1. Context
Head trauma is one of the most costly sections of health care in European countries. The World Health Organization estimated that approximately 90% of mortality due to trauma occurs in low- and middle-income countries (1-3). Each year, about 1.5 million people die due to brain trauma and about 10 million people are hospitalized due to this type of traumas (4). Preventive measures seem to play an important role in managing treatment and preventing further complications in these patients (5).
Attention to patients is essential (6-9). Chronic subdural hematoma is a common hemorrhage in the intracranial space, consisting of blood serum and decomposed blood products. Blood in the subdural space leads to an inflammatory reaction and the cascade progression of the disease. This type of hematoma may be due to trauma, as the most important and common cause, coagulation disorders, arachnoid cyst, vascular disorders, metastatic cancers, and brain meningioma (10, 11). Chronic subdural hematoma (CSDH) is an important hemorrhage that occurs after head trauma, which is considered the most important treatable cause of death and disability in trauma victims in almost half of the patients injured with severe trauma (12). Surgery is one of the most important treatments used for these patients. On the other hand, various medicines, such as ACE inhibitors (13), steroids (14), tranexamic acid (15), and herbal medicines (16), are used along with surgery.
Tranexamic acid (TXA) belongs to the family of lysine-derived antifibrinolytics. TXA requires a simple molecular breakdown in the liver to be metabolized and has a high renal excretion (3). TXA is an anti-fibrinolytic agent and a synthetic lysine-derived reducing hemorrhage through reversible competition for the lysine junction in plasminogen with fibrin. This medicine further inhibits the activation of plasminogen and prevents the destruction of fibrin by plasmin (17, 18). The TXA antifibrinolytic power is higher than the existing antifibrinolytic medicines while it has the fewest side effects. TXA does not increase thromboembolic complications such as deep vein thrombosis and pulmonary embolism (19, 20).
Medication can improve patients' condition (3, 21, 22). Antifibrinolytic medicines improve homeostasis, reduce hemorrhage, and consequently reduce the need for blood transfusions. These medicines have been addressed in various surgeries such as cardiovascular, liver, and orthopedic surgeries (19, 23-25). One of these medicines is TXA, the effects of which have been shown in various surgeries (26).
2. Objectives
Given the importance of identifying an effective medicine for reducing the status of subdural hematoma (SDH), this study aimed to determine the effect of TXA on SH treatment using the SR method.
3. Methods
3.1. Study Protocol
This SR study was conducted to evaluate the effect of tranexamic acid on SH treatment using articles published from 2000 to 2020.
3.2. Search Strategy and Study Selection
The search was performed by two researchers who were dominant in various types of SR studies and specialized discussion of neurosurgery. Table 1 presents the searched databases. The search method was according to PICO criteria, which are presented in Table 2 (Tables 1 and 2).
| Status of Included Databases | Name of Database |
|---|---|
| National | SID, Iran Doc, RICST |
| International | Web of Science, Science Direct, Scopus, Embase, PubMed/Medline, EBSCO, CINAHL, Cochrane Library, EMBASE. |
| Search Term | Boolean Keywords |
|---|---|
| Outcome term | subdural hematoma |
| Population term | Patients with subdural hematoma |
| Descriptive term | Tranexamic acid OR subdural hematoma OR chronic subdural hematoma OR subdural OR Recurrence OR Medical treatment |
3.3. Inclusion and Exclusion Criteria
A checklist that contained the following items was used to collect the data: surname, year of study, year of publication, population, sample size, age, intervention, and outcomes. Data were also classified and reported using Word software.
3.4. Data Extraction
The checklist contained information on surname, year of study, year of publication, population, sample size, age, intervention, and outcomes. Data were also classified and reported using Word software.
4. Results
4.1. Search Results and Features
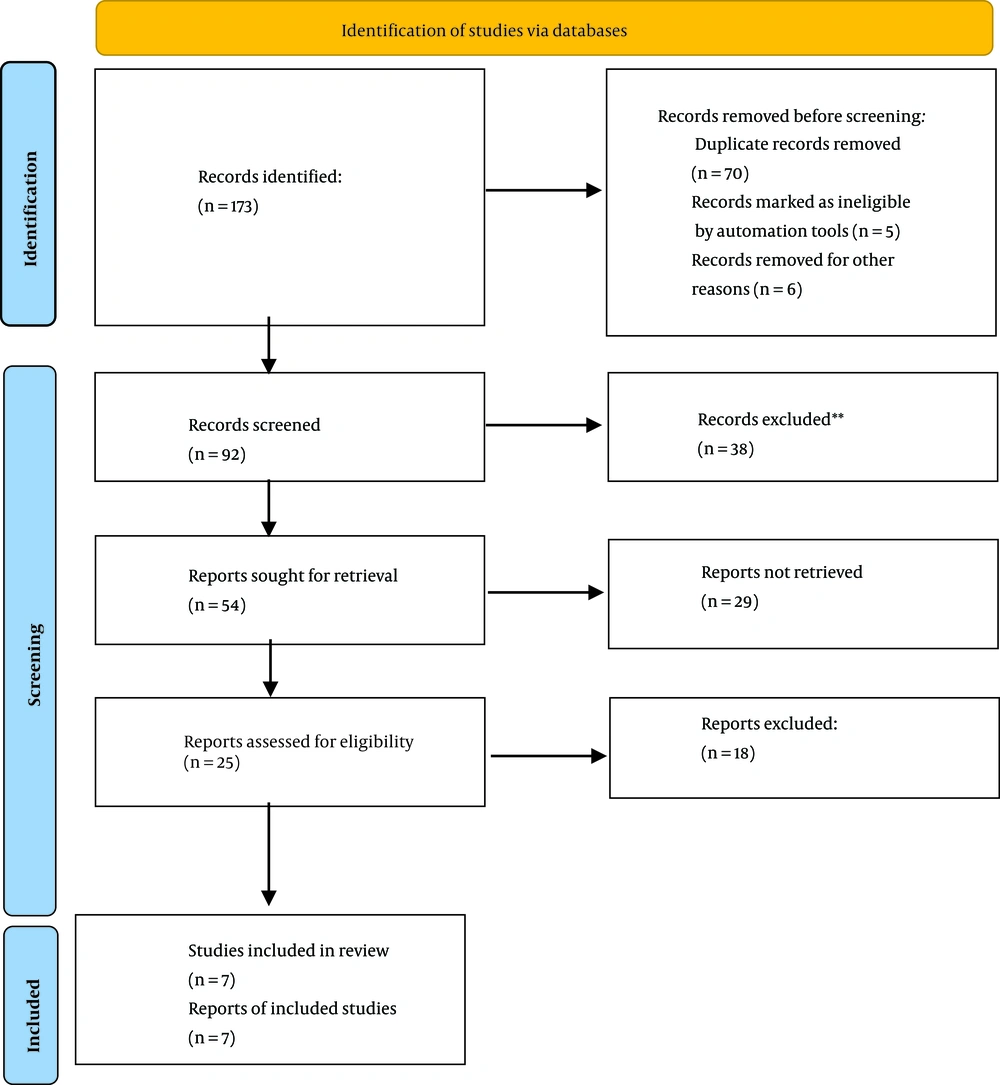
Initially, 178 articles were identified, out of which 118 were removed due to the relevance of the title and method, 44 due to duplication, six due to following the SR method, and three due to having a case report design.
4.2. Demographic Characteristics Articles Reviewed
Seven studies were found as eligible, as follows: the study by Wakabayashi et al. (27) with a sample of 199 patients, Kageyama et al.’s study (28) with 21 patients, Wan et al.’s study (29) with 90 patients, Kutty et al.’ study (30) with 27 patients, Tanweer et al.’s study (31) with 14 patients, Yamada and Natori’s study (32) with 193 patients, and Lodewijkx et al.’s study (33) with 7 patients (Table 3).
| Row | Author | Year | Study Design | Population | N | Age a | Intervention | Outcomes |
|---|---|---|---|---|---|---|---|---|
| 1 | Wakabayashi et al. (27) | 2012 | - | CSDH | 199 | 77.7 ± 10.5 | Patients were given Gorei-san, TXA, and a combination of these two medicines. | The recurrence rate was 8.3% in patients treated with Gorei-san, 10.9% in patients receiving TXA, 2.9% in patients with a combination of the two medicines, and 5.7% in patients without medication. |
| 2 | Kageyama et al. (28) | 2013 | Clinical article | CSDH | 21 | 79 Years | 750 mg of orally every day | Before treatment with TXA, the hematoma was 58.5 mL, which decreased to 3.7 mL after TXA. Also, hematoma did not recur or progress in any of the patients. |
| 3 | Wan et al. (29) | 2020 | Clinical article | CSDH | 90 | - | 500 mg/twice day | The use of TXA can reduce SDH. |
| 4 | Kutty et al. (30) | 2019 | Prospective Observational Study | CSDH | 27 | 39-87 years | 750 mg/day | The recovery rate of hematoma in patients was appropriate, and no progress was reported in any of the patients in terms of increased hematoma severity. Generally, TXA is an effective medicine in reducing hematoma status in patients with CSDH. |
| 5 | Tanweer et al. (31) | 2019 | Retrospective | SDH | 14 | 56.4 ± 16.3 | 650 mg daily orally | There was no increase or delayed recurrence of SDH during TXA treatment. There was also no thromboembolism in the patients. The average volume decreased from 9.44 ± 4.84 mm to 4.44 ± 3.29 mm. |
| 6 | Yamada and Natori (32) | 2020 | prospective study | CSDH | 193 | TXA Group: 78.2 ± 9.8; Goreisan Group: 79.2 ± 8.7 | 750 mg of orally | Mean (standard deviation) of hematoma in ml in the TXA group decreased from 108.1 (39.5) before intervention to 39.4 (23.2) on the day after intervention, 9.8 (16) 2 months after intervention and 5.6 (12.4) in 3 months after the intervention. While in the no-administration group, it decreased from 109.2 (38.7) before intervention to 36.8921.9) on the day after intervention, 17.9 (20) two months after intervention and 11 (16.5) three months after the intervention. Generally, the remaining hematoma volume in the TXA group was less than in other groups in 1, 2, and 3 months. |
| 7 | Lodewijkx et al. (33) | 2021 | prospectively | CSDH | 7 | 78 Years | 500 mg twice daily | The recurrence rate was 8.3% in patients treated with Gorei-san, 10.9% in patients receiving TXA, 2.9% in patients with a combination of the two medicines, and 5.7% in patients without medication. |
a Values are expressed as mean ± SD.
4.3. Role of TXA in Reducing SH
All studied articles showed that TXA could reduce SDH.
As shown in Figure 1, initially, 173 articles were extracted, of which seven were found as eligible (Figure 1). All articles were on patients with CSDH, and published from 2012 to 2021. In all articles that included the SR phase, TXA could reduce SDH.
5. Discussion
According to the findings, TXA could reduce SDH. Given the fact that no SR article has been conducted on the effect of TXA on SDH reduction, the findings are compared with other related articles. Two articles have investigated the TXA effect on hemorrhage reduction following the SR design. Gluud et al. investigated seven articles with a total of 1754 patients to evaluate the effect of TXA on the status of gastrointestinal hemorrhages, and showed that TXA could reduce hemorrhages in the upper gastrointestinal tract (34). In the same vein, Lee et al. performed an SR study (13 articles with a sample size of 2271 cases) and indicated that TXA could reduce continued bleeding, mortality, and urgent endoscopic intervention (35), which is consistent with the results of this study. TXA is a cheap and affordable medicine that can reduce the rate of hemorrhage and, consequently, the accessibility and need for blood, which has been shown to be effective in a variety of surgeries, including spine, obstetrics, plastic, and chest surgery (36, 37). In an SR study published by Scarafoni, with 23 articles in which TXA was prescribed to plastic surgery patients, it was indicated that TXA reduced hemorrhage, edema, and postoperative ecchymosis in plastic surgery patients (36).
Also, in an SR study published by Lee et al. with 11 articles in which TXA was prescribed to 1842 patients, it was shown that TXA could reduce operative time, postoperative hemorrhage, complications, and hospital stays in patients undergoing surgery with PCNL (38). Zhang et al. performed another SR study on the status of TXA on hemorrhage, with 13 articles in which TXA was prescribed to patients with femoral fracture surgery, and reported that it could reduce transfusion rate (39), which is consistent with the results of this SR study on the positive effect of TXA on patients with SDH.
Given the fact that the effect of TXA on patients with different medical diagnoses has been evaluated in various articles, so far, no study has investigated the effect of TXA on SDH, and this issue has been considered for the first time in this study. Therefore, conducting this study is a strength. One of the limitations of this study is the lack of meta-analysis in this field, which is suggested to be performed on recently published articles.
5.1. Conclusions
According to the positive effect of TXA on reducing SDH, the administration of this medicine is recommended to treat patients with CSDH.