1. Background
Stroke is the most common cause of mortality and permanent disability worldwide (1). Cell death and functional loss after stroke have been associated with excitotoxicity, calcium dysregulation, oxidative stress, inflammation, and pre-apoptotic stimuli (2). To develop new treatments to prevent stroke and promote recovery from stroke, it is essential to investigate the mechanisms of pro-inflammatory responses associated with ischemia (3). The level of interleukin-6 (IL-6) has been shown to increase following ischemia and stimulate the expression of adherent endothelial cells (4), which eventually block microvessels and cause cell damage (5). However, study of cytokines in the nervous system injuries is challenging due to their complex pre- and anti-inflammatory activities through interdependent pathways, such as the neurotrophic properties of IL-6 (4-6).
The adverse effects of inflammatory mediators on cell proliferation, migration, and survival (7) may be prevented by neurotrophic factors, which are vital components in neuronal survival, growth, differentiation, and plasticity (8). Nerve growth factor (NGF), along with other trophic factors, is widely found in the brain, including the hippocampus. It positively regulates neurogenesis, synaptogenesis, and neurovascular integrity (9). It seems that NGF has a direct effect on releasing glutamate in the hippocampus (10), which is the most abundant excitatory neurotransmitter in the brain, involved in neuronal survival and cell death (11).
Moreover, glutamate plays an important role in long-term neuronal excitability, synaptic organization, and neuronal migration during neuronal development and maintenance (12). Nevertheless, significant amounts of glutamate can have neurotoxic effects; therefore, excitotoxicity in ischemic injuries can exacerbate cellular loss, especially in the hippocampus (13). It has been shown that increasing the level of endogenous adenosine through adenosine A2A receptors inhibits the function of inflammatory cells (14), decreases glutamate release (15), and increases neurotrophic factors, such as NGF (16). Consequently, endogenous adenosine is recognized as a potent physiological mediator, regulating various physiological processes through adenosine receptors (17) that may be potential therapeutic targets for stroke, as well. Previous studies have suggested the positive effect of endogenous adenosine on functional recovery after brain ischemia; however, the effect of exogenous adenosine has not been investigated yet.
Additionally, the regulation of inflammatory, excitotoxic, and trophic processes following an ischemic injury can be influenced by physical activity. Physical exercise shows neuroprotective activities against ischemic stroke by reducing primary risk factors, such as hypertension, dyslipidemia, diabetes, sedentary lifestyle, and obesity (18). Besides, preconditioning with endurance training on a treadmill can lead to neuroprotection and improved neuronal survival in ischemia/reperfusion injuries, resulting in reduced infarct volume and improved functional recovery (18, 19).
Previous studies have demonstrated that exercise preconditioning facilitates functional recovery after strokes by decreasing inflammatory responses, such as IL-6 expression, inhibiting glutamate overactivation, and increasing the NGF expression. Besides, modification of the molecular cascade seems to be associated with reduced neuronal apoptosis, restored function of the blood-brain barrier (BBB), and enhanced angiogenesis (20, 21). Nevertheless, endurance training on a treadmill may induce physiological and psychological stress (22) and increase the release of stress hormones (23), suggesting the dual role of training in pre- and post-ischemic stroke inflammatory responses (21).
2. Objectives
Since it is essential to identify more potent treatments for cerebral ischemia (24), the present study aimed to evaluate the effect of endurance training and exogenous adenosine infusion, as a preconditioning intervention, on inflammatory, excitotoxic, and neurotrophic processes in a rat model of stroke. By inducing an ischemia/reperfusion injury by transient common carotid artery clamping, the results indicated the synergistic effects of preconditioning with physical exercise and exogenous adenosine infusion on neuronal and functional integrity.
3. Methods
3.1. Animals
Fifty-six adult male Wistar rats (weight: 250 - 300 g) were purchased from Pastor Institute of Iran (Tehran, Iran). The rats were housed in standard Plexiglas cages under controlled ambient conditions (22 - 24°C, 48 - 55% humidity) in a 12: 12 h light/dark cycle with ad libitum access to food and water. The rats were randomly divided into five groups: control (group 1, n = 8); ischemia (group 2, n = 12); endurance training + ischemia (group 3, n = 12); adenosine infusion + ischemia (group 4, n = 12); and endurance training + adenosine infusion + ischemia (group 5, n = 12). The animals were included to study based on weight, age, and sex. The exclusion criteria were diseases and lack of racial or physiological homogeneity. Also, animals were excluded if they had injuries during exercise or expired after cerebral ischemia. In the experimental groups, factors, such as performing aerobic exercises, lack of injury during exercise, and lack of sudden weight change, were examined; if these conditions were not met, the animals would be eliminated from the study (Appendix).
3.2. Endurance Training and Physical Exercise Protocol
The rats in the training group were habituated to treadmill walking for 10 - 15 minutes at a speed of 15 m/min at zero slope in three alternating sessions per week before the main training sessions. Next, an eight-week endurance training protocol was applied for training the rats on a 10-line treadmill in five sessions per week. The training program started at 18 m/min for 20 minutes at zero slope in the first week. The duration, intensity, and treadmill slope were increased gradually; in the eighth week of training, the animals underwent a 50-minute training at a speed of 30 m/min at a 10° slope (25). After completing the exercise protocol and 24 hours of rest, cerebral ischemia/reperfusion surgery was performed.
3.3. Adenosine Treatment Protocol
In this study, 3 mg/mL of adenosine (Adenocard & Adenoscan) was purchased from the Faculty of Pharmacy of Tehran University of Medical Sciences, Tehran, Iran. The adenosine-treated groups received 0.1 mg/mL/kg of adenosine daily at the beginning of each week of endurance training. Adenosine was infused intraperitoneally slowly over one minute (26) at three hours after endurance training; the rats received eight doses of adenosine in total. In the adenosine-treated groups, cerebral ischemia/reperfusion surgery was performed one week after the final adenosine injection.
3.4. Cerebral Ischemia/Reperfusion Surgery
To induce transient cerebral ischemia, the rats were anesthetized by an intraperitoneal (IP) injection of 50 mg/kg of ketamine and 5 mg/kg of xylazine (Merck, Germany) (27). The vagus nerve was carefully separated from the carotid arteries. Next, both the common carotid arteries were clamped with a microsurgical clip for 45 minutes, after which the blood flow was allowed to reperfuse the carotid plate freely (26). Reperfusion of the carotid arteries was confirmed by visual examination. During surgery, the rectal temperature of the animals was maintained at 36.5 ± 0.5°C, using a heating system. The animals were kept separately for 48 hours after surgery with ad libitum access to water and food to facilitate recovery (28).
3.5. Tissue Preparation
The rats were deeply anesthetized at 48 hours after the induction of ischemia with a combination of 50 mg/kg of ketamine and 5 mg/kg of xylazine (Merck, Germany) (27). The brain tissue was immediately dissected and placed on ice and then frozen in liquid nitrogen. Next, the brains were embedded in paraffin blocks, and coronal sections with a thickness of 7 µm were prepared with a microtome for staining. The remaining tissue samples were stored at -80°C for gene expression assays.
3.6. Cresyl Violet (Nissl) Staining
Cresyl violet (Nissl) staining was used to identify the basic structure of healthy neurons and necrotic neurons. For staining, slices with a thickness of 7 μm (three slices per animal) were transferred to silane-coated slides and stained with a 0.1% Cresyl violet acetate solution. Next, the slides were dried and covered with Entellan (Sigma-Aldrich, USA). They were then visualized under a light microscope (AX-70 Olympus, Japan) at 400 × magnification, and cell counting was performed using an imaging software along a length of 400 μm in the hippocampal CA1 region. Only irregular and dark cells with unidentifiable nucleus and nucleolus were counted as dead cells.
3.7. RNA Extraction
RNA extraction from the tissue was performed in all groups, according to the manufacturer's protocol (Qiagen, Germany). Initially, 200 - 300 µL of Kiazol QIAzol (QIAGEN, Germany) was added to the hippocampal tissue and kept at -80°C for 24 hours. After 24 hours, the plaque in the Cryotube was crushed in a semi-frozen state with a micropipette and pipetted slowly. Next, about 100 µL of chloroform was added to the sample to lyse the cells; the solution was left in contact with the cells for about one minute and then centrifuged at 12,000 rpm for 10 minutes.
After centrifugation, 1 mL of isopropanol was poured onto transparent RNA and stirred for one minute. The samples were then centrifuged at 12,000 rpm for 10 minutes. Next, the supernatant was discarded, and 1 mL of 70% alcohol was added. After vortexing, the mixture was centrifuged at 7,500 rpm for 10 minutes. The supernatant was discarded with a sampler, and the plaque was dried in a microtube. To dissolve RNA, 20 µL of distilled water (60°C) was poured onto the plaque in the microtube. It was then pipetted slowly and placed on a 60°C plate for five minutes. The extracted RNA was stored at -80°C until further use.
3.8. cDNA Synthesis
After extraction of high-purity RNA from all samples, cDNA synthesis was performed according to the manufacturer's protocol (Fermentas, USA). For reverse transcription reactions, all primers designed for the genes were analyzed, and gene expression analysis was performed by a quantitative real-time polymerase chain reaction (qRT-PCR) assay. The expression ratio of genes was also evaluated by the comparative CT method (ΔΔCT) (29) (Table 1).
| Genes | Primer Sequences |
|---|---|
| IL-6 | Forward: AGGCAGAGTCATTCAGAGC |
| Reverse: CATTGGTAGTTGGGGTAGGA | |
| Glutamate | Forward: TCCTCCCTCTCATCATTTCCA |
| Reverse: CAGGATGACCCCCATCACA | |
| NGF | Forward: GCC TGT TTG TCG TCT GTT GT |
| Reverse: GCC CCG AAT CCT GTA GAG AG | |
| A2A | Forward: TCT AAA TGC TGG GAG GTC AA |
| Reverse: CTC ACG GTG GTC CTT TGT TG | |
| GAPDH | Forward: CAT ACT CAG CAC CAG CAT CAC C |
| Reverse: AAG TTC AAC GGC ACA GTC AAG G |
3.9. Statistical Analysis
Data are reported as mean and standard deviation (SD). Shapiro-Wilk test was performed in SPSS Version 16.0 for Windows (SPSS Inc., USA) to verify the normal distribution of data. One-way ANOVA test was also used to compare differences between the groups, followed by Bonferroni post-hoc correction test if there was a significant difference. The significance level was set at P ≤ 0.05. Also, to estimate the sample size of this experimental study, previous studies were used to find if the intervention was effective. The sample size was calculated using G*Power software.
3.10. Ethical Statement
This study was performed according to the guide for the care and use of laboratory animals, published by the US National Institutes of Health (NIH Publication No.: 85–23, revised in 1996). The study protocol was also approved by the local ethics committee (IR.IAU.SRB.REC.1397, 134). All efforts were made to minimize animal suffering and reduce the number of animals used.
4. Results
4.1. Cell Death
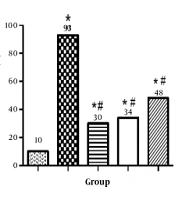
Based on the results, endurance training preconditioning reduced ischemia/reperfusion-induced cell death in the CA1 region of the hippocampus. The Nissl staining revealed that in the CA1 region, ischemia led to the irregular shape and dark color of the cells, with their nucleus and nucleolus frequently unrecognizable, which is indicative of necrotic cell death (Figure 1). Cell death in the CA1 region was more significant in group 2 compared to group 1, as 93% of the cells were necrotic (P < 0.05). Based on the results, the number of necrotic neurons in group 3, group 4, and group 5 decreased significantly compared to group 2 (P < 0.05 for all).
I, cresyl violet staining of the hippocampal CA1 region; A, non-lesion control group; B, ischemia only group; C, endurance training (ET) + ischemia group; D, adenosine + ischemia group; E, endurance training (ET) + adenosine + ischemia group. The ischemic lesion induced a significant rate of necrotic cell death. Note that endurance training and adenosine alone or in combination visibly reduced the rate of cell death (400x magnification); II, comparison of mean percentage of necrotic cells in the hippocampal CA1 region. All groups except for the controls received a transient cerebral ischemic lesion induced by common carotid artery occlusion. Note that cell death was significantly reduced by exercise and adenosine alone, and synergistic effects of both treatments slightly attenuated their benefit [symbols indicate significances: * significant difference compared to untreated controls group (P < 0.05); # significant difference compared to ischemia only group (P < 0.05)].
4.2. Gene Expression
The relative expression of IL-6 gene was significantly different between the groups. The IL-6 gene expression decreased in group 3, group 4, and group 5 by 8136, 1077, and 13 folds, respectively, compared to group 2 (P < 0.05 for all). The relative expression of glutamate gene also decreased in group 3, group 4, and group 5 by 19, 12, and four folds, respectively, compared to group 2 (P < 0.05 for all). On the other hand, the relative expression of NGF gene increased by seven, four, and two folds in group 3, group 4, and group 5, respectively, compared to group 2 (P < 0.05 for all). Finally, the relative expression of A2A gene increased by six and three folds in group 3 and group 4, respectively, compared to group 2 (P < 0.05 for all) (Figure 2).
Impact of endurance training pre-conditioning on IL-6, glutamate, NGF, and A2A gene expression. Impact of endurance training pre-conditioning on A, IL-6 (P < 0.05); B, glutamate (P < 0.05); C, NGF (P < 0.05); and D, A2A gene expression (P < 0.05) after induction of ischemia in different groups. Note that, out of all therapies, endurance training had the largest effect on gene expression patterns following ischemia, while adenosine increased expression of NGF and A2A and reduced expression of IL-6 and glutamate genes. Synergistic effects were rather negligible. Symbols indicate significances [* significant difference compared ischemia only group (P < 0.05)].
5. Discussion
According to the present study, after 45 minutes of cerebral ischemia/reperfusion, 93% of hippocampal CA1 cells were necrotic. Presumably, this cell death is associated with excessive glutamate release and excitotoxicity by overactivation of receptors, to which the hippocampus is particularly vulnerable (12). Besides, inflammatory factors, such as IL-6 and tumor necrosis factor-α (TNF-α), potentially contribute to ischemia-induced neuronal death, as increased inflammatory cytokine release commonly occurs in stroke and brain injury (4). In the present study, glutamate and IL-6 gene expression significantly increased in group 2.
The lack of effective therapies for stroke patients, despite promising preclinical findings, has prompted extensive investigations of molecular pathways linked to cell death (30). Adverse responses to inflammatory mediators in various stages after ischemia can explain the failure of clinical strategies (31). Endurance training represents an effective preventive, and even partially therapeutic strategy, by reducing the risk factors and protecting neurons against ischemia/reperfusion injury (32). Moreover, it has been reported that endurance training produces endogenous neuroprotective effects that will promote neuronal survival following an ischemia-induced damage (18, 19).
Studies have shown that endurance training preconditioning can decrease glutamate release and overexpression of glutamate receptors, leading to excitotoxicity resistance and reduced post-stroke brain injury (20). It has been also shown that preconditioning with endurance training increases the expression of glutamate transporters, which in turn reduces cell death following cerebral ischemia by increasing glutamate re-uptake and clearance (33). In line with previous findings, the present study showed that endurance training preconditioning significantly diminished glutamate gene expression.
The glutamate gene expression and cell death significantly reduced in the endurance training group, as well as adenosine infusion and endurance training/adenosine infusion/ischemia groups. In another study, a significant and rapid increase in the level of adenosine after ischemia/reperfusion attracted the researchers’ attention to this purine for therapeutic use (34). Adenosine is an endogenous neural regulator with neuroprotective properties by regulating cell proliferation and survival. Moreover, adenosine may limit cell death through inhibition and reduction of molecular events, such as reduced glutamate release and inhibition of inflammatory responses (35). In the present study, the Nissl staining showed that adenosine injection significantly reduced ischemia-induced cell death in the hippocampal CA1 neurons.
The present study revealed that endurance training and adenosine infusion, either alone or in combination, significantly decreased the expression of IL-6 compared to the ischemia group. Recently, Chio et al. (2017) argued that the role of endurance training preconditioning in modulating inflammatory responses after ischemic injury was not well understood (36). Cross-sectional studies have suggested that regular endurance training plays a protective role against inflammatory diseases (37). This effective protection may result from the release of IL-6 (38), which can play a neuroprotective role in brain ischemia by protecting neurons and inhibiting glutamate release (39). It may also trigger NGF secretion by stimulating astrocytes that can improve the survival of neurons following a brain injury (40).
Moreover, the level of NGF seems to increase in inflammatory and neuropathic pain states (41). Although, as previously stated, the dual role of IL-6 after a brain injury is not well understood, methodological differences that affect the dual role of this cytokine may be influential (42). Nevertheless, the results of the present study showed the significant effects of endurance training preconditioning, adenosine infusion, and their combination on increasing the level of NGF expression.
Previous studies have reported that adenosine reduces glutamate gene expression from glial cells through adenosine A2A receptors (15) and plays a protective role against oxidative damage (43). Since adenosine production is dependent on the amount of adenosine triphosphate (ATP) catalysis, the level of adenosine increases under stress conditions, such as endurance training or increased use of ATP over time. It seems that adenosine plays an important role in the complex adaptation of the body to endurance training; because of its rapid production, it is considered an ideal molecular agent for many regulatory mechanisms (44).
Besides, ischemic conditions increase the demand for energy and cellular oxygen and subsequently, increase adenosine levels with potentially protective effects (45). However, the mechanisms of adenosine depend on its effect on adenosine receptors, such as the strength and amount of binding to receptors, which can also produce various effects, because the A2A receptor, as one of the most important adenosine receptors, has a wide distribution in the brain and regulates many physiological processes (17). In the present study, cell death and A2A receptor expression increased in the adenosine infusion groups. However, its role in neurodegenerative processes is controversial due to the activity of A2A receptor as a mediator of both potential neuroprotective and neurotoxic effects. More recent studies reported that activation of this receptor may play a protective role against neurodegenerative diseases, such as Alzheimer's disease, Parkinson's disease, and ischemia (46).
Additionally, A2A facilitates neurotransmitter and synaptic transmission in the hippocampus (47). However, there is insufficient information regarding the signaling effects of endurance training on A2A in the brain tissue. Recent studies have shown a close relationship between A2A signaling and neurotrophic factor expression in neurons, as interactions between A2A and neurotrophic effects are effective in controlling the brain’s protective responses to cerebral ischemia (48).
Considering the vital role of A2A receptors in improving the neurotrophic activity of the hippocampus (16), tyrosine kinase receptors activate specific signaling pathways, and regulate neuronal release by A2A expression (39). The present study also showed that A2A expression increased significantly, with a higher NGF expression in adenosine-treated and endurance training groups. Moreover, A2A plays an important role in improving memory and learning (49) and increasing the sensory-motor function by promoting the release of neurotransmitters (26). One of the hallmarks of adenosine effect on target tissues is activation of adenosine receptors. The present study revealed that the protective effect of adenosine in the injured hippocampus is related to the activation of A2A receptors, resulting in decreased cell death factors and increased NGF expression. However, further studies are needed to identify the processes downstream the signaling cascade.
Some limitations of stroke models include the risk of hemorrhagic events, moderate recanalization rates, and hyper/hypothermia. Hypothalamic damage always occurs in animal models of stroke, whereas it rarely occurs in human strokes. Also, hypothalamic ischemia produces a hyperthermic response in rats, which may affect further analyses. Hypothalamic damage is also observed in rats after stroke; however, the surface/volume ratio of the damaged hippocampus regions leads to temperature loss in the postoperative period in rats.
5.1. Conclusions
The results of the present study indicated the most significant reduction in inflammatory processes and neuronal growth in the group of endurance training alone. Nevertheless, analysis of treatment with adenosine infusion and adenosine infusion-exercise combination in the experimental groups indicated the intensified effect of adenosine-exercise combination on increasing neuronal resistance and cell death.
![I, cresyl violet staining of the hippocampal CA1 region; A, non-lesion control group; B, ischemia only group; C, endurance training (ET) + ischemia group; D, adenosine + ischemia group; E, endurance training (ET) + adenosine + ischemia group. The ischemic lesion induced a significant rate of necrotic cell death. Note that endurance training and adenosine alone or in combination visibly reduced the rate of cell death (400x magnification); II, comparison of mean percentage of necrotic cells in the hippocampal CA1 region. All groups except for the controls received a transient cerebral ischemic lesion induced by common carotid artery occlusion. Note that cell death was significantly reduced by exercise and adenosine alone, and synergistic effects of both treatments slightly attenuated their benefit [symbols indicate significances: * significant difference compared to untreated controls group (P < 0.05); # significant difference compared to ischemia only group (P < 0.05)]. I, cresyl violet staining of the hippocampal CA1 region; A, non-lesion control group; B, ischemia only group; C, endurance training (ET) + ischemia group; D, adenosine + ischemia group; E, endurance training (ET) + adenosine + ischemia group. The ischemic lesion induced a significant rate of necrotic cell death. Note that endurance training and adenosine alone or in combination visibly reduced the rate of cell death (400x magnification); II, comparison of mean percentage of necrotic cells in the hippocampal CA1 region. All groups except for the controls received a transient cerebral ischemic lesion induced by common carotid artery occlusion. Note that cell death was significantly reduced by exercise and adenosine alone, and synergistic effects of both treatments slightly attenuated their benefit [symbols indicate significances: * significant difference compared to untreated controls group (P < 0.05); # significant difference compared to ischemia only group (P < 0.05)].](https://services.brieflands.com/cdn/serve/3170b/bb7f490b9bae05daecb2a3756f1f9e727fdfac01/ans-119236-g001-F1-preview.webp)
![Impact of endurance training pre-conditioning on <i>IL-6</i>, glutamate, <i>NGF</i>, and <i>A2A</i> gene expression. Impact of endurance training pre-conditioning on A, <i>IL-6 </i>(P < 0.05); B, glutamate (P < 0.05); C, <i>NGF</i> (P < 0.05); and D, <i>A2A</i> gene expression (P < 0.05) after induction of ischemia in different groups. Note that, out of all therapies, endurance training had the largest effect on gene expression patterns following ischemia, while adenosine increased expression of <i>NGF</i> and A2A and reduced expression of <i>IL-6</i> and glutamate genes. Synergistic effects were rather negligible. Symbols indicate significances [* significant difference compared ischemia only group (P < 0.05)]. Impact of endurance training pre-conditioning on <i>IL-6</i>, glutamate, <i>NGF</i>, and <i>A2A</i> gene expression. Impact of endurance training pre-conditioning on A, <i>IL-6 </i>(P < 0.05); B, glutamate (P < 0.05); C, <i>NGF</i> (P < 0.05); and D, <i>A2A</i> gene expression (P < 0.05) after induction of ischemia in different groups. Note that, out of all therapies, endurance training had the largest effect on gene expression patterns following ischemia, while adenosine increased expression of <i>NGF</i> and A2A and reduced expression of <i>IL-6</i> and glutamate genes. Synergistic effects were rather negligible. Symbols indicate significances [* significant difference compared ischemia only group (P < 0.05)].](https://services.brieflands.com/cdn/serve/3170b/e30dbc7add61be150b3d24bb29554b01c3e4a264/ans-119236-i001-F2-preview.webp)