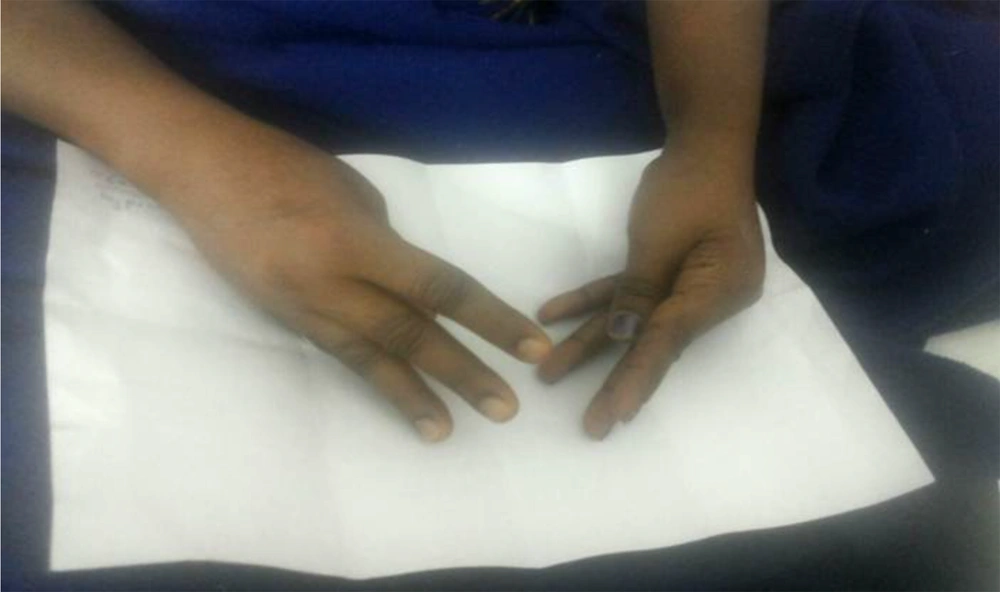
A 47-year-old homemaker presented to our hospital with complaints of weakness of both upper and lower limbs of one-day duration. The weakness was ascending, symmetric, and rapidly progressive in nature. There was no history of numbness or tingling sensation. There was no history of loss of consciousness, bowel or bladder involvement. There was no significant history of trauma and neck pain prior to the onset of the weakness. Her family gave no history of any drug intake or recent vaccination. They also could not recall any similar episodes in the past. She gave a history of intermittent fever, joint pains, and skin rash. She was evaluated in our outpatient department and was found to have evidence of anti-SSa and anti-SSb antibodies. A diagnosis of Sjogren’s syndrome was made, and she was started on hydroxychloroquine. When she presented to the emergency room her blood pressure, heart rate, and rhythm were normal. Her respiratory rate was 14 per minute, and she had flaccid quadriparesis. Her deep tendon reflexes were absent. No sensorimotor deficits were noted. As she was in impending respiratory failure, she was immediately intubated and put on mechanical ventilation. She was noted to have oral ulcers during her endotracheal intubation. Her initial laboratory parameters revealed severe hypokalemia (serum potassium -1.4 meq/L Normal range 3.5 - 5.1 meq/L) with hyperchloremic metabolic acidosis (Table 1). The ECG showed flattened T waves and prolonged QT interval (Figure 1). She was treated with intravenous potassium supplementation. She required more than 300 milliequivalents/day of elemental potassium in the first 48 hours to normalize her serum potassium levels. Her weakness started to improve as her serum potassium started improving. On the second day of admission, she developed a severe carpal spasm (Figure 2). This spasm was intermittent and lasted for more than 12 hours. A repeat ionized calcium was assessed, which was normal. A magnesium level was also assessed, which was low (0.9 mg/dL, normal range, 1.7 - 2,2 mg/dL) that was corrected with intravenous supplementation.
| Investigations | Case 1 | Laboratory (Normal Value) |
|---|---|---|
| Hemoglobin, mg/dL | 12.7 | 12.1 - 15.1 |
| Total count, /mcL | 6390 | 4000 - 11000 |
| Platelets, lakh/mcL | 2.19 | 1.5 - 4.5 |
| Serum creatinine, mg/dL | 0.8 | 0.6 - 0.9 |
| Serum sodium, mEq/L | 142 | 135 - 145 |
| Serum potassium, mEq/L | 1.4 | 3.5 - 5.1 |
| Serum chloride, mEq/L | 118 | 96 - 106 |
| Serum calcium, mg/dL | 8.26 | 8.1 - 10.6 |
| Serum albumin, g/dL | 2.7 | 3.5 - 5.5 |
| Corrected calcium, mg/dL | 10.1 | 8.5 - 10.2 |
| Ionised calcium, mmol/L | 1.3 | 1.1 - 1.35 |
| Serum phosphorus, mg/dL | 1.3 | 2.5 - 4.5 |
| Serum magnesium, mg/dL | 0.9 | 1.7 - 2.2 |
| Vitamin D3, ng/mL | 16.8 | 20 - 50 |
| Arterial blood gas | ||
| pH | 7.34 | 7.35 - 7.45 |
| Bicarbonate, mEq/L | 13 | 22 - 26 |
| Pa O2, mmHg | 84.2 | 80 - 100 |
| Pa CO2, mmHg | 24.7 | 35 - 45 |
| Thyroid profile | ||
| T3, ng/mL | 0.61 | 0.6 - 1.81 |
| T4, mcg/dL | 5.5 | 5 - 10.7 |
| TSH, mIU/L | 1.62 | 0.35 - 5.5 |
| Urine analysis | ||
| Specific gravity | 1.010 | 1.003 - 1.035 |
| pH | 8 | 4.7 - 7.5 |
| Albumin | 1+ | NIL |
| Sugar | NIL | NIL |
| Pus cells, /HPF | 2-3 | 0 - 5 |
The spasms persisted over 48 hours and resolved after the serum potassium and magnesium levels were stabilized over the next few days. She was started on oral potassium citrate supplementation.
The clinical presentation of hypokalemia, metabolic acidosis, and alkaline urine was suggestive of a distal renal tubular acidosis. Investigations for a probable secondary cause were done, and her antibodies against SSa and SSb came as strongly positive again (Her antinuclear antibody levels were negative).
Normocalcemic tetany and hypokalemic paralysis were diagnosed in a patient with Sjogren’s syndrome accompanied by distal renal tubular acidosis. She was started on low-dose steroids and hydroxychloroquine. She was discharged after a week. Currently, she is well and is on regular follow-up. She continues to take her potassium citrate, steroids, and hydroxychloroquine. Her electrolytes and divalent cation levels were normal in the last year and on follow-up a fortnight back.
3. Discussion
Hypokalemic paralysis, as a clinical presentation of Sjogren’s syndrome, is reported in the literature (5, 6). However, the combination of tetany (a form of neuromuscular irritability) and flaccid paralysis is rare in literature. One of the reasons for this is the presence of coexisting hypomagnesemia (associated with hypokalemia and chronic metabolic acidosis), which is often not tested (the forgotten cation). Hypomagnesemia has been shown to be prevalent in patients with hypokalemia and chronic metabolic acidosis (7). Our patient had both of these electrolyte and acid-base abnormalities. Hypomagnesemia has been shown to cause multiple neurological abnormalities, including tetany (8).
There is also literature to suggest that both periodic paralysis and tetany can be explained by the presence of hypokalemia (9, 10). The mechanisms by which hypokalemia causes tetany are all speculative. It is believed that extracellular potassium level alone is not responsible for neuromuscular irritability, but the ratio of the intracellular and extracellular potassium (Ki/Ke) and its transmembrane conductance plays a definitive role. A combination of low extracellular potassium levels and metabolic acidosis can cause severe intracellular potassium depletion and membrane depolarization (11). Also, potassium has a differential effect on muscles and peripheral nerves. Hypokalemia affecting muscles causes paralysis, whereas it causes tetany if it affects peripheral nerves preferentially (12).
Even though hypokalemia can theoretically present with hypotonic muscular paralysis and tetany, very few reports are available in the literature, especially in patients with Sjogren’s syndrome (13). We believe that the underlying tetany was secondary to hypomagnesemia as the symptoms were improved with magnesium supplementation.
3.1. Conclusions
This case illustrates the important role of electrolytes and divalent cations in neuromuscular manifestations in a patient with Sjogren’s syndrome. There is a complex interplay between fluid, electrolyte, and acid-base disturbances in patients with renal diseases with predominant tubular defects. It also clarifies that even though a single electrolyte/cation abnormality may be classically associated with the underlying disease, it is important to look for other abnormalities and try to correct them.