1. Background
Traumatic brain injury (TBI) can lead to long-term injuries in patients (1). TBI is one of the leading causes of disability and death with a prevalence of about ten million people annually (2). By 2020, TBI became the third leading cause of death and disability (3). This type of trauma can also lead to complications such as depression, anxiety, and reduced quality of life. In this group of patients, the depression rate was 5 - 10 times higher than other patients (4, 5). Head trauma, including decreased level of consciousness, brain contusion, post-traumatic syndrome, subdural hematoma, and skull fracture, is one of the most important causes of mortality in hospitalized patients in the world (6, 7).
Causes of TBI include falls from heights, rocks falling from mountains, collision of heavy and hard objects with the head, and accidents. Also, the injuries are mostly in the head and neck areas and eventually lead to brain injuries (8, 9). Brain injuries range from mild to severe and can be graded based on the Glasgow level of consciousness. In many TBIs, cerebral hemorrhage occurs and is divided into primary and secondary types. In the initial hemorrhage, there is evidence of bleeding on the patient's CT scan up to the first six hours after the injury, while in the delayed hemorrhage, bleeding may appear within six hours of the injury. Therefore, it is very important to study the progression of the disease in these patients based on diagnostic findings (1, 10-12).
Diffuse axonal injury (DAI) is an injury that occurs after the onset of TBI, leading to many problems for patients and imposing high costs on the treatment system (13). This disease has been diagnosed in 56% of patients with moderate (56%) and severe (90%) TBI injury (14). DAI is caused by axonal injuries that occur in accidents, falls, as well as contentions and can lead to axonal amputation at the time of injury. This disease leads to secondary axotomy by disrupting axonal transmission and normal homeostasis (15), which can lead to signal and cognitive dysfunction (16).
Axonal injury is one of the main causes of DAI, the occurrence of which depends on the severity of the injury and is more common in traumas with higher injury, especially in accidents (17). DAI is defined as the most important factor for complications in patients with TBI, as well as mortality in these patients. DAI can lead to physical, cognitive, and behavioral changes, causing decreased quality of life, disability, and disruption of daily activities (17, 18). Due to the fact that brain tissue is functionally impaired but not destroyed, the brain may gradually regain its normal function and the individual's neural function is restored (19, 20).
2. Objectives
Due to the importance of examining the effect of laboratory variables status on the prognosis of patients' progress and also the importance of considering health and disease control in patients with DAI, this study was conducted to investigate the status of laboratory variables in patients with DAI.
3. Methods
3.1. Study Design
This cross-sectional study was conducted in Ilam, Iran (ethics code: IR.MEDILAM.REC.1397.013).
3.2. Study Population
In this study, 140 patients with DAI were included.
3.3. Inclusion and Exclusion Criteria
3.3.1. Inclusion Criteria
All DAI patients diagnosed by magnetic resonance imaging (MRI) were included in the study. The DAI status was determined as follows: if the patient had DAI lesions confined to the white matter of the lobar or cerebellum, they were assigned into the stage 1 group; if the patient had DAI lesions located in the corpus callosum (possibly with or without stage lesions 1), they were assigned into the stage 2 group; and if the patient had DAI lesions located in the brainstem (possibly with or without stage lesions 1), they were assigned into the stage 3 group (21). The consent of the patient's companions or legal guardian to participate in the study, the definitive diagnosis of the treating physician for DAI, and the absence of other comorbidities were also considered as inclusion criteria.
3.3.2. Exclusion Criteria
The exclusion criteria were as follows: incomplete information, patient death, and dissatisfaction of the patient's companions or legal guardian to participate in the study.
3.4. Data Gathering
Data collection tools included a demographic profile form (age, gender, Glasgow Coma Scale (GCS) score, and causes of injury) and MRI to diagnose DAI.
3.5. Research Method
Laboratory tests, including glucose, LDL-C, HDL-C, total cholesterol, triglycerides, Hb, HCT, PT, PTT, INR, BUN, creatinine, and CRP were evaluated. Specialized devices were used to study the laboratory and radiology variables. Lipids and lipoproteins were considered at 7 AM within one week after injury. Also, to determine the validity of the obtained results, the relevant tests that could be assessed by a common device, were evaluated by a specific diagnostic device.
Regarding MRI, all cases were performed 30 days after the injury using a device in the hospital. The criteria for dividing patients into DAI and non-DAI were confirmed by MRI. Interpretation of test results was performed by a pathology specialist and interpretation of all MRIs was performed by a neurologist. The validity and reliability of all devices were confirmed.
3.6. Data Analysis
SPSS 16 software and descriptive-analytical statistical tests were used for analysis.
4. Results
As Table 1 shows, most (61.5%) of the patients were male, 47.1% had a non-governmental occupation, and 55.7% were less than 30 years old. Also, in 87.9% of cases, traffic accidents were the cause of DAI, and the GCS was less than 7 in 65% of patients.
| Variables | No. (%) |
|---|---|
| Gender | |
| Male | 85 (61.5) |
| Female | 54 (38.5) |
| Job | |
| Unemployed | 33 (23.6) |
| Housewife | 21 (15) |
| Non-governmental | 66 (47.1) |
| Employee | 20 (14.3) |
| Causes of injury | |
| Traffic accidents | 123 (87.9) |
| Clash | 15 (10.8) |
| Other cases | 2 (1.3) |
| Age | |
| < 30 | 78 (55.7) |
| 30 - 40 | 29 (20.7) |
| 40 - 50 | 24 (17.1) |
| > 50 | 9 (6.5) |
| Mean ± SD | 28.2 ± 12.6 |
| Glasgow Coma Score | |
| Range | 3 - 13 |
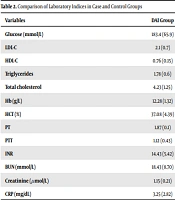
Table 2 compares the laboratory indices in case and control groups.
| Variables | DAI Group | Non-DAI Group | P-Value |
|---|---|---|---|
| Glucose (mmol/L) | 183.4 (65.9) | 143.40 (45.12) | < 0.05 |
| LDL-C | 2.1 (0.7) | 2.4 (0.9) | < 0.05 |
| HDL-C | 0.76 (0.15) | 1.03 (0.6) | < 0.05 |
| Triglycerides | 1.78 (0.6) | 1.8 (0.8) | < 0.05 |
| Total cholesterol | 4.23 (1.25) | 4.6 (0.7) | < 0.05 |
| Hb (g/L) | 12.28 (1.32) | 11.07 (3.9) | < 0.05 |
| HCT (%) | 37.08 (4.39) | 36.9 (4.32) | < 0.05 |
| PT | 1.87 (0.1) | 1.32 (0.21) | < 0.05 |
| PTT | 1.12 (0.43) | 1.32 (0.54) | < 0.05 |
| INR | 14.43 (5.42) | 12.24 (6.5) | < 0.05 |
| BUN (mmol/L) | 18.43 (8.70) | 13.32 (4.52) | < 0.05 |
| Creatinine (µmol/L) | 1.15 (0.21) | 0.9 (0.1) | < 0.05 |
| CRP (mg/dL) | 3.25 (2.82) | 1.28 (0.42) | < 0.05 |
5. Discussion
It is important to pay attention to emergency patients (22-24). Evaluating laboratory variables is also important and provides complete and accurate information (25-27). The present study aimed to investigate the relationship between laboratory variables and disease prognosis in patients with DAI. Most patients were in the age range of less than 50 years; 78 (55.7%) patients were under 30 years of age, 29 (20.7%) patients were in the age range of 30 - 40 years, and 24 (17.1) patients were in the age range of 40 - 50 years. Also, the mean (standard deviation) age of patients was 28.2 (12.6) years. In the study by Jatav et al., 29 (29%) patients were between 20 - 29 years, 24 (24%) were under 40 years, and 18 (18%) were under 50 years old, which is consistent with our study (28). Also, the mean age of patients in the study by Zhong et al. was 42 (16.6) years (29), and 31.2 in the study by Abu Hamdeh et al. (30), which is consistent with the results of the present study.
According to our results, most of the patients were male. In the study by Humble et al., 71% of patients were male (31), in the study by Mata-Mbemba et al., 73.3% were male (32), and in the study by Benjamini et al., most of the patients were male (16). This is consistent with our results indicating that the number of male DAI patients is high compared to females. Since the most important causes of TBI include traffic accidents, conflicts, and falls, males are more exposed to such injuries due to their job type and environment (33, 34).
Also, 65% of DAI patients had GCS less than 7 and the GCS range was between 3 and 13 for all patients. In the study by Kim et al., 43% of patients had GCS less than 13 (35), in the study by Ljungqvist et al., the GCS range was between 3 and 14 (36), in the study by Tong et al., the range of GCS was 3 - 15 (37), in the study by Xie et al., the range of GCS was between 6-9 (38), and in the study by Jatav et al., all patients had GCS less than 8 (28). These results are consistent with the results of this study in which GCS score was low in patients with DAI. The cause of most DAIs was traffic accidents; in the study by Jatav et al., 43.6% of the DAIs were caused by vehicle accidents, especially motorcycles (28), in the study by Zhong et al., 51.4% of cases were due to traffic accidents (29), in the study by Xie et al., 89.2% of DAI patients were due to traffic accidents (38), and in the study by Rabinowitz et al., this rate was 70% (39), which are consistent with the results of the present study.
In our study, DAI led to disruption of laboratory variables (P < 0.05). In the study by Zhong et al., it was shown that after DAI, the rate of laboratory variables is disturbed, which is consistent with the results of the present study based on the disturbance of the results of laboratory variables such as HDL-C, LDL-C, triglycerides, and total cholesterol after DAI (29). Also, in the present study, the hemoglobin level was equal to 12.28 (1.32), while in the study by Lee et al., it was equal to 12.99. 2.30 (g/dL) (40), which is consistent with the results of the present study.
According to the findings, laboratory variables in patients with DAI had a statistically significant difference compared to the case group, which indicates the negative effect of DAI on laboratory variables. However, further studies are needed to confirm our results.