1. Context
Decompressive craniectomy (DC) has proven its worth in different scenarios (1-4) but still has three unresolved issues:
(1) It requires a second surgery in the form of cranioplasty with its monetary and logistic implications incredibly distressing for the elderly, economically underprivileged, and socially isolated.
(2) During the interval between the DC and cranioplasty, there is a disruption of Monro-Kellie doctrine, giving rise to problems like hydrocephalus and trephination syndrome (5, 6).
(3) There is an incongruity between the outcome in cases of traumatic brain injury (TBI) after DC even after satisfactory correction of brain shifts compared to the cases of malignant infarct (7).
We propose step-ladder expansive cranioplasty, a single-stage surgery, to solve all three drawbacks of DC. The resolution of the first two issues is evident if the bone can be placed back to cover the craniectomy defect in the immediate postoperative period. Our claim that DC, in isolation, can be responsible for grey cell injury and “expansion cranioplasty” can be of added benefit in preventing it needs elaboration.
2. Pathophysiological Basis of the Concept of Pressure Injury to Cerebral Cortical Grey Cells Following DC
2.1. Unique Anatomical Properties of Cerebral Blood Flow
All the cerebral capillaries are known to be perfused with blood at any given time (8). It has also been documented that almost every neuron in the brain possesses its own capillary (9). Structurally, cerebral arteries do not have external elastic lamina, and their adventitia is very thin in contrast to the systemic arteries (10). The cerebral cortex receives its perfusion from penetrating arterioles that descend approximately to a depth of 2.5 mm from the cortical surface, descending to the grey-white intersection (11). On the other hand, the deep white matter (DWM) arteries pass through the cerebral cortex with only a few branches to the cortex running straight through the DWM. These arteries are concentrated ventriculopetally to the white matter around the lateral ventricle. The adventitial sheaths of the subcortical and DWM arteries are relatively thicker than the cortical vessels (12).
2.2. DC Makes the Blood Supply of Cortical Grey Cells Vulnerable at the Region Underneath the Craniotomy Scalp Flap
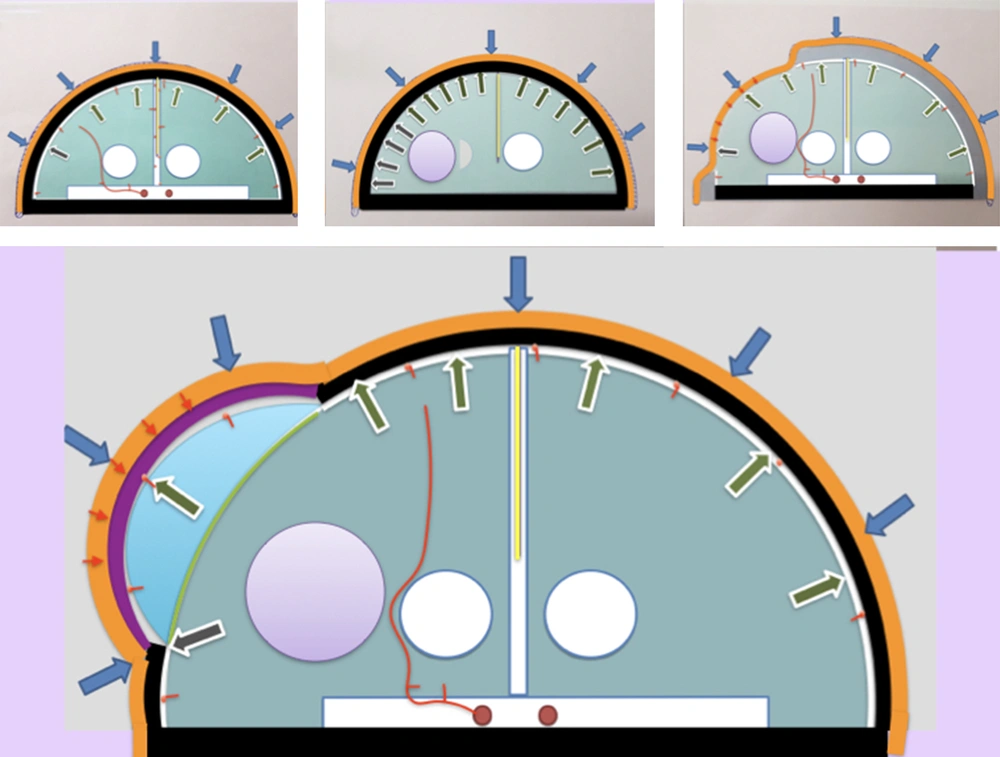
Brain rests protected from the physical forces of the outer environment by the cranium and CSF in which the brain and cerebral vasculature float (Figure 1A). The cerebral vessels enjoy protection from any direct compression due to the hydrostatic properties of the CSF in the subarachnoid space on the brain surface and fluid occupying Virchow-Robin spaces in the brain parenchyma. In the face of an intracranial space-occupying lesion or increasing brain volume secondary to any pathology, when no more shifting of the CSF or blood outside the cranium is possible to compensate for it, there is an exponential rise in ICP. Opening the lid with a DC is the only surgical solution (Figure 1B). However, DC takes the protective barrier of bony calvaria away. Dural opening, documented to be the most effective surgical step for ICP reduction, neutralizes the mechanical advantage enjoyed by the cerebral vessels of floating inside the CSF. This breaches the second line of defense. The pulsatile nature of the brain may act as a protection against cortical pressure injury by intermittently relieving the pressure. However, in the face of unresolved refractory raised ICP, even this may lose its effectiveness.
Diagrammatic representation of the concept with its pathophysiological basis. Orange line, scalp; black line, calvaria; purple line, cranioplasty construct; yellow line, falx cerebri; red line, artery; green area, brain parenchyma; blue area, volume expansion achieved at the craniectomy site; white area, CSF; purple area, intracerebral bleed/edema causing mass effect. Arrows represent the vector of forces: blue arrows, atmospheric pressure; green arrows, ICP; red arrows, tensile strength of the scalp; red dots, blood vessels; small dots, pial arteries supplying the outer 2.5 mm of the cortex; large dots, large cerebral arteries supplying DWM. A, Normal ICP with intact calvaria; B, Unilateral space-occupying lesion with resistant raised ICP; C, DC with volume expansion with compression of pial vessels by the antagonistic forces of ICP acting centrifugally and combination of atmospheric pressure and tensile strength of scalp acting centripetally; and D, Cranioplasty construct placed over the craniotomy defect protecting the underlying brain from the centripetal forces.
2.3. Possible Pressure Injury to Underlying Cortical Grey Cells
Decompressive craniectomy and various modifications of hinge craniotomy reported upon are based on the shared principle that the swollen brain can push the scalp flap, with or without a free-floating bone flap underneath it, to achieve the desired cranial volume expansion (13-16). Theoretically, compression of pial vessels by the antagonistic forces of ICP acting centrifugally and a combination of atmospheric pressure and tensile strength of scalp acting centripetally is a valid possibility at the scalp-parenchyma interface (Figure 1C).
2.4. Does It Really Matter?
Over the years, with CT scan and 1.5 T MRI being the only available neuroimaging modalities, our attention and efforts have entirely been on white matter changes and their correction. Decompressive craniectomy achieves it well. With the advancement in technology, we can now better document the loss of grey matter. Three critical aspects have come to light in this regard that deserve attention.
Firstly, there is always an associated loss of grey cells in TBI, with or without a documented contusion. This loss of grey cells may be widely dispersed and located away from the site of injury (17, 18). Secondly, documented damages to the white matter will often result from an undocumented grey cell loss proximally (19). Thirdly, even in mild TBI, extensive white matter injury escapes detection by commonly available neuroimaging modalities (18, 19). The overlying grey matter may be intact at the time of injury. There is no strong reason to think otherwise for severe TBI if it is true for mild TBI.
Therefore, the undamaged grey matter needs as much care and effort to safeguard it as is done for the “penumbra”, since the ultimate functional outcome will depend on the loss of both grey and white matters, which are most likely strongly interrelated. We can achieve this by devising surgical techniques to create a cranial volume expansion on their own, taking pressure off the brain surface while doing so. Thereby, cortical vessels will not have to withstand physical compression while ensuring reduction in ICP.
2.5. Solution
The dual aim of cranial volume expansion and resultant ICP reduction on the one hand and preventing any pressure on the cortical grey cells and its vasculature, on the other hand, can be achieved by fixing the cranioplasty construct after displacing it centrifugally from the craniectomy defect to obtain an assured cranial volume expansion. This is unlike all other modifications of DC, including the various hinge craniotomies where the brain is left to actively push against the combination of centripetal forces of the tensile strength of the scalp and atmospheric pressure (Figure 1D).
3. Evidence Acquisition
To begin with, there were three questions to be answered:
- How much volume augmentation is achieved by decompressive hemicraniectomy?
- Can the craniectomy bone flap be fashioned to give the same amount of volume augmentation as achieved by a classical decompressive hemicraniectomy?
- How to achieve scalp cover in the expanded cranium after an expansive cranioplasty?
3.1. Start from a Scratch
An extensive literature search in March 2014 revealed a limited number of studies that had reported upon the craniectomy surface area and the resultant volume augmentation and/or the lateral bulge of the scalp flap (20-22). The problem in drawing any inference from these studies was as follows:
- The flap displacement and volume expansion were measured from the level of the craniectomy margin using the Flint Method (23). Since there were no records of preoperative parameters in the existing studies, the actual expansion from the preoperative state could not be obtained.
- The craniectomy surface area and corresponding volume augmentation were not charted for each case in the studies, and lateral flap displacements were recorded only in the study by Cavusoglu et al. (21). Since the volume augmentation would be a product of craniectomy surface area and lateral flap displacement, it lacked crucial information.
A mathematical model was devised to decode the available data and determine an actual intracranial volume expansion achieved by the decompressive hemicraniectomy reported in the existing literature (Figure 2A). This model was based on two fundamental assumptions. Firstly, intracranial space is geometrically equivalent to a 1500 cm3 volume hemisphere, which works out to have an 8.945 cm radius (rounded off to 9 cm for calculations). The flat circular surface of the hemisphere represents the base of the skull. Secondly, it was also assumed that a circular area of the equal surface area could mathematically represent any craniectomy defect of any shape (circular, rectangular, or oval), having the same surface area (24).
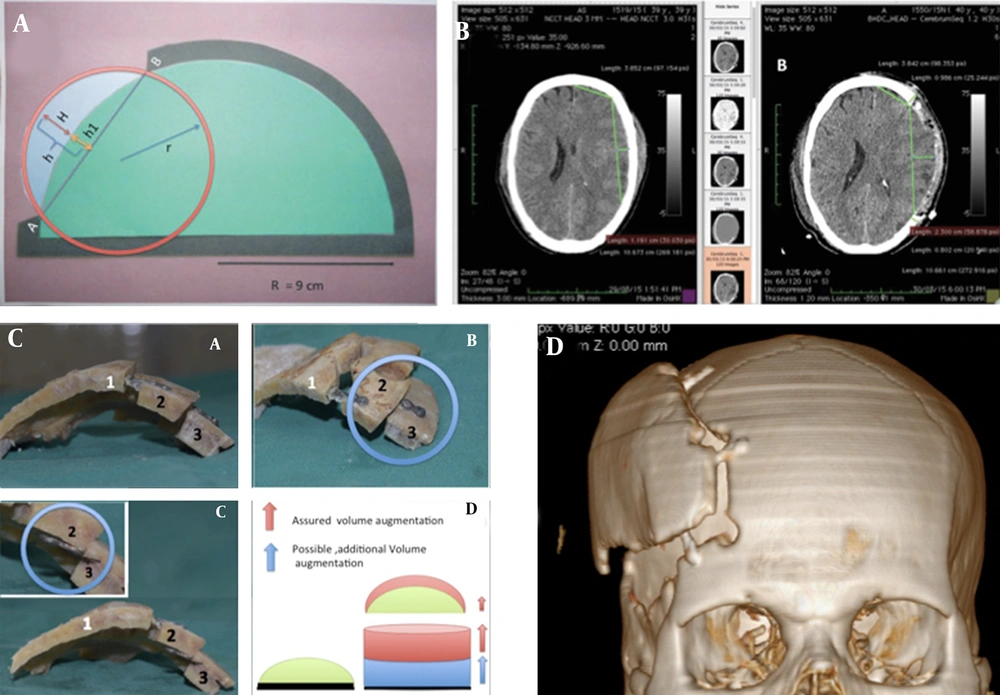
From drawing board to surgery: A, Mathematical model: A green half-circle with radius ‘R’ representing the brain. After volume augmentation of the part of the dural sac (represented by blue area), it is postulated to form part of an imaginary circle of radius r represented by the red circle. In this model, we found out ‘h’; the centrifugal displacement of the bone flap required before fixing to obtain adequate volume expansion; B, Feasibility study of pre and postoperative CT scans measuring the maximum projection of the dural out-pouching, which can be expected in the worst scenario and will require to be covered by the cranioplasty construct; C, Study on cadaveric model experimenting with different alternatives; and D, First surgery performed: Three-dimensional volumetric reconstruction images showing the step-ladder cranioplasty construct.
On our mathematical model, we found that a unilateral FTP decompressive hemicraniectomy of 12 cm × 8 cm in size and oval in shape (equivalent in surface area to a 9.79 cm diameter circle) has a surface area of 75.42 cm2 and the maximum distance from the line joining the craniectomy margins to the outer margin of pre-craniectomy unexpanded dura (or the inner table) is 1.45 cm.
Subsequently, my colleagues and I published an anatomical study, trying to validate the findings of the mathematical model on CT scan images (25). The findings of this anatomocal study closely replicated Cavusoglu et al. findings and our results in the previous study using a mathematical model (21, 24). In our anatomical CT scan-based study, the mean surface area of craniectomy defects was found to be 66.89 cm2. Measured from the craniectomy margin, the maximum projection of the dural out-pouching was 2.7 cm. This implied that we required the construction of expansive cranioplasty, which would achieve 1.6 cm of centrifugal displacement of the inner table of the free bone flap from its preoperative position (Figure 2B).
3.2. Pleasant Surprises
The mathematical model and the NCCT-based Anatomical study brought out another apparent fallacy. The findings of both studies indicated that though measured from the craniotomy margin, the height of the dural out-pouching (lateral or centrifugal scalp flap displacement) increases proportionally to the size of the craniectomy; but, for a given volume expansion, the centrifugal displacement of dura beyond its preoperative limit is inversely proportional to the craniectomy size. We call the volume expansion “apparent volume expansion” when measured from the craniectomy margin. The “true volume expansion” is the crescentic part of the brain that prolapses out of the craniectomy defect beyond the preoperative limit of the dura (Figures 1D and 2A).
At this stage of our research, we were under the impression that the height or centrifugal displacement of the cranioplasty construct was based solely on the thickness of the calvaria. With this perception, we went ahead with a cadaveric study trying to design various alternative construct designs with their unique pros and cons (Figure 2C). However, as we conducted the cadaveric study, we realized that the volume expansion was not much dependent on the bone thickness (26). There were two more important factors that would matter more while designing a cranioplasty construct. The first was the angulation of the titanium miniplates, and the second was the length of the bridging portion of the titanium miniplate connecting the bone flap and the cranium. By increasing the length of the bridging portion of the miniplates, the bone flap can be displaced more and more laterally, increasing and adjusting the volume expansion at will (Figure 2D).
3.3. Last Hurdle Before Clinical Implementation
Realizing that after volume augmentation, the cranium will have a proportionate increase in its surface area required to be covered. Leaving any open wound or suture-line under stress, with a risk of flap necrosis or breaking down over a craniotomy, would have disastrous consequences. Hence, we had to plan a solution before embarking upon applying the technique. It was planned to extend the rostral-most end of a standard question mark incision across the midline to create an inverse S-shaped scalp incision. We planned to raise two separate flaps. The first was to resemble a standard trauma flap to be turned laterally. The second subgaleal scalp flap was planned to be raised using the contralateral extension of the incision as a rotation flap medially. It was decided that if the flap appears to be under tension during the closure, a portion of the scalp wound on the contralateral side, away from the craniotomy site, will either be covered with an SSG or left for closure by secondary intension.
4. Results
We performed our first case of “Step-ladder Expansive Cranioplasty” for a case of traumatic brain injury with acute subdural hematoma in 2016 (27).
4.1. Operative Technique
- Position: Head tilted 60 degrees to the contralateral side; the operating table can be tilted for convenience if deemed necessary.
- Draping should keep the contralateral parietal eminence exposed.
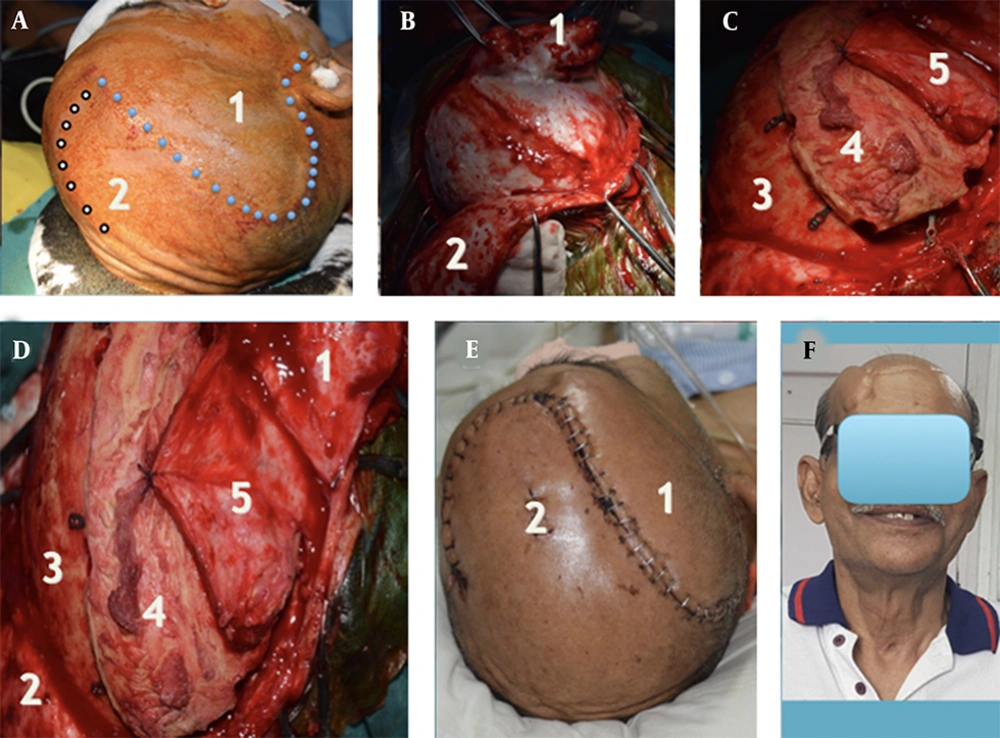
- Incision: A standard, reverse question mark incision is made extending from 1 cm anterior to the tragus, extending anteriorly to the hairline. The most anterior point of the incision is then connected to the most posterior point approachable, on the contralateral superior temporal line with a gentle curvilinear incision with convexity to the contralateral side; thereby, an inverse S-shaped scalp incision is made (Figure 3A).
Step-ladder expansive cranioplasty: The Procedure; A, B, C and D, Operative images. Blue dots represent the standard question mark incision line with an additional inverted S-shaped extension along the white dots to cater for a rotation flap. (1) The standard subgaleal scalp flap raised. (2) The medial subgaleal flap is used as a rotation flap during the closure. (3) Cranium at the margin of the craniotomy defect. (4) Craniectomy bone flap fixed to the outer table of the cranium with the titanium miniplates fixed to the inner table of the free bone flap with screws. (5) Temporalis muscle with temporalis fascia covering the temporal craniectomy defect. E, Postoperative photographs showing the S-shaped suture line: (1) and (2) the lateral and medial scalp flaps, respectively. F, The cosmetic outcome two years’ post-surgery showed the obvious “step-ladder” deformity.
Two separate scalp flaps are raised. A standard trauma flap is raised and turned laterally. A second subgaleal flap is then raised using the contralateral extension of the incision. This medial flap is used as a rotation flap subsequently during closure (Figure 3B).
A frontotemporoparietal (FTP) free bone flap of at least 12 cm × 8 cm in size is raised, and the squamous part of the temporal bone is nibbled off to the extent necessary.
Durotomy is done, and basic pathology is dealt with (in our case, the evacuation of acute SDH and achieving haemostasis).
We recommend an expansive duraplasty in all cases to ensure closure of the dural bag. We use a pericranial flap.
An assistant on the side table should prepare the bone flap, fixing titanium miniplates to the inner table of the free bone flap in four places. All the fixation sites should be on the part of the bone medial to the superior temporal line.
The margins of the bone flap should be drilled at the outer table to create a gentle slope on bone edges. Failing to do so may create an angulation of the scalp flap overlying it, jeopardizing vascularity.
Expansive cranioplasty creates abundant space underneath the bone flap. Dural edges need to be hitched at the margin to prevent epidural hematoma collection (we did not resort to this step for our index case).
The construct is placed back over the craniectomy defect as a cap. It is fixed with miniplates and screws on the outer table (Figure 3C), creating a single-step step ladder cranioplasty. The miniplates are fixed on the outer table of the cranium and the inner table of the free bone flap, thereby creating the elevation. The lateral displacement of the cranioplasty construct can be adjusted by shifting the points of fixation over the cranium laterally or medially.
Temporalis muscle with fascia is sutured to a thin rim of the temporalis muscle and fascia, left over the superior temporal line (Figure 3D).
The medial scalp flap is rotated laterally and sutured to the lateral scalp flap covering the cranioplasty construct. A wide subgaleal undermining is done, and the remaining wound on the medial side is closed in two layers (Figure 3E). During scalp closure, the following sequence is recommended:
- Suturing should commence from the pre-auricular region and continue to complete closure at the posterior part of the wound first.
- The scalp over the frontal bone is highly mobile. Subgaleal mobilization further eases the closure of the wound’s frontal part, which should be done next.
- The rotation flap should now be turned medially to complete the closure of the question mark incision.
- The principles of closure of a rotation flap should now be followed to close the residual wound.
4.2. Postoperative Follow-Up in the Index Case
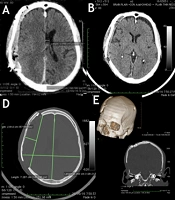
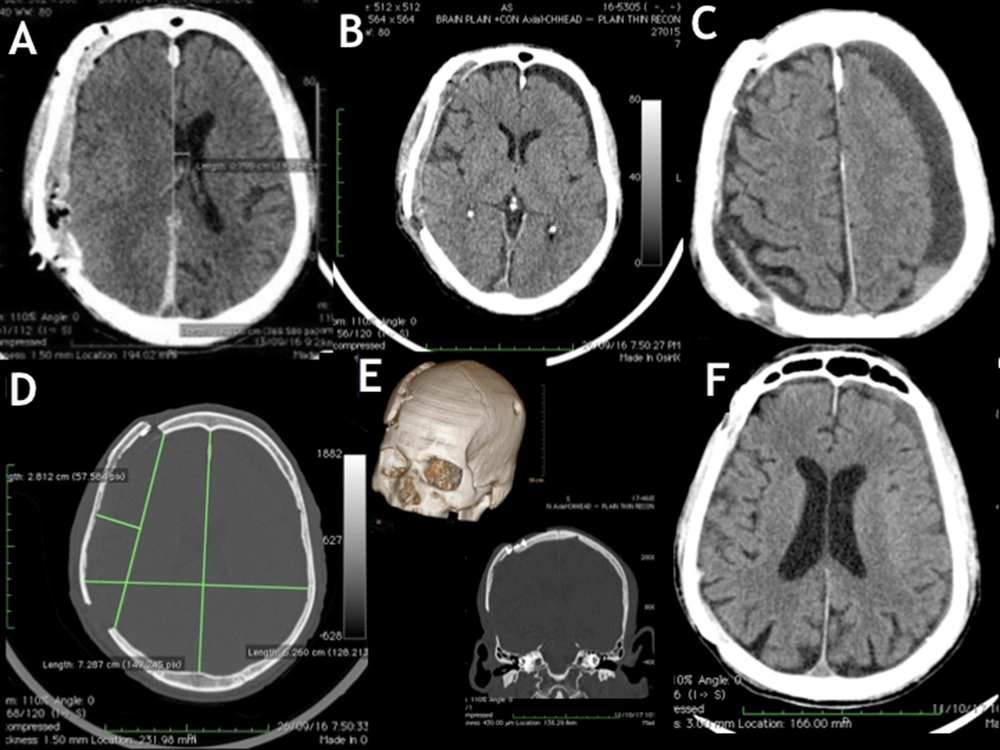
In the bone window of an axial NCCT image, the distance of the inner table at the level of the parietal eminence was found to have increased by 10.2 mm (72.8 mm on the affected side as compared with 62.6 mm on the normal side), when measured from the midline. The lateral limit of the dural out-pouching was 28.1 mm when measured from the craniectomy margin (Figure 4D). Immediate postoperative neuroimaging revealed an extradural collection resolved by the third postoperative week, leaving behind a CSF hygroma over the contralateral FTP region instead (Figure 4A). The individual presented in the fourth postoperative month with Generalized Tonic-Clonic Seizures (GTCS). On evaluation, he was detected to have developed chronic Frontoparietotemporal SDH (Figure 4C). It was managed with a single burr hole and drainage under local anesthesia. The subsequent recovery was uneventful. Two years after surgery, he had an obvious step deformity in the scalp (Figure 3F); he was happy to cover under the cap and was not keen to undergo any corrective surgery for the same. There was no radiological evidence of implant failure (Figure 2F). There was a near-complete resolution of the CSF hygroma, and the brain appeared to have expanded to occupy nearly the entire available space in the volume augmented cranium (Figure 4F). We lost this extremely valuable patient of ours due to the COVID-19 pandemic.
Follow-up CT scans: A, Immediate postoperative images revealed an extradural collection under the cranioplasty construct; B, Both the extradural collection and midline shift resolved in two weeks, while there was contralateral CSF hygroma; C, Contralateral chronic SDH with significant mass effect evident in 16 weeks postoperative NCCT image; D, Axial section showing an increase in the biparietal diameter of 10 mm on the cranioplasty side. The outer margin of the brain was measured to be 2.81 cm beyond the level of the craniectomy defect; E, Step ladder expansion was achieved by fixing the cranium and the bone flaps on two opposite surfaces of titanium miniplates. Volumetric 3D reconstruction also shows the contralateral burr hole; F, Eighteen months’ post-surgery, NCCT of the head showed near-complete resolution of CSF hygroma.
4.3. How Does It Fare as Compared to Hinge Craniotomies?
We do not have the audacity to compare our surgical technique, at its nascent state, with the wide variety of well-established single-stage techniques, but there are two points worth noting.
At least two neurosurgical centers have published small case series with similar but minor tweaking techniques with satisfactory results (28, 29).
Most hinge craniotomies have been reported to have achieved a lateral out-pouching of the dural bag through the craniectomy defect (5 mm), hence indirectly the volume expansion (16, 17, 19); only half of that (> 9 mm) reported for DC (25, 27, 30).
4.4. What Is Holding Us Back
Apart from the fact that it can logically resolve several neurosurgical enigmas (6), there is no documented proof to authenticate our hypothesis of cortical pressure injury happening at the craniectomy defect site.
Exact cranial volume expansion required to be achieved, and hence, the ideal or acceptable range of centrifugal displacement of the expansive cranioplasty construct is yet to be established. Also, there is likely to be a difference between different pathologies being dealt with. A consensus is required to be achieved after a series of focused research for the missing data. Lesser than adequate volume expansion may prove detrimental because fixing the bone plate obviates any possibility of further volume expansion. A disproportionate increase in cranial volume, on the other hand, is likely to lead to its own set of complications.
5. Discussion
Decompressive craniectomy achieves ICP reduction by creating cranial volume expansion. Maximum volume expansion achieved so far has been in the range of 107.2 cc to 157.6 cc (20-22) with a proportionate limit to ICP reduction. The extent of ICP reduction directly bears the outcome (31). In our endeavor to save the imminent threat to life, DC leaves Monroe-Kellie doctrine disrupted and cerebral cortical surface exposed to various physical forces and consequences of both (6). Step-ladder expansive cranioplasty is the ONLY procedure that can achieve cranial volume expansion and hence ICP reduction of desired proportions, guided by peroperative ICP monitoring during closure. After surgery, this technique also restores the Monroe-Kellie doctrine and puts the brain back into cranial protection.
5.1. Conclusions
The step-ladder expansive cranioplasty offers a single-stage alternative that takes the centripetal pressure exerted by the combination of the tensile strength of the scalp and atmospheric pressure off the brain surface while achieving an assured augmentation of intracranial volume that can be optimized on a case-to-case basis, based on our future understanding of the subject. Though acceptable to many as a single step surgery, in those uncomfortable with the cosmesis, taking down the construct at a later date is likely to be easier than a routine cranioplasty. Not requiring a host site for subcutaneous pocket or elaborate arrangements of bone preservation is an obvious advantage. In an emerging concept, neurosurgeons can apply their ingenuity to create unending possibilities. For one applying an absorbable material in place of titanium miniplates would allow the bone flap to settle after a stipulated period and change the dynamics to our advantage.
Full-scale clinical trials can begin to evaluate the procedure for reproducibility as soon as a consensus is achieved regarding the ideal or acceptable range of volume expansion necessary to take care of raised ICP in different cases scenarios or the hypothesis of cortical pressure injury be established.