In conclusion, PAs can be divided to functional and non-secreting. While a great deal of research attention has been devoted to the control of hormone secretion from these adenomas, less attention is paid to molecular events that give rise to the development of such tumours.
Prior research has shown that the deregulation of programmed cell death appears to play a crucial role in multistep tumorigenesis (
3,
4). A number of candidate genes, among which p53 and bcl-2 deserve special mention, contribute substantially to the regulation of programmed cell death (
13,
17). Not least important, the deregulation of these genes is essential in the development of malignant tumours (
18,
19).
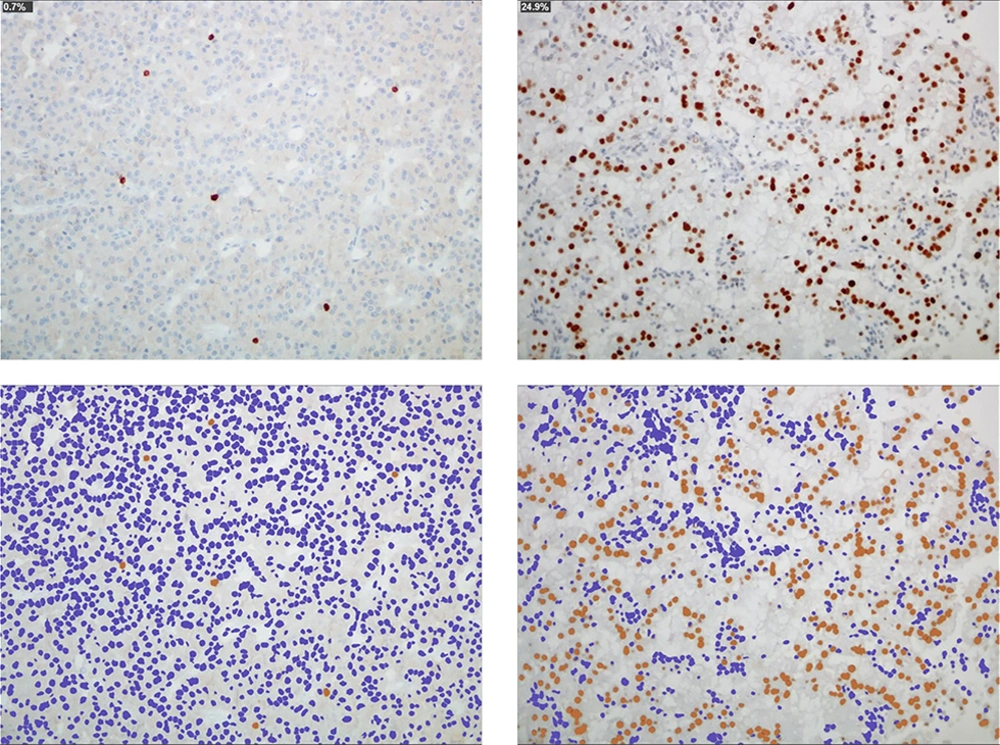
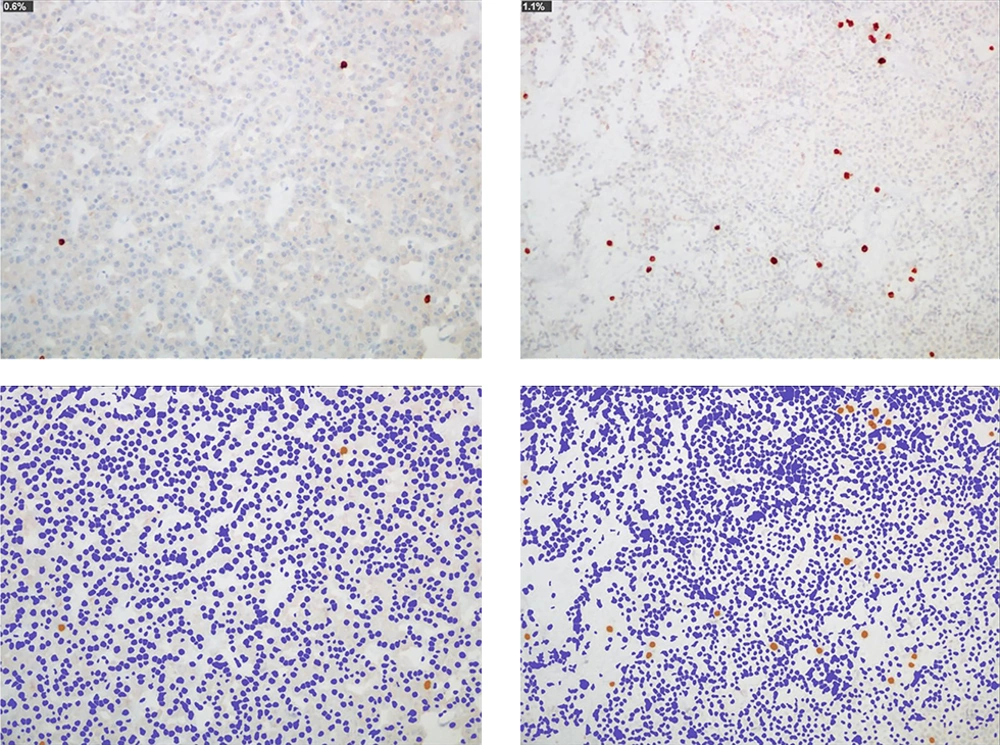
The research reported in this paper concerns the expression of the p53 and bcl-2 oncoprotein human pituitary tumours. Out of a total sample of 51 PAs, p53 immunoreactivity was detected in 35 and bcl-2 immunoreactivity in 26 PAs. Still other 19 cases turned out to be positive with regards to both oncoproteins, and thus it is proposed that there is an association of expression between p53 and bcl-2.
P53 is crucial in protecting the integrity of the cellular genome as well as defending the cell from malignant transformation. A more stable protein, which can now be effectively detected via immunohistochemistry, may be produced through the mutation of the gene. The data demonstrated p53 immunoreactivity in 68.63% of adenomas. At the same time, no evidence was found concerning immunoreactivity in any of the normal pituitary tissues. These findings point to the apparent disconnect between earlier studies showing that p53 gene abnormalities constitute the minority of PAs and the majority of pituitary carcinomas (
18), and the data showing a high level of p53 immunoreactivity. The reason for such a disparity in the results may be related to the high number of “atypical” PAs in the study group. Indeed, in line with Wierinckx et al.’s (
19) results, the current study observed that adenomas with immunohistochemical positivity for prolactin were associated with higher expressions of p53. In another study (
20), Ki-67, p53, and bcl-2 expressions were found significant in the PRL-secreting group. The routine use of p53 immunohistochemistry for PAs was controversial. These mixed findings seem to suggest that p53 cannot be considered an independent prognostic factor capable of determining the aggressive behavior of pituitary tumors.
The expression of bcl-2 in a number of PAs leads to the conclusion that it plays an important role in their pathogenesis. The reason for this is that in these kinds of tumors, which are slow growing, it could represent an initial event leading to indolent tumour growth. The current research found bcl-2 immunoreactivity in 50.98% of adenomas as compared to 75% of Ozer et al. (
7) as well as in 20% of normal pituitary tissue, thus pointing to its potential as an important marker in the event of tumor progression. Bcl-2 is of utmost importance for the regulation of apoptosis. It has been well-established that bcl-2 guides cell survival even in the event of low rate of cell proliferation, hence providing a growth advantage that may in turn give rise to neoplastic transformation.
Despite the fact that studies investigating the possible co-localisation of p53 and bcl-2 in individual cells were beyond the intended scope of this research, it is nevertheless worth pointing out a notable association concerning the presence of p53 and bcl-2 immunoreactivities across tumours. The above-mentioned co-expression could indicate that bcl-2 establishes a mechanistic basis for oncogenic synergy that takes place between these two proto-oncogenes in PAs. The establishment of such a basis is achieved by lessening the apoptotic effects of deregulated p53 expression without affecting its ability to stimulate continuous cell growth. Decreased expression of bcl-2 and accumulation of p53 has been associated with bromocriptine-induced apoptosis in PA cells (
21).
One study revealed that the p53 protein was co-expressed with the bcl-2 protein during the formation of the normal pituitary gland (
10). In another study, Ahlbom et al. demonstrated that the cell remodeling of the anterior pituitary, after the termination of lactation, occurs through the process of apoptosis and involves changes in Bax, bcl-2, and p53 (
22). It could be speculated that in regions of intense cell division, the expression of p53 might be necessary to prevent inappropriate and uncontrolled proliferation that is generally related to severe modifications in cell alignment, as well as the appearance of different disorders.
While Ozer et al. (
7) suggested a significant relationship between the proteins related to apoptosis and hormonal function in PAs, Green et al. (
23) found no significant association between apoptosis and p53 protein expression.
Although the sample size (51 patients, with a wide range of age and different histological groups of PA) was larger relative to other studies in this area, it is nevertheless still limited, allowing for limited ability to detect significant differences between groups. The study’s limitations include the retrospective nature of the data collection that can introduce bias because of differential loss to follow-up. Although the current results differed from those reported in earlier studies, one cannot but notice that there is still a paucity of research on p53 and bcl-2 expression in pituitary tissues. Thus, further exploration of this topic is warranted to confirm these preliminary findings.
In summary, the abnormal expression of p53 and bcl-2 in certain pituitary tumors could play a role in their carcinogenesis. The simultaneous genetic complementation of deregulated p53 and bcl-2 could be implicated through multistep tumorigenesis of pituitary tumours.