1. Background
Studies on the category-specific effects in semantic categorization tasks have become increasingly important for investigating the structure of the semantic system of the speaker’s language. This field of study takes its roots from research projects in the field of semantic memory, as well as from neuropsychological studies of brain-damaged patients, who occasionally exhibit a selective loss of semantic knowledge in some categories. A large number of brain-damaged patients are reported to have lost their semantic knowledge about living (e.g. animals) categories, while still having knowledge about objects from non-living (e.g. tools) categories (1, 2). The opposite pattern has also been observed, but less frequently (3, 4).
Similarly, studies on healthy subjects have revealed category-specific brain activation during picture naming (5-7). In a study using PET, Martin et al. noticed that naming tools elicited stronger activation than naming animals in the left frontal, as well as the left temporal lobe, but less activation in the left occipito-temporal region (7).
Different hypotheses have been proposed to explain such dissociation between living and non-living categories. One of these theories, called “Feature-based account of semantic memory” (1, 8), also referred to as the sensory-motor theory (9), considers the organization of conceptual knowledge according to the semantic object’s features (such as visual, auditory, action, functional properties) (10). According to this theory, category-specificity is not the underlying organizational principle, but it is an apparent category-structure that emerges, since concepts rely differentially on sensory, action, and verbally acquired knowledge. In particular, category-specific semantic deficits for living and non-living items are explained by their differential associations with sensory and action features. While sensory features are important for distinguishing between living items, action semantics play a critical role in the representation of inanimate items (especially tools) (1, 8).
There is also an alternative hypothesis, known as the “domain specific account” (11, 12), which tries to explain category-specific dissociation on different grounds, such as evolutionary pressure. According to this theory, neuroanatomically and functionally specialized neural circuits for processing of particular semantic domains may have originally evolved, because rapid and efficient identification of objects from particular categories could have survival advantages (13).
This study tries to investigate this category-specific semantic organization of tool and animal words among Persian speakers, using the event-related potentials (ERPs) technique. Although the ERP technique is not yet widely used to study this dissociation effect, due to its poor spatial resolution in comparison with neuroimaging techniques such as fMRI or PET, its high temporal resolution makes it a good potential candidate for investigating early cognitive processes. It also allows researchers to demonstrate that the differences in processing animals and tools occurred specifically within the time-window encompassing semantic analysis, such as differences in ERP components like N400.
N400 is a negative wave with onset latency between 200 ms and 250 ms post-stimulus and maximal amplitude around 400 ms. This wave has been related to semantic processing, since its amplitude is enhanced with semantic incongruence between word and context (14, 15) and decreases for conditions in which the context enables the subject to predict the word (16), or when semantically related stimuli have previously been presented (17-23).
In the present study, two different categories (animal and tool words) were used, to dissociate semantic sources underlying category-specific effects in normal healthy subjects.
According to the feature-based account of semantic memory, it is hypothesized that the two different categories of tools and animals may have different semantic sources. Since the sensory features are more important for distinguishing the living items, we expect more activation for animals’ stimuli in occipito-parietal regions. Action features, on the other hand, have an important role in processing inanimate objects; hence, we expect larger amplitude in fronto-central regions in functional judgment tasks for tools’ stimuli. Furthermore, since the importance of the motor-related aspects of representation for tool concepts, we expect tools’ activation to be more left-lateralized in all right-handed subjects.
2. Objectives
The ultimate goal of this work was the investigation of semantic organization of two distinctive domains.
3. Materials and Methods
A cross-sectional study using event-related potentials was performed to investigate categorization of two distinctive domains. The study was approved by the ethics committee of the Iranian center of neurological research (ICNR) of Tehran University of Medical Sciences.
3.1. Subjects
Twelve male right-handed university students, all native speakers of Persian (age range: 18 - 21 years; mean = 19.9, SD = 0.9) participated in this experiment. Since in the case of long hair, the gel injection and decreasing impedance is more difficult and may cause harm to the results of the research, only males were enrolled in this study. All subjects signed informed consent and reported to be free from neurological or psychiatric disorders, and had normal or corrected-to-normal vision. Data from two additional subjects were excluded from the analysis due to non-correctable blinking artifacts and technical problems during recording.
3.2. Stimuli and Task
A set of 100 common words, composed of 50 tool words and 50 animal words, were selected from a contemporary Persian dictionary. The animal category included mammals, reptiles, marine creatures and insects. The tool category was a variety of man-made objects (e.g., a comb, a broom, an axe). Since the words were similar with respect to visual processing across categories, the use of words instead of pictures would help us in focusing on semantic processing of category-specificity, instead of perceptual processing, which is related to earlier ERP components such as N1 (24-26). Also, the use of verbal stimuli enables us to investigate semantic sources of category-specificity, particularly in the Persian language, for the first time.
The words were matched for familiarity and concreteness, as well as number of letters. For these tasks, three examinations were conducted in a separate sample of subjects, thirty students, with the same range of age. There were no significant differences between animal and tool words in terms of familiarity (t = -1.731, P = 0.09), concreteness (t = -1.552, P = 0.127) and number of letters (t = -0.393, P = 0.696). It should be mentioned that due to the lack of any established language corpus in Persian, familiarity is used instead of frequency.
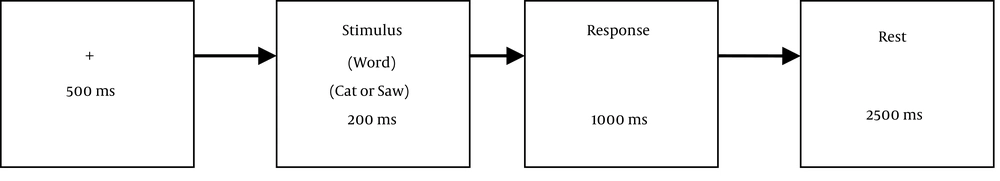
In a semantic judgment task, subjects were seated in a dark, sound-attenuated and shielded room facing a PC monitor at a distance of 100 cm with their chins positioned on a chinrest. Participants were instructed to fixate on a cross, centrally located on a white background, which was presented 500 ms before the onset of the stimuli. Each stimulus was presented for 200 ms, followed by a 1000 ms time for response and a 2500 ms time to rest and blink (Figure 1). Participants were instructed to respond as quickly and accurately as they could to the words by pressing a button on the response box with their right hand. The index finger was used for animals and the middle finger was used to press another button for tool words. Before the main experiment, subjects went through a training phase in which they were able to become acquainted with both animal and tool words. During the training session, a group of 50 words (25 words from each category, different from the main trial list) were shown to the participants. Experimental trials started only when subjects detected 85% of the target stimuli and responded to them within 1000 ms. Otherwise the training continued for another trial block with the same fifty stimuli. Once subjects passed the training, the experimental trial started, comprised of a series of 100 words, 50 from the tool category and the rest from animals. Stimuli were presented in a pseudorandom sequence. Reaction time and error rates were recorded.
3.3. EEG Recording
The electroencephalogram (EEG) was recorded continuously from 32 scalp sites using high input impedance amplifier and Ag/AgCl electrodes mounted in an electrode cap. The electrode positioning on the scalp was according to the 10 - 20 international system. Electrodes on the right side of the scalp were: FP2, F8, FT8, T8, TP8, P8, F4, FC4, C4, CP4, P4; on the central side: FZ, FCZ, CZ, CPZ, PZ; on the Left side: FP1, F3, FC3, C3, CP3, P3, F7, FT7, T7, TP7, P7; and on the occipital lobe: O1, OZ, O2.
To ensure that fixation was maintained, the horizontal and vertical EOGs were also recorded. Vertical EOG was recorded bipolarly from electrodes placed below and above the left eye, whereas horizontal EOG was recorded bipolarly from electrodes placed on the outer canthi of the two eyes.
A reference lead was linked to the bilateral mastoids. EEG data were filtered with a band pass of 0.05 Hz to 30 Hz and digitized at 1000 Hz (0.024 Micro volt per precision) for later offline analysis.
3.4. Data Analysis
EEG data analysis was performed using EEGLAB (version 4.515), a freely available open source toolbox running under MATLAB version 7 (Mathworks) (27).
First, data were down-sampled to 250 Hz to save later computation time. Data epochs were extracted (-200 ms to 1000 ms) and baseline was corrected (-200 ms to 0 ms).
Trials contaminated by eye or body movements were rejected. Artifact rejection by extreme value was used before averaging to discard epochs in which eye movement, blinking, excessive muscle potentials or amplifier blocking occurred. The criterion for artifact rejection was peak-to-peak amplitude exceeding ±40 microvolts of the EOG and HOG electrodes, and the rejection rate was about 15%.
ERP epochs were averaged offline from 200 ms before to 1000 ms after stimulus onset.
The average amplitude of the N400 was measured in a range of 300 - 600 ms post-stimulus, both for animal and tool trials. Average amplitude in the ranges of 0 - 150 ms, 150 - 200 ms, and 200 - 300 ms was also measured.
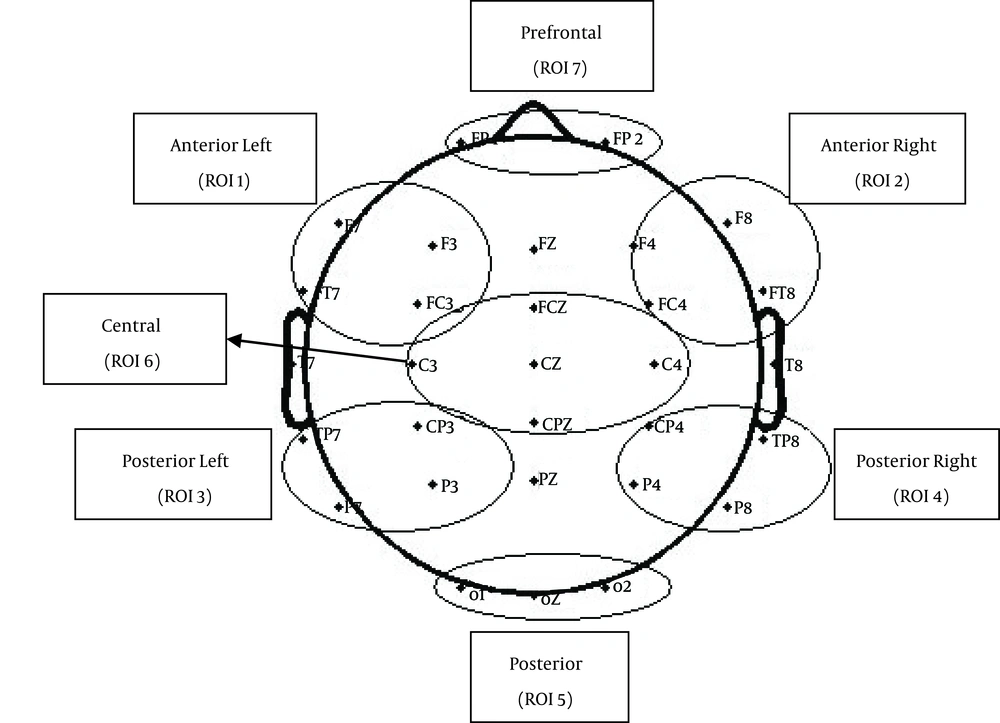
Similar electrodes were grouped to form a region of interest. This resulted in defining seven different regions of interest (Figure 2): posterior, posterior-left, posterior-right, central, anterior-left, anterior-right and prefrontal (Table 1). ERP waveforms were averaged across all the electrodes of each region to compute an average waveform for the whole region.
| ROI | Electrodes |
|---|---|
| Posterior | O1,Oz, O2 |
| Posterior-right | CP4, TP8, P4, P8 |
| Posterior-left | P7, P3, TP7, CP3 |
| Anterior-right | F4, F8, FC4, FT8 |
| Anterior-left | F7, F3, FT7, FC3 |
| Central | FCz, C3, Cz, CPz, C4 |
| Prefrontal | FP1, FP2 |
Regions of Interest (ROIs)
The paired t-test for behavioral data and repeated measures analyses of variance (ANOVAs) on ERPs average amplitude were performed, with region of interest and time window as the dependent factors and category as the independent factor.
4. Results
4.1. Behavioral Data
The task was performed equally well in the two conditions by all subjects (97% correct in the semantic judgment task between the tools and animals categories). Analyzing mean reaction times with t-paired test revealed a significant difference between animal and tool words (t = −7.995, P ≤ 0.001): Subjects responded faster to animal words (Mean: 530 ms, SD: 71 ms) than to tool words (Mean: 559 ms, SD: 79 ms).
4.2. Electrophysiological Results
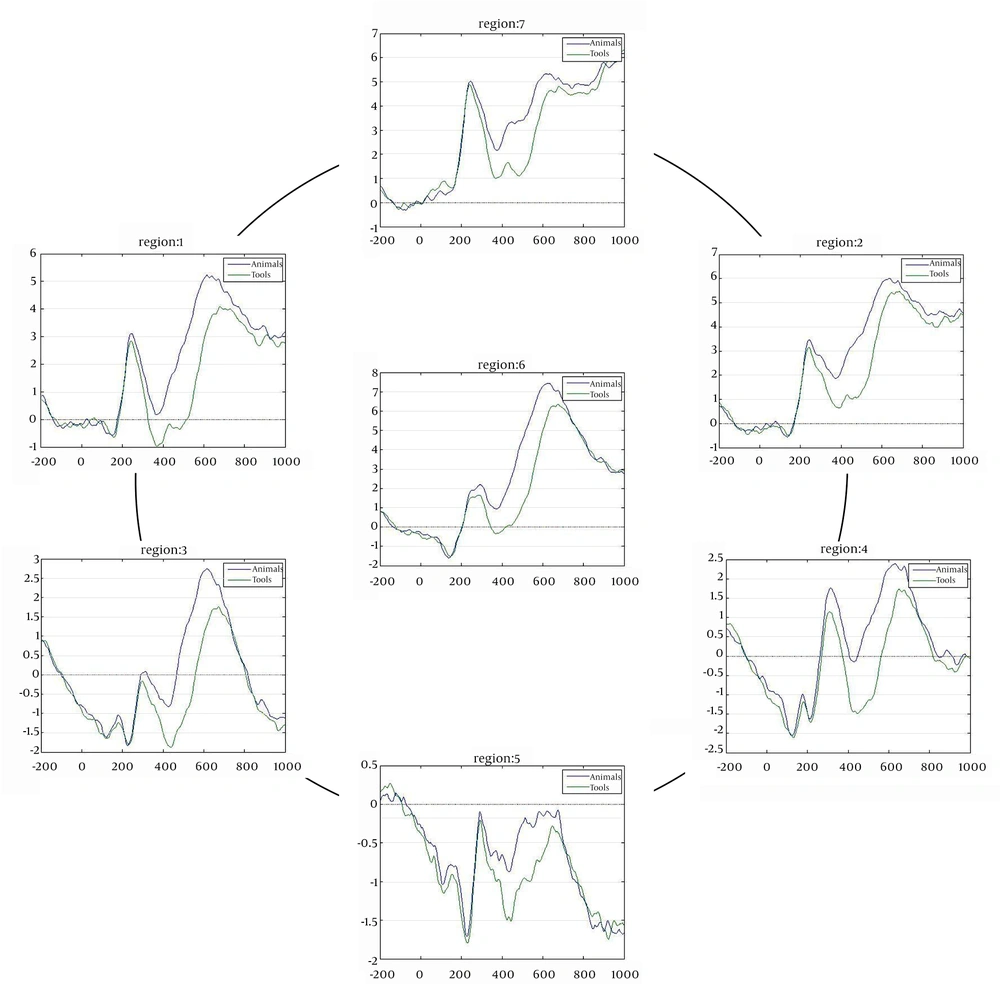
Figure 3 shows grand mean ERP waveforms for animal vs. tool words in all regions of interest. In both conditions, the tools category indicates more negativity in topography than the animals category.
Table 2 shows mean average amplitude in all regions and all time-windows relative to both categories. In most of the regions and time-windows, tool words elicited more negative amplitude than animal words.
| Stimulus | 0 - 150 | 150 - 200 | 200 - 300 | 300 - 600 |
|---|---|---|---|---|
| Anterior-left (n=48) | ||||
| Animal | 1.91 (8.16) | 1.77 (9.2) | 0.38 (9.67) | 0.88 (7.49) |
| Tool | −2.06 (8.6) | 0.12 (9.47) | −2.53 (7.9) | −1.53 (8.76) |
| Anterior-right (n=48) | ||||
| Animal | 1.8 (9.11) | 2.74 (9.63) | 1.62 (9.78) | 2.3 (6.82) |
| Tool | 0.66 (8.9) | −2.47 (7.68) | 0.69 (6.36) | −2.22 (7.87) |
| Prefrontal (n=24) | ||||
| Animal | 1.69 (11.24) | 1.83 (7.37) | −0.05 (11.27) | 1.75 (6.07) |
| Tool | 2.84 (11.84) | −0.56 (10.93) | 2.44 (9.77) | −2.5 (6.17) |
| Central (n=60) | ||||
| Animal | 1.54 (9.01) | −0.11 (8.03) | 1.75 (8.25) | 0.15 (6.34) |
| Tool | −2.85 (7.23) | −5.21 (6.99) | −3.49 (6.28) | −5.3 (6.04) |
| Posterior−left (n=48) | ||||
| Animal | −2.31 (5.78) | −2.17 (5.42) | −1.43 (6.01) | −0.26 (6.81) |
| Tool | −6.95 (6.93) | −4.71 (8.12) | −6.15 (5.28) | −4.79 (6.57) |
| Posterior−right (n=48) | ||||
| Animal | −4.64 (6.03) | −3.84 (6.75) | −1.16 (4.8) | −1.33 (6.27) |
| Tool | −7.13 (6.64) | −5.53 (6.21) | −5.15 (6.8) | −4.94 (7.5) |
| Posterior (n=36) | ||||
| Animal | −3.9 (3.05) | −3.58 (7.16) | −2.45 (3.79) | −1.29 (5.82) |
| Tool | −3.76 (5.16) | −1.39 (8.97) | −2.96 (4.75) | −2.15 (6.71) |
Mean Average Amplitude of ERP Waveform in Different Regions of Interest and Time-Windows, Compared in Two Categoriesa
Repeated measures analyses of variance (ANOVAs) on ERPs average amplitude was used for main factors and interactions (Table 3). Relatively, in all time-windows moving from anterior to posterior for both conditions the amplitude shifts to more negative, in which ROIs indicate a significant difference (P < 0.001). For all time-windows, tool amplitude is more negative than animal amplitude, and this difference is significant for all time-windows (P = 0.009).
| Main factor | df | F | P Value |
|---|---|---|---|
| Stimulus type | 1 | 8.534 | 0.009 |
| Time window | 1.78 | 2.443 | 0.108 |
| ROI | 2.527 | 10.675 | 0 |
| Two level interactions | |||
| Stimulus × time window | 1.78 | 0.034 | 0.954 |
| Stimulus × ROI | 2.527 | 2.419 | 0.088 |
| ROI × Time Window | 4.15 | 3.131 | 0.018 |
| Three level interactions | |||
| Stimulus × time window × ROI | 4.15 | 0.242 | 0.919 |
Repeated Measures Analyses of Variance (ANOVAs) on ERPs Average Amplitude
Also, the region of interest has a significant effect on time of processing (time-window) and it is right for all time-windows and regions of interest.
5. Discussion
We used ERPs to investigate using category-related brain activation to represent semantic memory organization. ERPs that were divided into two distinctive categories, animal and man-made tool words, were recorded. Results indicated no advantage for N400 as a semantic processing characteristic, because there was no significant difference between time-windows for both conditions. Also we see increasing negativity in all time-windows when moving from anterior regions to posterior. Tool words elicited more negativity in the central region and animal words in the posterior regions. Notably, we did not see increased negativity for tool words in left frontal sites.
Generally, average amplitudes elicited for tool words were greater in negativity than for animal words. This finding, along with behavioral results suggesting tool words have broader distinction than animal words, has to be made (26, 28-31). This is in contrast with naming tasks, in which members of natural categories are identified more slowly than members of artifactual categories, because categorical decisions require fine-grained distinctions between competing alternatives (24, 25).
There is a significant difference between processing of animal and tool words in all time-windows. This different spatial distribution of two categories, as well as different average response times for animal and tool words (faster RT for animal words), can be suggestive of different semantic representations and processing mechanisms being involved in semantic categorization of tool and animal concepts.
Also, the more left-anterior-oriented spatial distribution of the tools N400 can be attributed to the motor-related aspects of man-made objects, and the representation and processing in the semantic knowledge organization of the brain. This is in accordance with the feature-based account of semantic memory (1, 8, 31), in which conceptual knowledge is organized according to semantic object features. In this context, a category-specific semantic effect for living and non-living items is explained by their differential associations with sensory and action features. While sensory features are important for distinguishing living items, action semantics play a critical role in the representation of inanimate items (especially tools) (1, 8, 31).
The results of this study have indicated that semantic processing can happen in other time-windows and that the tool category has more specific features than the animal category for processing. Therefore, the feature-based theory of categorization is confirmed.