1. Context
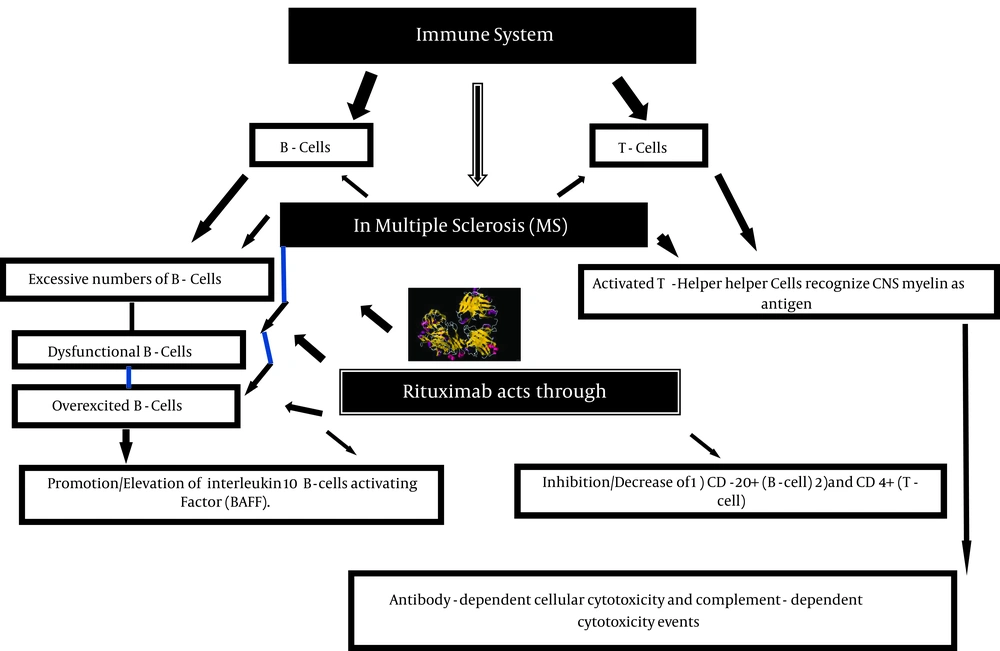
The annulled U.S patent in 2015; rituximab (C6416H9874N1688O1987S44) was first issued in 1998. The drug is against the protein CD20, which mainly originates on the surface of immune system B cells (1). Figure 1 shows the mechanism of rituximab action in Multiple Sclerosis (MS). Two groups of cells called B and T cells belong to white blood cells within immune system. The chimeric (mouse/human) monoclonal antibody, Rituxan, MabThera, Zytux or Rituximab, destroy B cells, therefore they are used in autoimmune disorders described by excessive number of B cells, overexcited B cells, or dysfunctional B cells.
There are three forms of T-cells: 1) Helper T-cells, 2) Suppressor T-cells and 3) Cytotoxic T-cells. Recognition of foreign antibody is the duty of helper T-cells by motivating antibody production. It also acts as a biological messenger by creating antibodies. Suppressor T-cells are in charge of turn-off related to response within the immune system. To attack and abolish antigenic materials is the duty of cytotoxic T-cells (1-3). Due to unidentified stimulus, in MS, activated helper T-cells recognizes central nervous system myelin as an antigen (1, 2). According to previously published data, anti-B cell drug rituximab considerably decreases the deteriorations and new brain lesions in a stage two clinical trial of patients with relapsing-remitting (2).
Rituximab is used in many neurological disorders such as MS, neuromyelitis optica, myasthenia gravis, Lambert-Eaton myasthenic syndrome, chronic inflammatory demyelinating polyradiculoneuropathy, paraneoplastic neurological syndromes, primary central nervous system lymphoma, inflammatory myopathy, and some other autoimmune-mediated neurological disorders (4-8).
Rituximab is considered as a class C for pregnant women. The molecular mass of drug is 143859.7 g/mol. With a half-life of 30 to 400 hours which is varied by dose and length of treatment, the drug has a bioavailability of 100 %. The administration is only by intra venous (1, 2). A recent publication suggests that subcutaneous rituximab may become a useful option in patients with B-cell non-Hodgkin’s lymphoma (9).
B cells have puzzling responsibilities in autoimmunity, applying both pathogenic and defensive properties (3, 10-12). Pathogenesis may be antibody independent, as B cell reduction treatment directs to improvement of disease irrespective of autoantibody ablation (1, 3). Previous publications show that B cell reduction treatment can strongly decrease disease development in relapsing-remitting MS and in trial autoimmune encephalomyelitis (13). Thus, in addition to their recognized repressive capacity, B cells also help the inflammatory response in MS (1, 2, 5, 9). Even though polymorphisms in genes scheming T-cell commencement demonstrate the strong relationship with illness vulnerability, B cell activation is ,besides being a common irregularity in Relapsing-Remitting MS (RR-MS), tinted by the attendance of intrathecal oligoclonal immunoglobulin bands in > 90% of patients (9, 14). It is therefore clear that B cells participate in MS (2, 5, 6). Adverse effects could be raised in > 50%, while most of them could be insignificant (15). Humoral immunity constantly suppressed a few months after prescription that might be followed by myelosuppression. It could cause recurrence of Hepatitis B Virus Progressive Multifocal Leukoencephalopathy (PML), and severe opportunistic infections, some of which are deadly (8).
Finally an early talented intervention in MS related to pharmacotherapy management might improve patients’ outcome, or even decrease the number of lesions and plaques. To achieve individualized drug therapy based on inter and intra individual variations in Iranian population of patients with MS (10), optimistic magnitudes were detailed for rituximab since 1998 (1-15). Therefore, a comprehensive review related to pharmacokinetics parameters of rituximab in terms of efficacy, safety and adverse effects especially in MS were updated.
2. Evidence Acquisition
2.1. Survey Method
Directory of open access journals (DOAJ), Google Scholar, United States National Library of Medicine (PubMed, NLM), LISTA (EBSCO), and the Web of Science were searched. The key words were relevant to rituximab, rituximab efficacy and safety, rituximab pharmacokinetics, rituximab in MS, rituximab efficacy and safety in MS. A total of 14053 (December 1994 - May 2015), 808 (May 1998 - March 2015), 319 (September 1997 - May 2015), 292 (October 2003 - April 2015), and 23 (October 2007 - December 2014) were found. In the next step articles relevant to the clinical properties of rituximab were selected and reviewed individually.
3. Results
Table 1, shows pharmacokinetics parameters of rituximab (Rituxan, MabThera, Zytux). It could be prescribed as the first targeted therapy used in B-cell malignancies. The category-1, anti-CD20 monoclonal antibody, rituximab, alleviates CD20 on lipid bundles, helping the antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity undertakings (16). It is reported that in addition to reduction of B-cells from cerebrospinal fluid at 24 weeks after preliminary treatment, rituximab could also reduce T-cells in cerebrospinal fluid (5). A combination of azathioprine, oral prednisolone and rituximab in patients with neuromyelitis optica defined longer time to next attack (17).
| Parameter | Reported Value |
|---|---|
| F, % | 100 (IV) |
| T1/2, h | Variable by D and DT (30 - 400) |
| Excretion | Not clear, may undergo phagocytosis and catabolism in RES. |
aAbbreviations: D; Dose, DT; Duration of Treatment, E; Excretion, F; Bioavailability, RES; Reticuloendothelial System, T1/2; Half-Life.
bMonoclonal antibody Ritoximab; Type: Whole antibody; Source: chimeric (mouse/human).
In patients with rheumatoid arthritis, since 2006 the prescription of rituximab was approved. Due to an increase in the level of tumor necrosis-α, risk of acute pulmonary fibrosis seems to be high (18-20). Durability, pharmacodynamics and efficacy were reported similar in subcutaneous and intravenous route of administration. Effectiveness of rituximab to treat neuromyelitis optica was reported in two patients (1, 21, 22). In pediatric neuromyelitis optica the use of rituximab and MS in a cohort study was reported overall safe and effective. As a new and optimistic approach in progressive form of MS, intratechal therapeutic appears to be a practiced management. Nonetheless, the need for trials on the intrathecal use of rituximab in MS is suggested and needs to be supported by further clinical investigations (23-26).
4. Conclusions
According to the previous reports, more than 80% of patients with MS experience a relapsing-remitting disease course. Nearly 10 years after disease onset, an estimated 50% of patients with relapsing-remitting convert to secondary progressive form of the disease (11, 27, 28). Therapeutic trials using rituximab for CD20-targeting and B-cell diminishing have specified the escalation to encourage the methods related to the duty of B-cells in the pathogenesis of MS in young adults. The monoclonal antibody, rituximab, by depleting CD20+ B-cells demonstrated efficacy in reducing disease activity in relapsing-remitting MS. Other developing therapies included alemtuzumab, daclizumab, laquinimod, estriol, 3-hydroxy-3methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors (statins), and vitamin D (26-31). In exceptional individuals with MS, mostly fulminant or types that fail to recover subsequent management with steroids and plasma exchange, cytotoxic drugs such as cyclophosphamide or B cell-depleting regimen such as rituximab might be prescribed (31). Monoclonal antibodies seem to quickly reallocate to the peripheral section subsequent to intrathecal injection. Ultra-low doses of rituximab administered intrathecally are adequate to cause whole diminution of peripheral B lymphocytes, representing that low-dose intrathecally management is possibly effective in both the central nervous system and systemic compartments. Rituximab-induced decrease in CD4+ and CD8+ T-cells may increase the risk of infection in susceptible individuals. However among different autoimmune diseases the drug efficacy seems vary, but collective clinical data would recommend that in a vast majority, rituximab might have an encouraging role in the treatments. The drug effectively reduces B-cells and may affect otter cells of the immune system by re-forming immune homeostasis and tolerance (32-35).
In conclusion, attributed pharmacotherapy intervention and clinical practice, in terms of prescription based on specific benefit-risk assessment, necessitates direct thoughtful and well-adjusted rituximab data in the Iranian patients with MS. Finally, despite the standard care for such patients worldwide, including Iran, there is still the need for development of pharmacotherapy command in terms of research on efficiency and adverse effects.