1. Context
Temporal lobe epilepsy (TLE) is the most frequent drug-resistant epilepsy. It represents approximately two thirds of the intractable seizure population requiring surgical management (1). Surgical treatment for these patients is typically a safe, effective and well-established option, with a success rate of 70% to 90% (2).
The best surgical outcomes are obtained when the epileptic zone (EZ) (3) is accurately localized during the pre-surgical evaluation. The EZ usually includes the ictal-onset zone (IOZ), the cortical region where seizures start and, in a variable and not well-defined degree, the interictal or irritative zone (IZ), the area showing interictal epileptiform discharges (IEDs). The ancillary tests used during the pre-surgical evaluation include (2, 4) video-electroencephalography (v-EEG), magnetic resonance imaging (MRI), single photon emission-computed tomography (SPECT) and positron emission tomography (PET). When results of the tests are not functionally and anatomically consistent, invasive recordings, such as foramen ovale (FO), subdural or depth electrodes are required (5). Pharmacological activation inducing or increasing interictal activity is also used as a complementary method, together with v-EEG to improve accuracy in the pre-surgical diagnosis. The tested drugs include: methohexital (6), clonidine (7), pentylenetetrazol (8), thiopental (9) and opiates (10). Nevertheless, the results were not specific and adverse effects were poorly tolerated, decreased safety and precluded the use of many drugs. Etomidate is a non-barbiturate imidazole derivative hypnotic agent with a rapid onset, a short duration of action, and minor side effects associated with intravenous perfusion. It acts as a selective modulator of the gaminobutyric acid receptor A (GABAA). It is shown that etomidate can be safely used to activate epileptogenic activity (11-16)
In fact, Pastor et al. (2010) showed that etomidate induced interictal spiking activity ipsilateral to the ictal onset zone (IOZ) and can correctly lateralize 95% of the patients with TLE. Furthermore, as this drug facilitates the reliable identification of the IOZ, it could be used to diagnose patients who do not experience seizures during v-EEG recordings or influence decisions regarding the placement of intracranial electrodes. In a recent work, authors showed that etomidate perfusion induce changes in the underlying epileptic network related to the ones found during spontaneous seizures (17).
The current paper reviewed the evidence found to date, about the effects of etomidate in the interictal and ictal activities and epileptic network in patients with temporal lobe epilepsy, in an effort to support its potential value as an additional diagnostic tool for the preoperative assessment in patients with drug-resistant epilepsy.
2. Evidence Acquisition
2.1. Bioelectrical Activity Induced by Etomidate
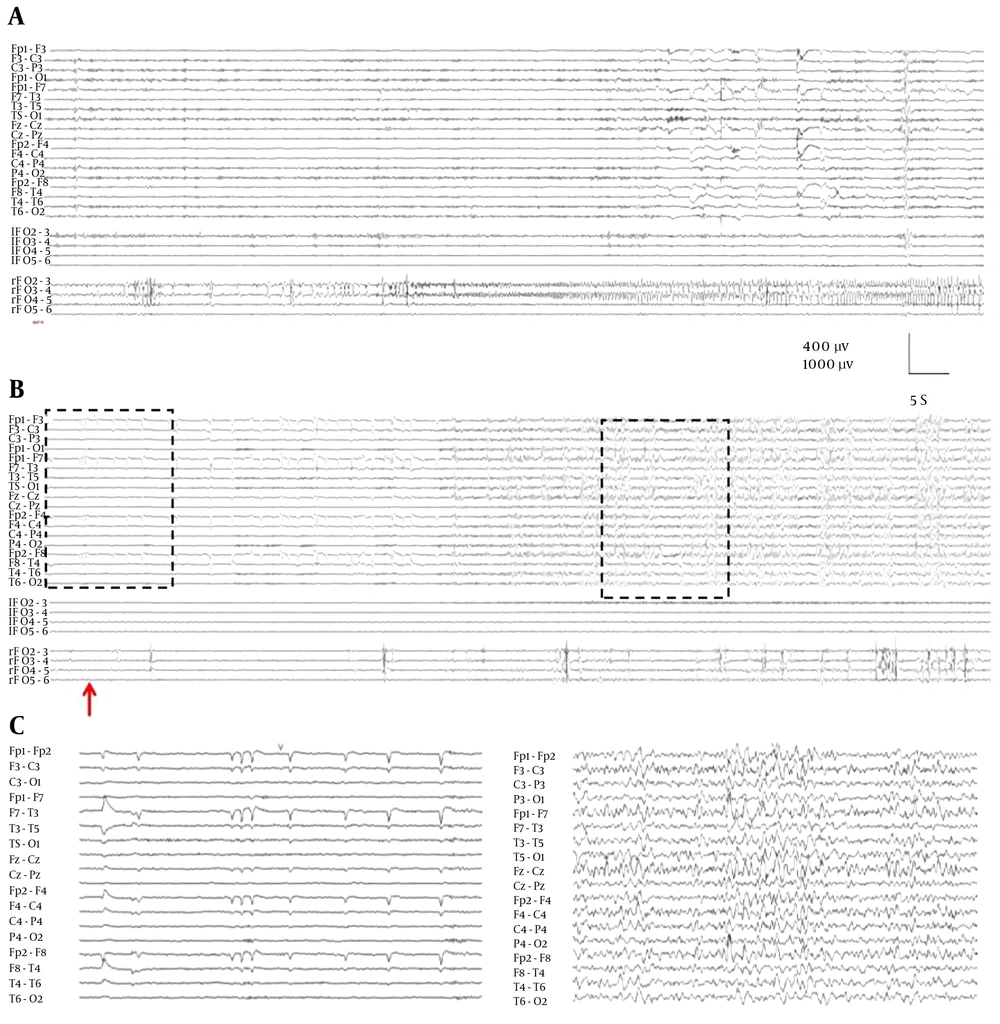
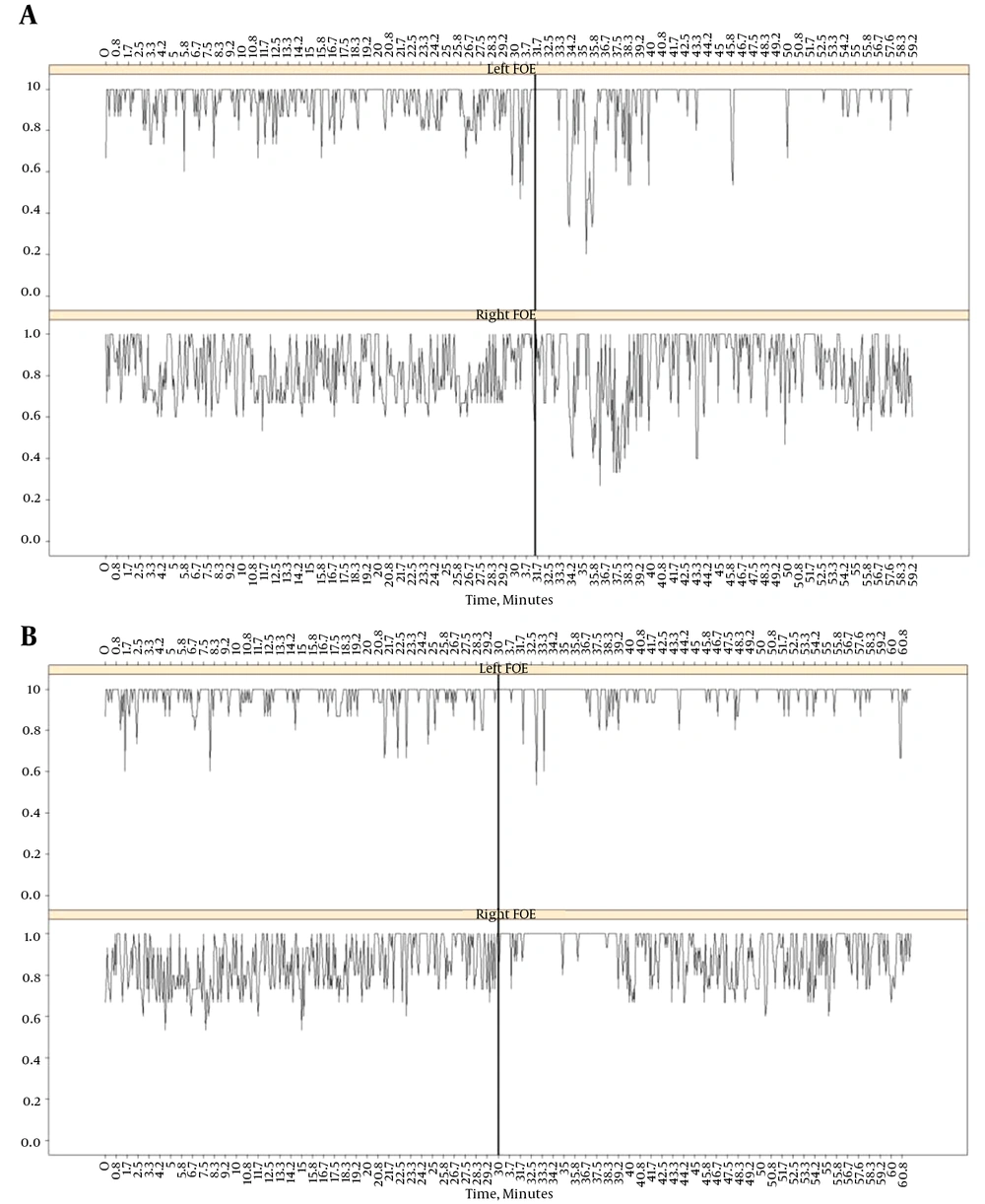
The changes induced after etomidate administration onto scalp EEG was described in an accurate way (14, 18, 19). In stage 1, small increases in amplitude and frequency were observed in the scalp EEG, followed by a generalized and high-amplitude delta activity (stage 2). Besides, studies using scalp EEG and foramen ovale (FO) electrode recordings described the high-voltage spikes and sharp-waves superimposed on this pattern (14). This interictal activity never appeared in different areas from the ones included in the IZ, i.e. they appeared only in the areas where interictal epileptiform discharges appeared in basal conditions. In the study, they compared the lateralization induced by etomidate with the IOZ identified spontaneous seizures recorded by VEEG + FO. Lateralization induced by etomidate perfusion was assessed through lateralization coefficient (LC), comparing the frequency of IED (spikes/min) in the left and right areas, considering the mesial and lateral areas.
Etomidate correctly identified the temporal lobe in 21 of 22 patients (Figure 1A). In addition, there was a strong correlation between the lateral and mesial values of the LC.
A, scalp and EFO record (left, top and right, bottom) during a spontaneous crisis originated in right mesial temporal region; B, record in the same patient during etomidate administration (arrow). In the right mesial temporal region a marked increase in irritative activity is observed; C, box detail records in scalp. Note the increase in scalp generalized delta activity in response to etomidate (right).
Likewise, the activity was grouped from the temporal areas where seizures occurred, and, the activity from areas where seizures did not occur. In order to access the kinetics of activity after etomidate infusion they used the equivalent to the first derivative for the discrete time-series of the frequency of spikes.
It was found that activity increased in both the mesial and lateral epileptic areas, although the increase was higher in the mesial region. Conversely, practically no increase was observed in the non-epileptic areas following etomidate administration (Figure 1B and 1C). Furthermore, the epileptic region displayed a higher frequency and also faster kinetics.
There are different theories about why etomidate activates the irritative area; one could be explained by the fact that etomidate almost exclusively acts on the β2 and β3 subunits of the GABAA receptor at clinical concentrations (20). Studies on rats show that β2 subunits seem to be preferentially located on GABAergic interneurons and excessive activation of these receptors could cause disinhibition of cortical activity and seizures. Studies on cultured astrocytes show that etomidate inhibits glutamate uptake, increasing the extracellular glutamate concentration to a level that can escape the synaptic cleft and activate extra-synaptic receptors. As a consequence, irritative activity increases (21). Besides, examination of slices obtained from patients with epilepsy revealed a decrease in the reversal potential of Cl- anions (22). This change could induce a depolarization instead of hyperpolarization after GABA release, driving the irritative activity.
2.2. Voltage Sources in Mesial Temporal Lobe
An early study aimed to examine different electrophysiological properties of voltage sources in the records obtained through FO electrodes (23) in mesial TLE. The primary purpose of the study was to ensure that pharmacologically induced irritative activity was generated by the same cortical regions observed under basal conditions. It was found that etomidate could induce irritative activity in the mesial temporal region in an extremely specific manner, i.e. triggering the same cortical structures responsible for spontaneous IED.
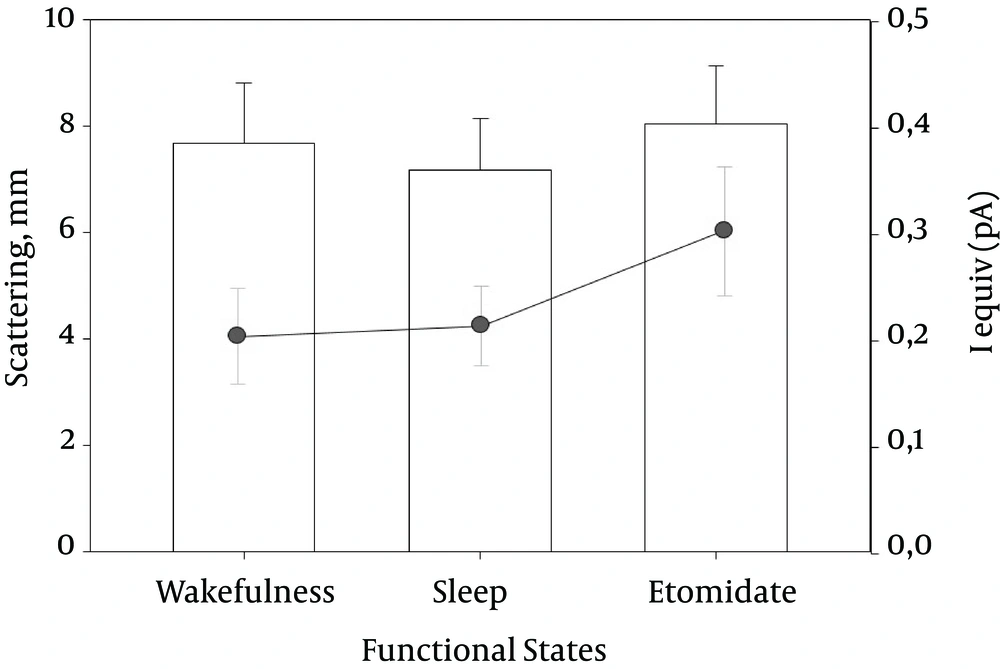
A more recent study by the authors also proposed to analyze the electrophysiological properties of interictal activity induced by etomidate (24), and consider whether it matches or not with interictal activity at baseline conditions, it also considered its relationship with ictal activity. A classical electrostatic theory was applied to derive mathematical expressions of IED (i.e. spikes and sharp waves) recorded using FO (25, 26). An infinite and homogenous volume conductor and an isotropic medium were assumed.
It was found that the scattering and iequiv were similar for interictal and pharmacologically induced activities (Figure 2A), suggesting that the single source model could reliably explain interictal and recorded etomidate-induced spiking using FO electrodes in TLE.
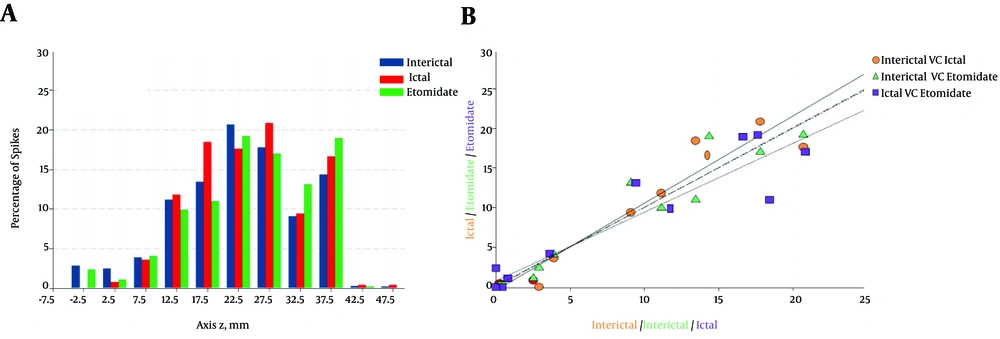
Furthermore, the spatial distribution of the voltage sources responsible for interictal baseline activity was similar to the ones obtained for etomidate-induced activity. In addition, there was a close correlation between areas where irritative activity occurred under basal conditions and the ones induced through etomidate. The analysis of normalized sources showed that 76.9% of the patients show localised distribution of voltage sources at either the interictal baseline or in the presence of etomidate. A more scattered distribution along the axis z, with IP20 - 75 > 20 mm (Rank: 21, 5 - 28,3 mm), was observed in 22.1% of the patients. A linear relationship was also found among the three functional states (Figure 2B).
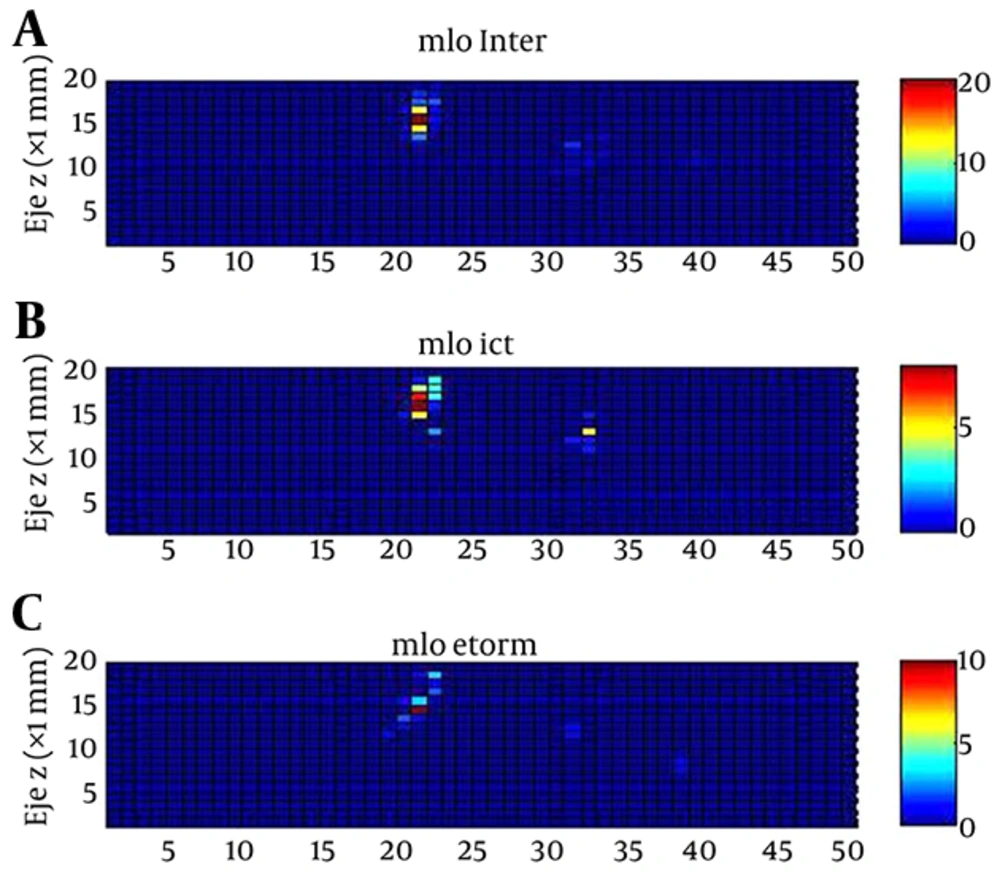
In order to analyze the topographical relationship between pharmacologically induced activity and that of the IOZ, the degree of superposition was established among sources obtained from interictal, ictal and pharmacologically induced activities (24). Thus, the zr-plane or configuration space, was divided into small non-overlapping patches of Δx × Δy, where Δx = 1 mm, Δy = 1 mm and area = 1 mm2. This tessellation covers the entire theoretical surface. The active area was defined (where current-source appears, irrespective of the functional state) in two steps: (i) patches where spiking activity 1 spike/mm2; and (ii) spikes less than a minimum distance (dmin) from other spikes. The dmin = 1.44 mm was selected (Equation 1). In this way, spurious spikes were excluded from the active area and the total area where the current sources appear for each type of activity and the superposition between them was obtained.

There was a great degree of superposition of the current-source topography for interictal, ictal and pharmacologically induced activities. For each pair of activities, the area and the spikes of superposition were interictal-ictal: 10.0 ± 1.5 mm2 (31%) and 74.7 ± 12.6 spikes/ mm2 (12%); interictal-etomidate induced: 7.3 ± 1.4 mm2 (24%) and 74.7 spikes/ mm2 (12%) and ictal-etomidate induced: 4.7 ± 1.0 mm2 (24%) and 30.0 spikes/mm2 (45%), respectively.
2.3. Seizure Networks Dynamics and Etomidate
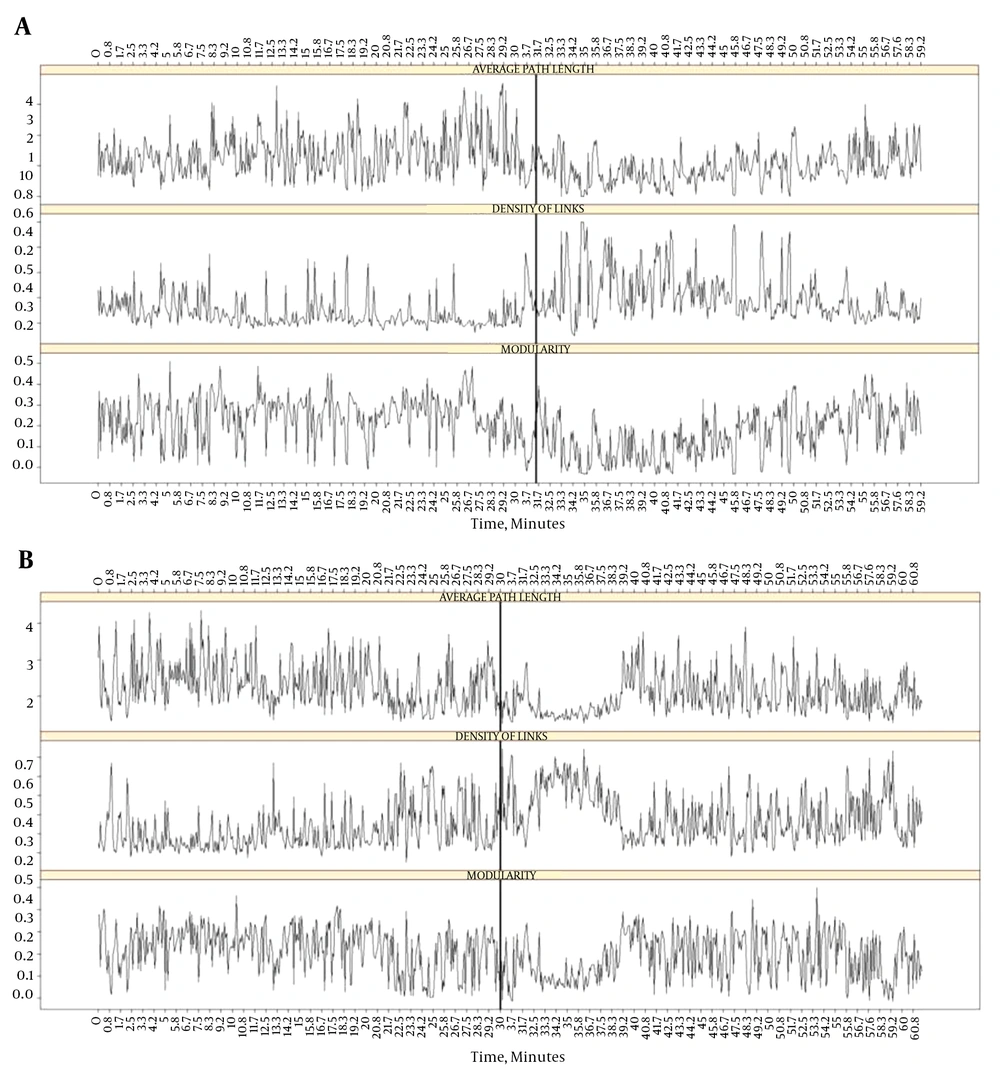
A recently published work addressed the very relevant issue whether temporal lobe seizures produce network changes comparable to the ones elicited by etomidate administration (17). Scalp and FOE recordings of nine patients with temporal lobe epilepsy were analyzed employing analysis of complex networks. To evaluate the global aspects of the cortical dynamics, several variables in network theory were calculated (see Vega-Zelaya et al., 2015b for more details): the average path length (APL), density of links (DOL), average clustering coefficient (ACC), modularity (Mod) and spectral entropy (SE).
The APL is calculated as the average of all the shortest paths in terms of the number of steps along the network nodes between every pair of vertices in the network. In this sense, low values for the APL imply efficient and fast communication across the network functional topology. The DoL is the ratio of the actual number of edges in the network to the number of all possible edges between the network nodes. A network with many links or a high density of links implies highly synchronous behavior at both short and long distances. A high DoL decreases the APL in the network because a greater number of edges allows for a greater number of alternative paths between the two nodes. The ACC characterizes local connectedness in a network by measuring how well neighbours are connected in a given node. Modularity (Mod) measures how well a given partition or division in a community in a complex network corresponds to a natural or expected sub-division. The averages for each community were calculated, and the overall spectral entropy was defined as the average over all communities.
Two different groups were evaluated. The first group corresponded to the whole network (scalp + FOE), and the second one corresponded to the mesial sub-networks (FOE).
Based on the previous published results regarding ictal network dynamics (27, 28), it was expected to find that the ACC and DoL would increase with respect to the pre-ictal values both during the seizure and etomidate administration. Since the increase in the DoL would increase the available paths, it was assume that they lower the APL. Also it was expected that the increase in DoL would make intra-regional links more available and lower the modularity. Likewise, since anesthetics are known to lower SE (29), it was proposed that SE would decrease in both cases.
Considering the whole network, it was observed that etomidate fits better to the previous statement, except for modularity (Figure 3A). In four of nine patients, the APL did not decrease during etomidate administration. As expected, SE decreased after etomidate administration in all patients and also decreased during the seizure (Figure 3B), implying that the frequency content during the seizure reduced to a few relevant ones. During the seizures, the network measures demonstrated disparate results. DoL and Mod best followed the previous statement (in seven out of nine seizures) and the worst measures were APL and ACC.
A, scattering along the antero-posterior theoretical axis; B, linear regression of the location of current sources for pairs of functional states, shown in the same colors. The linear regression of interictal/ictal is y = 1.09x - 0.50, r = 0.930, for interictal/etomidate y = 1.01x + 0.01, r = 0.922 and for ictal/interictal y = 0.856x + 0.952, r = 0.867.
Therefore, it was found that the changes showed similar behavior in both situations: an increase in the density of links (DoL) and a decrease in the spectral entropy (SE). The other measures did not show higher coincidences.
Regarding the mesial sub-networks, etomidate and seizures produce similar changes (Figure 6). Specifically, during etomidate, APL increases and DoL decreases on the ipsilateral side of the seizures than on the contralateral side, mimicking the effect of seizures on these network measures. Previous findings already showed impaired connectivity in the ipsilateral side in patients with TLE (30-32). Thus, decreased connectivity was shown in the mesial structures, ipsilateral to the SOZ, compared with the contralateral side during resting-state fMRI studies (30, 31) and during the inter-ictal stage through electrophysiological methods (32). All these results were consistent with recent reports suggesting that a loss of connectivity within specific network structures are involved in seizure generation (33, 34).
3. Results
Etomidate might be considered as a useful tool to identify the EZ and IOZ in patients with TLE. It is a safe, efficient and well tolerated drug. The described side-effects are not severe and can be monitored adequately by scalp EEG. The most frequent ones include, myoclonus affected small distal muscles, pain, paradoxical and euphoric reactions (14). Hemodynamic effects are observed (35) with higher doses and include tachycardia and increases in mean arterial pressure (19).
This method can activate the EZ, inducing irritative activity only in the areas where spontaneous activity was recorded (14). Additional evidence shows that interictal, ictal and etomidate-induced activities greatly overlap; indicating that the physiophatological mechanisms are similar, and that presumably the cortical areas corresponding to each of the activities are likely the same or closely related (24). These findings are of great significance, since etomidate might facilitate the identification of different patients regarding the activity topography, and it can be used for new surgical approaches, e.g., high-definition radiosurgery in patients with well-localized sources. An important issue to emphasize is that etomidate induced changes are produced with a low probability to eliciting seizures (14). This is an important advantage because it was observed that etomidate reproduce the same biophysical characteristics at the same location as the seizures do, without triggering an ictal episode.
Moreover, it is proved that etomidate uses the same neural network that the seizures do. Therefore, the findings show that the changes etomidate produces in the limbic network are very similar to the ones occurring during temporal seizures. The most remarkable finding in relation to the epileptogenic network is that mesial synchronization ipsilateral to the IOZ is reduced compared with those of the contralateral side under basal conditions in both situations; implying that etomidate and seizures produce similar changes in the mesial sub-networks. From the viewpoint of a functional brain network in epilepsy, etomidate could be used in research to increase knowledge about network dynamics during seizure activity.
4. Conclusions
This method offers several benefits as a diagnostic tool during the pre-surgical evaluation of patients with TLE. It may help to confirm results derived from other methods. It can facilitate diagnosis in patients without seizures during v-EEG recording. Besides, it has the potential to influence decisions regarding the need to use intracranial electrodes, and also the best placement of those electrodes. Considering all the mentioned points this technique can be used to better define the region resected during epilepsy surgery.