1. Background
Long-term physical exercise is an important method to promote health with relatively low cost (1-4). However, thousands of people die annually from diseases caused by physical inactivity worldwide (5, 6). One of the greatest current challenges is to promote adherence to physical activity (7, 8). There is supporting evidence that behavioral response, mediated by a single bout of exercise, predicts participants engagement during the next six to 12 months (9). Therefore, the term adherence to exercise refers to chronic issues; the study of acute effect of different courses of exercise on subjective and neurobiological behavioral variables becomes relevant.
According to Ekkekakis and Petruzzello (10), there is a U-shaped association between aerobic exercise intensity and affective state, characterized by the interaction of behavioral factors like happiness, euphoria, relaxation and stress reduction. This theory suggests that the optimal effort intensity with the most positive affective response would be a moderate one that is near the ventilatory threshold (~ 65% VO2max) (11). However, a meta-analysis conducted by Reed and Ones (12) showed that low-intensity exercises with a duration of 35 minutes or less induced a strong activation of positive affect. Contrarily, recent researches have shown that an exercise protocol, including high-intensity intervals generated more pleasure than a program with continuous stimulus of moderate intensity (13). There is also evidence suggesting that positive behavioral outcomes tend to occur after exercise at a self-selected intensity (14). Although, intensity seems to be the main regulator of variable affective responses, different results are found in the literature. Observing more objective variables can contribute to a broader discussion on the issue.
Various techniques like functional magnetic resonance imaging (fRMI), positron emission tomography (PET), single photon emission tomography (SPECT) and electroencephalography (EEG) are used to study possible changes in brain activity in humans. Standardized low-resolution brain electromagnetic tomography (sLORETA) is another method which represents a novel EEG approach, allowing bioelectrical location determination of three-dimensional signals and not just restricted cortical events (15). Mathematical models used by sLORETA are able to estimate the intracerebral volume and infer the specific activity of neuro-anatomical structures like Brodmann areas (BA). Although, this technique is economic and noninvasive with good spatial and temporal resolution compared to fMRI and PET (16-18), however, few studies used this method in the field of exercise, worldwide. Schneider et al. (19) found greater activity in areas associated with brain reward system, doing the preferred effort (treadmill) and cycle ergometer, in 11 recreational young runners who underwent three different types of maximal test (treadmill, cycle ergometer and upper body ergometer). In another study by the same authors on 22 recreational runners aged 21 to 45 years, activation of areas related to positive emotions was found (20). However, no behavioral subjective scales were applied in such studies. In our laboratory, we found greater activity in areas related to the integration of affective and cognitive information in 19 active young male, along with a reduction in total mood disturbance, followed by a 20-minute moderate effort condition at 80% heart rate reserve (21).
2. Objectives
The objective of this study was to investigate the acute response of submaximal, maximal and supramaximal efforts, on a cycle ergometer, on subjective states of mood and anxiety, and cortical activity of physically active young males. The sLORETA is a very modern technique in the area of exercise, which can contribute to further discussions on cortical phenomena, associated with subjective behavioral variables. Our hypotheses were; 1) Moderate exercise improves behavior, unlike very intense efforts and 2) Changes in subjective mood and anxiety are accompanied by specific changes in cortical activity.
3. Materials and Methods
3.1. Sample
We recruited 10 healthy men, practicing regular physical activity (at least one-hour sessions of aerobic, muscular strength and endurance exercises three times a week). Their age, weight and height ranged between 20 and 27 years, 62 and 82.5 kilograms and 1.66 and 1.85 meters, respectively. Those who were illiterate, colorblind, smoker, with mental illnesses or using any psychoactive substances (like antidepressants and anxiolytics) were excluded. One subject was excluded because of not completing the maximal effort protocol.
3.2. Experimental Procedure
Participants received written information about adopted routines, procedures and possible risks, through the consent term. This study was approved by the Ethics Committee of the Universidade Gama Filho (protocol number 172.2011). All volunteers were instructed not to consume stimulants and central nervous system depressants, like foods containing caffeine or alcohol, do not engage in strenuous exercise in 48 hours before the tests, not eating within three hours prior to interventions and sleep for at least seven hours the night before the tests. An anamnesis was applied prior to each protocol, to verify that all instructions were followed and whether there were other important changes.
The subjects performed three different combinations of intensity and duration of exercises on a cycle ergometer (Monark®, Brazil) including submaximal, maximal and supramaximal. The treatments were randomized with one-week intervals and always at the same time of day. Wasserman test was adopted for maximal effort (22). After three minutes of warming-up, there was a gradual increase in workload (10% VO2max) under a fixed rate (by using a metronome audiovisual), according to body weight and age (23) at each stage of one minute. Oxygen uptake (VO2), carbon dioxide output (VCO2), minute ventilation (VE) and respiratory exchange ratio (RER)were determined by indirect calorimetry (Vista Mini-CPX®, VacuMed®, Ventura, CA, USA). The subjects used a pneumotachograph (Hans Rudolph Inc®, Kansas City, MO, USA) inserted in a turbine with bidirectional gas flow (MIR Turbocharge®, VacuMed®, Ventura, CA, USA). Before each test, ergospirometer was calibrated in a closed loop. Perceived exertion was measured using 6- to 20-point Borg Scale. The test was stopped in case of three of the following criteria: VO2 plateau ≤ 150 mL/min or 2 kg, mL/min, heart rate ≥ 90% predicted by age (220-age), Borg Scale ≥ 18, RER ≥ 1.15 and voluntary failure to maintain the cadence.
The submaximal effort went on for 30-62 minutes ± 9% VO2max. This intensity was determined from the anaerobic threshold, obtained by two experienced researchers through the respiratory equivalent (EQV), (11) and simplified V-slope methods (24). The EQV was defined as the moment when the ventilatory equivalent for oxygen consumption (VE/VO2) increased, without a concomitant increase in the ventilatory equivalent for the excretion of carbon dioxide (VE/VCO2). [The EQV is the moment when there is an increase of the ventilatory equivalent for oxygen consumption (VE/VO2) without the concurrent increase in the ventilatory equivalent for carbon dioxide excretion (VE/VCO2)].The simplified V-slope method was examined in a Cartesian coordinate graph, with VO2 in the abscissa axis and VCO2 in the ordinate axis.. The VO2 at the anaerobic threshold was determined as the breakpoint in the plot of CO2 as a function of VO2, where the slope becomes > 1 by the simplified V-slope method (24).The load and the cadence remained stable during the whole test using an audio-visual metronome. Respiratory gases and perceived exertion sensations were checked following the same procedures described above. The supramaximal effort consisted of 30 seconds sprint against a workload corresponding to 0.075 kp body mass (kg-1) (25). [The subjects were instructed to pedal progressively for about five seconds to allow the workload to be adjusted manually and to remove the influence of inertia on the performance tests]. During 30 seconds, the subjects were verbally encouraged to pedal as fast as possible. The number of revolutions per minute was used to calculate the load in Watts obtained through a manual counter. The Borg Scale was used to check the perceived exertion sensations immediately after exercise.
The behavioral and cortical variables were analyzed before and immediately after the treatments. We used Profile of Mood State (POMS) (26) and State-Trait Anxiety Inventory (STAI) (27) scales to assess mood and anxiety, respectively. Assessment of cortical activity is described below. Figure 1 illustrates the experimental design of this study.
3.3. Acquisition, Reduction and Analysis of Electroencephalographic Signals (EEG)
EEG was recorded for five minutes in a relaxed sedentary position from 20 monopolar electrode sites (Fz, Cz, Pz, Oz, Fp1, Fp2, F3, F4, F7, F8, C3, C4, T3, T4, T5, T6, P3, P4, O1, O2). International 10/20 system (referred to linked earlobes) was used for electrode placement with a Brain-tech 3000 (EMSA-Medical Instruments, Brazil). Eye-movement artifact was monitored with a bipolar electrode montage, using two 9 mm diameter electrodes, attached superiorly to and on the external canthus of the right eye. Following digital filters were used: notch (60 Hz), high-pass of 0.16 Hz and low-pass of 35 Hz. Data was digitized at 240 Hz, with a 12-bit resolution. We used the linked ears reference given that this is the routine in our laboratory. After that an average reference was used a posteriori.
EEG data was analyzed using sLORETA, in which the image represents the standardized electric activity of each voxel in neuro-anatomic Montreal Neurological Institute (MNI) space as the exact magnitude of the estimated current density. Anatomical labels like Brodmann areas, are also reported using MNI space, with correction to the Talairach space (28). The intracerebral volume is partitioned in 6239 voxels at 5 mm spatial resolution. A minimum of 120 epochs of artifact-free resting EEG were exported for further analysis using the sLORETA software provided by the KEY Institute for Brain-Mind Research (University Hospital of Psychiatry, Zurich, Switzerland; http://www.uzh.ch/Keyinst/NewLORETA/LORTA01.htm). Next, data was log-transformed and calculated in delta, theta, alpha and beta activity for each subject in each treatment. Cross-spectra of each subject and for each frequency band were transformed to sLORETA files by using the sLORETA transformation matrix.
3.4. Statistical Analysis
Friedman nonparametric test and Wilcoxon were used for POMS and STAI deltas values (post-pre exercise) to compare the differences between treatments. The sLORETA software was used to analyze EEG activity. After a critical threshold was defined (t critical), voxels with statistical values exceeding this threshold were accepted. The omnibus hypothesis (that all the voxel hypotheses are true) was rejected if a voxel value exceeded the critical threshold for P < 0.05 defined by 5000 randomizations. Voxel-by-voxel t values in Talairach space were displayed as Statistical Parametric Maps (SPMs).
4. Results
The values (mean and standard deviation) of heart rate, oxygen consumption, power and perceived exertion sensations effort were 144 ± 7 bpm; 2.48 ± 0.37 L.min-1; 150 ± 26 Watts; 13 ± 2 Borg (submaximal effort), and; 185 ± 7 bpm; 4.03 ± 0.41 L.min-1, 303 ± 46 Watts, 20 ± 0 Borg (maximal effort). Maximal effort lasted from eight to 12 minutes. In the supramaximal treatment a power of 631 watts ± 4 and Borg of 20 ± 0 were observed.
The results of mood (POMS) and anxiety (STAI) are shown in Table 1. There was a difference in total mood disturbance between all of treatments (P = 0.001). Submaximal effort reduced the total mood disturbance, while maximal effort increased it and supramaximal effort caused an even greater increase. The other variables of POMS did not change significantly. Regarding anxiety, there was a significant difference between submaximal and supramaximal (P = 0.012), as between maximal and supramaximal (P = 0.037), indicating that it is increased in supramaximal effort compared to the other two protocols, which did not differ.
| Variables | Pre | Post | Delta | X2 | P | Wilcoxon |
|---|---|---|---|---|---|---|
| Tension | - | - | - | 4.514 | 0.105 | - |
| Sub | 3 (3; 5) | 3 (2; 4) | 0 (-2; 0) | - | - | - |
| Max | 3 (2; 5) | 5 (3; 6) | 1 (0; 3) | - | - | - |
| Supra | 4 (3; 4) | 6 (4; 7) | 2 (0; 4) | - | - | - |
| Depression | - | - | - | 4.846 | 0.089 | - |
| Sub | 0 (0; 1) | 0 (0; 0) | 0 (-1; 0) | - | - | - |
| Max | 0 (0; 1) | 0 (0; 2) | 0 (0; 1) | - | - | - |
| Supra | 0 (0; 1) | 0 (0; 2) | 0 (0; 1) | - | - | - |
| Hostility | - | - | - | 3.556 | 0.169 | - |
| Sub | 0 (0; 1) | 0 (0; 0) | 0 (-1; 0) | - | - | - |
| Max | 0 (0; 0) | 0 (0; 1) | 0 (0; 1) | - | - | - |
| Supra | 0 (0; 0) | 1 (0; 2) | 0 (0; 2) | - | - | - |
| Vitality | - | - | - | 3.706 | 0.157 | - |
| Sub | 12 (10; 16) | 16 (11; 17) | 1 (-1; 3) | - | - | - |
| Max | 14 (12; 18) | 14 (7; 15) | -1 (-7; 2) | - | - | - |
| Supra | 14 (11; 17) | 11 (4; 17) | -2 (-6; 0) | - | - | - |
| Fatigue | - | - | - | 4.941 | 0.085 | - |
| Sub | 1 (1; 3) | 3 (1; 4) | 0 (-1; 3) | - | - | - |
| Max | 3 (3; 5) | 13 (6; 14) | 6 (3; 10) | - | - | - |
| Supra | 2 (1; 4) | 8 (6; 16) | 6 (3; 13) | - | - | - |
| Confusion | - | - | - | 4.323 | 0.115 | - |
| Sub | 4 (2; 6) | 4 (3; 5) | 0 (0; 1) | - | - | - |
| Max | 2 (1; 5) | 4 (3; 7) | 2 (0; 3) | - | - | - |
| Supra | 4 (2; 5) | 7 (4; 9) | 3 (1; 4) | - | - | - |
| Total mood disturbance | - | - | - | 13.941 | 0.001 | - |
| Sub | 96 (92; 111) | 92 (90; 103) | -2 (-10; 2) | - | - | Sub*Max (P = 0.012) |
| Max | 98 (89; 115) | 107 (98; 125) | 9 (5; 12) | - | - | Sub*Supra (P = 0.008) |
| Supra | 94 (90; 104) | 118 (106; 126) | 18 (5; 28) | - | - | Supra*Max (P = 0.049) |
| Anxiety (STAI) | - | - | - | 10.364 | 0.006 | - |
| Sub | 33 (29; 34) | 30 (25; 36) | 1 (-3; 1) | - | - | Sub*Supra (P = 0.012) |
| Max | 33 (27; 36) | 34 (31; 44) | -1 (-2; 7) | - | - | - |
| Supra | 29 (27; 36) | 44 (33; 48) | 4 (2; 15) | - | - | Supra*Max (P = 0.037) |
aDelta values (post pre moment) expressed in median and quartiles (1st; 3rd)
bAbbreviations: Max, maximal effort; Sub, submaximal effort; Supra, Supramaximal effort; Treat, treatment
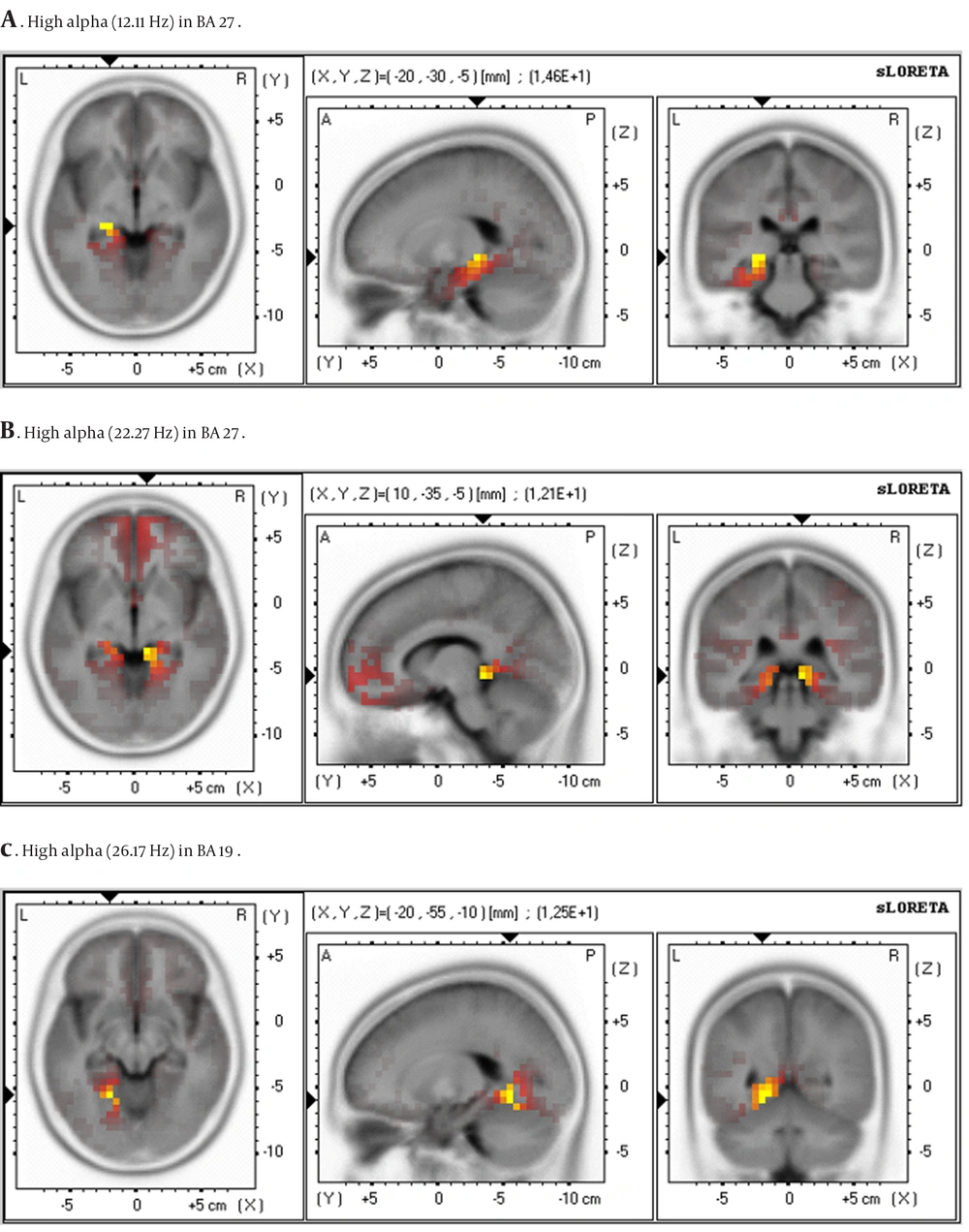
Through sLORETA analysis, significant increases in maximal effort at the post moment were seen, when compared to pre, for high alpha (14.62 Hz) and high beta (12.11 Hz) in BA 27 (parahippocampal gyrus-limbic lobe), and high beta (12.46 Hz) in BA 19 (parahippocampal gyrus-limbic lobe). Figure 2 illustrates the changes mediated by maximal effort. No significant differences were found between pre and post submaximal and supramaximal efforts.
Statistical parametric maps (voxel-by-voxel) of sLORETA differences comparing pre and post moment in maximal effort, A. High alpha (12.11 Hz) in BA 27, B. High beta (22.27 Hz) in BA 27, C. High beta (26.17 Hz) in BA 19,19Red and yellow colors indicate significant increase in POST measurement shown for high alpha in BA 27 (A), high beta in BA 27 (B), and high beta in BA 19 (C). For X (- represent left, + represent right), Y (- represent posterior, + represent anterior), Z (- represent inferior, + represent superior).
5. Discussion
The aim of this study was to investigate acute effect of three different patterns of exercise on a cycle ergometer on mood, anxiety and cortical activity of physically active young males, through manipulation of intensity and duration of the effort. The submaximal effort reduced the total mood disturbance. The maximal effort, of short and progressive character, activated areas of the brain related to pleasure and reward, but increased total mood disturbance. The supramaximal effort increased total mood disturbance and anxiety.
We agree with the review of Ekkekakis and Petruzzello (10) which suggested greater benefits in behavioral response to acute moderate effort. Although Bartlett et al. (13) concluded that a session with intervals containing maximal stimuli is a good strategy to motivate young and physically active subjects compared to a continuous protocol, but it is too early to affirm that short-term intense stimuli can promote behavior and adherence to exercise. In the present study, we reported a worsening in mood and anxiety due to a single supramaximal effort. Richards et al. (29), following a similar protocol, reported occurrence of dizziness and nausea in some subjects. We also observed the same in three subjects.
We did not support some findings in the literature regarding other factors of the POMS. For example, Fumoto et al. (30) submitted 10 healthy physically active young (nine men and one woman) to 15 minutes at moderate intensity on a cycle ergometer and found a significant reduction of tension and confusion, and a tendency to increase vigor factor and reduce depression factor. Woo et al. (31) submitted 16 young women to moderate intensities (61.4% VO2max) in three different durations (15, 30 and 45 minutes) and found a higher score after a half hour vigor activity. We found no significant differences regarding tension, depression, hostility, vigor, fatigue and confusion in moderate effort.
sLORETA revealed an increase of high alpha and beta activity in the parahippocampal gyrus of the limbic lobe (high alpha and beta in BA 27, and high beta in BA 19) in maximal exercise. These areas are associated with reward system, pleasure, positive emotion and memory. Schneider et al. (19) submitted 11 recreational young runners to three different types of maximal effort as treadmill, cycle ergometer and upper body ergometer. The authors found that the most preferred type of effort by the subjects, induced an increase of alpha in frontal (BA 6, 8 and 9) and limbic (BA 24 and 32) lobes, as well as beta in parietal lobe (BA 7). The cycle ergometer exercise increased alpha and beta in parietal (BA 7) and limbic (AB 23 and 31). Upper body ergometer effort increased alpha in frontal lobe (BA 45) and beta in parietal lobe (BA 7 and 40). We agree with the authors because we also found changes in the limbic lobe, especially in the same type and intensity of effort. However, we did not use the cap system for electrode placement, which gives an optimization of time in recording EEG signals. After 15 minutes, the time it takes to start EEG collecting, Schneider et al. (19) reported increases of alpha in frontal (BA 6 and 9) and limbic (BA 24 and 32) lobes, regions associated with reward and pleasure, only in the cycle ergometer (upper body ergometer increased alpha and beta in the temporal lobes). Although they found an increase of alpha in limbic lobe, they observed no changes in beta, as we found in the present study. Another relevant aspect was the absence of subjective measures of behavior parallel to biological measure in the study of Schneider et al. (19) to allow a fuller discussion of findings.
In another study, 22 young recreational runners underwent a maximal effort on treadmill and there were significant increases in low alpha in the left frontal gyrus (BA 8), related to the processing of positive emotions (32). However, after 15 minutes, which represents a real comparison with our protocol, there was a reduction in high alpha, low beta and gamma in areas accounting for language (BA 18, 20, 21 and 22). Moreover, Schneider et al. (32) in this study did not use subjective scales to analyze the behavior changes. Moreover, Moraes et al. (21) found significant changes in sLORETA followed by a reduction in total mood disturbance in 19 young active (11 Men and 8 Women) after 20 minutes of moderate exercise on a cycle ergometer. Increases in low alpha in the anterior cingulate cortex (BA 24), low beta in the same region (BA 33) and high beta in posterior cingulate cortex (BA 23) were observed. In this study, we observed a lower total mood disturbance after submaximal effort, but no significant electrophysiological change was observed by sLORETA.
Improvement in brain areas mediated by high intensities, especially in subjects with high level of physical fitness, could be justified by the postulates of Solomon (33). According to a study performed on a group of paratroopers about their emotional experiences, the feeling of fear to jump was followed by a very intense pleasure when the equipment is opened. The opposite feeling of fear, corresponding to the relief and pleasure, represents a resource to stabilize emotion, as well as a psychological reinforcement encouraging the repetition of action. This emotional duality has given rise to the opponent process theory or hedonistic hypothesis, later expanded to other situations, including intense physical exercise. High intensities of exercise in active subjects can promote positive reinforcement. We know that endurance and adventure athletes, or subjects with high levels of physical fitness, undergo numerous challenging sessions that cause fear or pain, repeating them probably due to “the costs of pleasure and the benefits of pain" (33). In the present study, although there was an increase in total mood disturbance immediately after maximal effort, activation of the brain reward system was verified by sLORETA. An important limitation probably explaining the difference in results between the subjective and objective variables was the time of application of the behavioral scales, which occurred immediately after the treatments, and the capture of EEG, which performed 15 minutes after the end of the sessions, time required for the placement of the electrodes. We do not know if the supramaximal effort, given the exposed context, could present some sort of neurobehavioral benefit after a long period (for example 60 minutes after the 30 seconds sprint).
We conclude that acute changes in mood, anxiety and cortical activity are dependent on the effort intensity. Physically active youth, but not athletes, show positive results on subjective or neurobiological behavioral variables in submaximal and maximal exercises. This information may be useful to promote adherence to regular physical activity.