1. Background
Migraine is a common disabling and chronic neurological disorder, representing a substantial personal and social burden worldwide, with a prevalence estimated at 13%. The disease frequency is considerably higher in female patients at 18.2% and specifically affects the 25-55-year-old age group (1, 2). After adjusting for age, gender, and income, the incidence of psychiatric diseases (depression, anxiety, and bipolar disease), respiratory problems (allergies, asthma, and chronic obstructive pulmonary disease (COPD)), cardiovascular diseases and related risk factors (hypertension, impaired lipid profile, obesity, cerebrovascular accident, and chronic pain) showed to be more common in migraine patients (1). The prevalence of this condition increases between the ages of 12 - 40 years and reduces thereafter in both sexes. Severe headache attacks are reported to cause profound impairment in patient activities or require bed rest in 43% of the patients. One-third of migraine patients missed at least one day of work or school in the previous three months due to headaches, and 51% reported reduced productivity at work or school (3).
Various physiological and environmental factors have been shown to facilitate the onset of attacks such as hormonal changes, changes in routine normal life like sleep time, working pressure, and seasonal changes (2). Migraine attacks distribution changes with the hormonal changes during the menstrual cycle. A study showed that a reduction in sex hormone levels in females, especially during the end days of the luteal phase and menstruation, is associated with an increased attack rate in 55% - 70% of migraine patients. However, true menstrual migraine occurs in 7% - 14% of the patients, which is defined as experiencing migraine attacks just two days before to three days after the onset of menses (4, 5). When menstrually related migraines are considered, more than 90% of the migraine attacks occur between the two days before menses and the first few days of menses. Thus, the distinction between “menstrual” and “menstrually related” migraine is important. It also should be considered that the exacerbation of attacks during the hormonal change has not been reported in all patients. Therefore, the presence or absence of a definite relationship between the pain and menses may involve different biochemical mechanisms in different subtypes of the disease (6).
The presence of specific receptors for progesterone has been reported in the bovine pineal gland (7). In addition, it has been shown that the circadian secretion of melatonin is modulated via native progesterone levels (8) and increased by progesterone administration (9) in healthy women. Thus, it could be deduced that melatonin secretion is influenced by gonadal steroids. However, this relationship has not been consistently confirmed in reported studies. Murialdo et al. (10) reported a significant decrease in nocturnal urinary melatonin during the ovarian cycle in common-type migraine patients compared to healthy controls. They reported significantly lower melatonin urinary excretion in migrainous subjects than in the control group in all phases of the menstrual cycle. The melatonin levels in both groups were higher during the luteal phase than during the follicular and menstrual periods. However, there were no significant changes in the melatonin level when the data were analyzed between different phases of the ovarian cycle (10). Melatonin level further decreases in migraine attack. Brun et al. (11) also emphasized that decreased urinary melatonin was associated with menses in female patients. It has been shown that the overall melatonin blood level was lower in migraine patients than in healthy controls (10-12).
Melatonin, as a promising treatment for migraines, has been investigated in different case-control studies. An open-label trial using melatonin for migraine prophylaxis showed a significant reduction in attacks in 64.7% of the migraine patients (13). Despite the conflicting results, a systemic review of the current study suggests that melatonin could be a suitable and beneficial alternative for migraine prophylaxis with doses corresponding to 3 mg and a therapy duration of about three months or more (14).
2. Objectives
In this study, we aimed to investigate the effect of melatonin on menstrual-related migraine and compare melatonin treatment efficacy with naproxen, which is currently used as preventive therapy in this specific type of migraine headaches.
3. Methods
This study is an open-label, randomized clinical trial. The study protocol was designed and registered at the Iranian Registry of Clinical Trials (code: IRCT20121110011424N4) and conducted in the Neurology Clinic of Imam Khomeini Hospital affiliated to Tehran University of Medical Sciences.
Menstrual-related migraine criteria: The study enrolled women aged 18 years or older who were experiencing regular menstrual cycles ranging 28 - 35 days, with a BMI of 20 - 35. They were assessed for eligibility with a definite previous history of migraine at least six months before the current outpatient visit and the experience of menstrual-related migraine attacks (according to the International Classification of Headache Disorders [ICHD-III beta]).
The participants who were included in this study met the criteria for pure menstrual migraine or menstrually related migraine based on the criteria proposed by ICHD-III beta. They fulfilled the criteria for migraine without aura and prospectively recorded migraine attacks over at least three consecutive cycles exclusively on day 1 ± 2 (i.e., days -2 to +3) of menstruation in at least two out of three menstrual cycles. In addition, if they experienced a migraine attack at no other time of the cycle, they were defined as women with “menstrual migraine”. If additional attacks occurred at other times of the cycle, they were classified as women with menstrually related migraine. Both subgroups were included in this study and labeled as “menstrual-related” migraine patients.
3.1. Inclusion and Exclusion Criteria
As mentioned, the patients were asked to report migraine attacks without aura, with exacerbations at least in two out of three menstrual cycles before the screening visit to be enrolled in this study. The patients who had a history of other types of primary headaches, medication overuse headaches, peptic ulcers, diabetes mellitus, or hypertension were excluded from the study. Pregnant women were also excluded.
3.2. Randomization Method, Treatment Protocol, and Study Visits
Eligible patients were randomly divided into two treatment groups A and B, by using computerized random digits. Neither the patients nor the physicians were blinded to the patient’s groups. Research investigators who evaluated the patient headache attacks during the follow-up visits and statistical analysts were blinded to treatment groups. All patients were evaluated in two follow-up outpatient visits one month and three months after the screening visit. The two other clinical visits were planned at the end of the first and third subsequent menstrual periods. Data regarding age, sex, body mass index (BMI), and marital status were recorded for all cases. The patients were assessed by a migraine dairy for attacks and sleep problems experienced in the previous month, Visual Analog scale (VAS) for pain severity, and the Berlin sleep questionnaire for sleep apnoeaby an external blinded research investigator. Data analysis was also assessed by an external investigator who was blinded to the intervention in each group. Reported side effects were assessed only for the melatonin group. The number of sedatives or analgesic drugs that were used for at least one month during migraine attacks was also recorded in each treatment group.
All eligible women signed an informed consent form in the first clinical visit. Demographic data, including age, body height, weight, and marital status, were recorded in a questionnaire at the time of the screening visit.
3.3. Treatment Strategies
In the melatonin group, 3 mg of melatonin was given overnight (30 min before bedtime) from three days before to five days after menstruation. The other treatment group was prescribed with 250 mg naproxen BID for the same time course.
3.4. Statistical Analysis
The SPSS version 16.0 software (IBM, Armonk, NY, USA) was used for data analysis. The Independent sample t-test, ANOVA, repeated-measures ANOVA, and χ2 test were used as needed.
4. Results
Sixty eligible women were enrolled in this study and randomly divided into two treatment groups, melatonin, and naproxen, and followed for three months. For the final analysis, four patients were excluded in the melatonin group due to their refusal to continue treatment or missing follow-up appointments.
4.1. Demographic Data
The mean age was 33.33 ± 6.34 in the naproxen group and 36.92 ± 6.81 in the melatonin group. The mean height and weight were 163.28 ± 5.38 vs. 164.84 ± 4.4 cm, and 65.80 ± 8.51 vs. 67.08 ± 9.07 kg, respectively. The independent sample t-test showed no significant differences in the measured demographic data between the two treatment groups (P value > 0.05) (Table 1).
| Variable | Naproxen Treatment Group | Melatonin Treatment Group |
|---|---|---|
| Patient number | 30 | 26 |
| Age | 33.33 ± 6.34 | 36.92 ± 6.81 |
| Weight | 65.80 ± 8.51 | 67.08 ± 9.07 |
| Height | 163.28± 5.38 | 164.84 ± 4.4 |
| BMI | 24.47 ± 6.31 | 24.94 ± 5.77 |
aValues are expressed as mean ± SD.
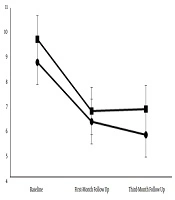
Figure 1 shows the number of days the patients experienced migraine attacks. A repeated-measures ANOVA with a Greenhouse-Geisser correction showed that the mean number of attack days was not significantly different between the two treatment groups (F = 0.46, df = 1.354, P > 0.05). The post hoc tests using the Bonferroni correction indicated that attack days in each group reduced significantly in both follow-up visits compared to the screening day (P < 0.001). However, no significant difference was observed in the one-month follow-up and three-month follow-up visits (P > 0.05).
The mean number of migraine attack days in the melatonin and naproxen treatment groups. The mean number of attack days was not significantly different between the two treatment groups (P > 0.05). In both treatment groups, the patients experienced a significantly reduced number of attack days in the follow-up visits compared to the screening day (P > 0.05).
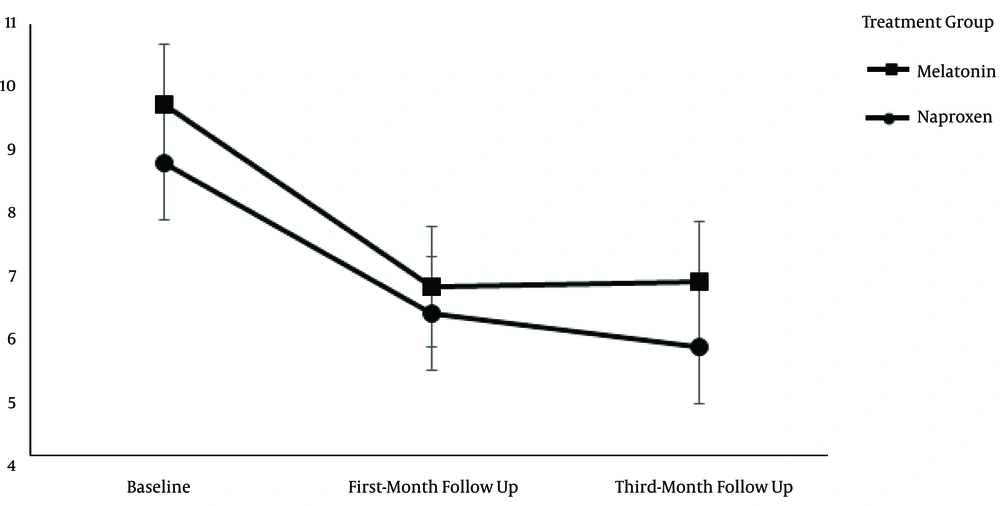
Figure 2 elicits the migraine pain severity based on the VAS in the two treatment groups. The repeated-measures ANOVA test using Greenhouse-Geisser correction showed no statistical difference in the subgroup analysis between the two treatment groups (F = 0.186, df = 1.171, P > 0.05) whereas the post hoc tests using Bonferroni correction depicted that the reduction of pain severity in both treatment groups was significant in the first month and third months after drug prescription (P < 0.001 and P < 0.001, respectively). Furthermore, no statistically significant difference was observed between the two follow-up visits (P > 0.05).
The mean score of pain severity during migraine attacks scored based on VAS in the melatonin and naproxen treatment groups. There is no statistical difference between the treatment groups (P > 0.05), whereas pain severity significantly reduced in both treatment groups in the one-month and three-month follow-ups (P > 0.05).
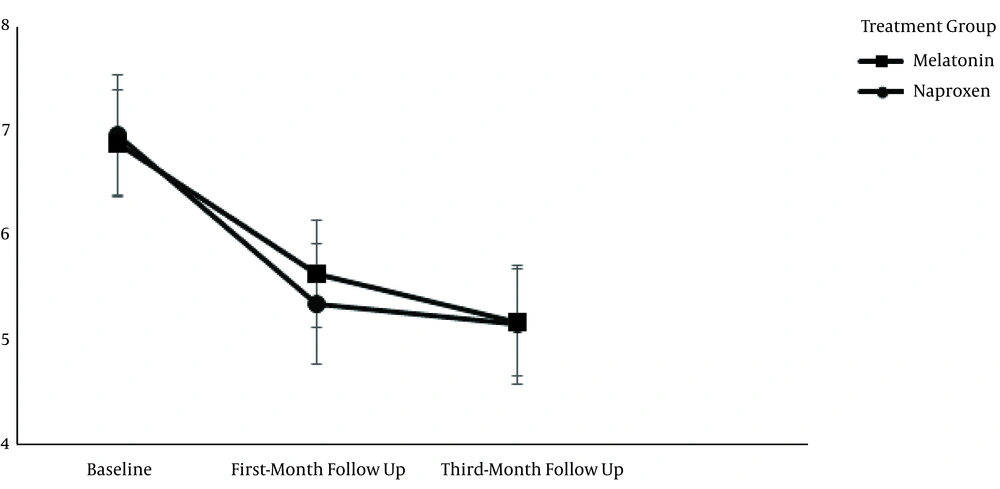
Figure 3 depicts the number of sedatives and/or analgesic drugs used during each migraine attack.
As shown, sedative/abortive drug use was significantly lower in the melatonin group than in the naproxen group (F = 11.33, df = 1.009, P < 0.05). The ANOVA using Bonferroni correction post hoc tests showed a significant reduction in the one- and three-month visits (P < 0.001 and P < 0.001, respectively). However, the sedative use was not significantly different between the two follow-up visits (P > 0.05).
Table 2 shows the frequency, and the name of sedative/analgesic drugs used during migraine attacks in patients enrolled in this study.
| Abortive/Sedative Drug Name | Frequency of Use in the Total Number of Abortive/Sedative Drug Used During Reported Attacks, % |
|---|---|
| ACA | 46.8 |
| Ibuprofen | 23.95 |
| Diclofenac | 5.2 |
| Dexamethasone | 4.2 |
| Sumatriptan | 2.1 |
| Acetaminophen codeine | 7.3 |
| Naproxen | 3.12 |
| Migrextra (compound sedative drug) | 3.12 |
| Indomethacin | 4.2 |
4.2. Sleep Problems
The complaints of snoring based on self-reports (26.7% in the naproxen group vs. 38.5% in the melatonin group) and family-reports (26.7% in the naproxen group vs. 30.8% in the melatonin group) were not significantly different by using the χ2 test based on Pearson correlation test (χ2 = 0.888, df =1, P > 0.05 and χ2 = 0.115, df = 1, P > 0.05, respectively).
A repeated-measures ANOVA using Greenhouse-Geisser correction depicted significant changes in the snoring rate and post-sleep tiredness (F = 4.821, df = 1.219, P < 0.05, and F = 5.424, df = 1.051, P < 0.05, respectively). The snoring rate reduction was significant using the ANOVA test based on Bonferroni correction post hoc tests in the one-month and three-month visits compared to the screening day (P < 0.05 and P < 0.05, respectively). However, no significant change was observed between the two follow-up visit days (P > 0.05). Post-sleep tiredness was significantly different based on the ANOVA test between the follow-up visits and the screening day (P < 0.05 for one-month follow-up and P < 0.05 for three-month follow-up); it was also significantly lower in the three-month follow-up than in the one-month follow-up clinical visit (P < 0.05).
Side Effects in the Melatonin Treatment Group: The most reported side effect was dizziness (53.8%). Other side effects in order of frequency were inattention (38.4%), daily fatigue (38.4%), and GI problems (15.38%).
5. Discussion
In this study, we compared the efficacy and safety of melatonin and naproxen in patients with menstrual-related migraines. The number of migraine attack days and the severity of migraine pain in the two groups improved significantly, and the decreasing trend overtime was the same in the two groups. This finding shows that melatonin is as effective as naproxen in reducing the number of migraine-related menstrual attacks. Treatment with naproxen or melatonin made a significant difference in the number of sedative/abortive drugs used in each attack, which was lower in the melatonin group. Melatonin also had a beneficial effect on reducing sleep problems such as the rate of snoring and the severity of subjective tiredness after sleep. The most reported adverse drug reactions in melatonin-treated patients were dizziness (54%), post-sleep fatigue (38.5%), inattention (38.5%), and GI problems (15%).
Similar to the results of the current study, the first pilot study of melatonin in 2004 for migraine cases showed a significant decrease in headache attacks in 64.7% of the patients. In the first month, a complete response to treatment was achieved in 25% of the patients. They reported a significant reduction in the attack rate and duration, pain severity, and the number of sedative drug use in patients. The reported side effects in the study included daytime sleepiness and alopecia (13).
Another randomized clinical trial, designed to investigate the effect of melatonin as a prophylactic treatment on migraine and its role in sleep quality of patients, demonstrated the superiority of melatonin to placebo in reducing the frequency of migraine attacks. However, this study did not show a significant effect on sleep quality (15). It is important to note a few points about different treatment responses reported by the aforementioned study compared to the current reported results. First, the dose of melatonin used in this study was 2 mg versus 3 mg in our study. Second, the baseline number of patients’ migraine attack days per month was lower in this study (4.2) than in ours (7.5). Third, in this study, the control group received a placebo while our study patients received naproxen, a known drug for the prophylactic treatment of menstrual-related migraine (15). In another similar study, a pilot trial was performed on 49 different primary type headache patients (37 with migraine headaches and 12 with tension headaches), which showed that treatment with melatonin (4 mg half an hour before sleep) for six months significantly reduced the attack frequency in patients compared to baseline (16). A recent systematic review study focused on melatonin as a prophylactic treatment for migraine (14). A total of seven studies were eligible although due to heterogeneity and the low number of patients, a meta-analysis was not performed. The authors claimed that because of contradictory results of the studies, the beneficial role of the melatonin treatment in migraine attacks could not be proven, but the treatment regimen with melatonin at a dose of 3 mg for intervals of three months appeared to show promising results (14).
There are limited data about the role of melatonin in migraine and menstrual-related headaches. Melatonin has anti-inflammatory properties and leads to a reduction in prostaglandin E production (activator of the trigeminovascular system) and inflammatory cytokines (17, 18). In addition, melatonin is a neurohormone that can control the vascular response of the brain and lead to vasoconstriction of the brain arteries (19). Melatonin reduction, therefore, can play a role in triggering migraine headaches. In this regard, trigeminal ganglion contributes to releasing neuropeptides such as substance P (SP) and calcitonin gene-related peptide (CGRP), which are involved in provoking migraine attacks (20, 21). A study by Ansari et al. revealed that melatonin decreases the expression of CGRP through its effect on the production of nitric oxide via inducible nitric oxide synthase (iNOS) enzyme activity in migraine patients (22). Furthermore, it has been shown that non-steroidal anti-inflammatory drugs (NSAIDs) also decrease melatonin synthesis in the body, which may lead to possible harmful effects of chronic NSAIDS use, such as naproxen, in migraine patients. The melatonin level significantly increases during the luteal phase of the menstrual cycle, possibly due to the increase of post-ovulatory progesterone. In addition, a study showed that progesterone receptors are present in the bovine pineal gland, which may support the melatonin-progesterone interaction in regulating the menstrual cycles (8, 9, 11, 23, 24). Physiological changes in the menstrual cycle have not been found in migrainous women, which may influence the attack severity during the menstrual phase (11). The fluctuation of the pain threshold and a dose-related analgesic effect for melatonin are shown in experimental animals, such as mice, and it may reflect the role of the pineal gland in the determination of the light-dark-linked circadian rhythm in the body (25). Therefore, the administration of melatonin in menstrual-related migraines may be effective in relieving attacks by interfering with hormonal and circadian rhythm changes.
In this study, sleep quality factors like snoring and post-sleep tiredness improved after melatonin treatment compared to pre-treatment. Therefore, there is a potential role for melatonin in improving the sleep quality of patients with menstrual-related migraines. Melatonin also showed beneficial effects on sleep quality in two other studies by Peres et al. (13, 25). They also previously reported the significant effect of sleep disturbances on migraine chronobiologic features in chronic or episodic types and showed that 46.5% of the patients experience headaches after changing their sleep schedule. A significant shift in the duration of sleep was observed in patients, ranging from -2.5 to +5 h. Most patients (69%) had a delayed sleep phase, whereas 31% slept earlier (18). Both episodic and chronic migraine cases reported waking up in the morning or being woken up during the night by headaches, and a significant number of migraine attacks occurring in the morning period were attributed to sleep disorders (26, 27). Although a few studies have looked into the effects of melatonin treatment on the sleep quality of migraine patients, their results generally suggest the lack of effectiveness in patients without sleep disorders (15).
Our results showed that the number of sedative drug use was significantly lower in the melatonin group than in the naproxen group. Although naproxen is the treatment of choice for menstrually related migraine, it has been shown that non-steroidal anti-inflammatory drugs (NSAIDs), which reduce pain via prostaglandin synthesis inhibition, may also decrease melatonin synthesis (28).
Therefore, we conclude that melatonin could be considered a potential treatment for menstrual-related migraines, with an effectiveness comparable to that of naproxen in reducing attacks. In addition, our study showed lower rates of medication overuse headaches and sleep problems in the melatonin treatment group than in the naproxen group, which are two challenging problems for physicians in managing chronic migraine patients. It is also an alternate treatment for patients when there is a contraindication for using NSAIDs. However, the number of cases and the lack of a placebo arm are the two limitations of the trial, which should be considered in future investigations of the role of melatonin as a potential treatment in women with menstrually related migraine headaches.