1. Context
Several pneumonia cases with no previously identified etiology were described in Wuhan, China, in December 2020, which rapidly spread worldwide. Genome sequencing analyses confirmed that this was a novel coronavirus (2019-nCoV). The World Health Organization (WHO) named the newly recognized coronavirus as Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Despite global efforts to control SARS-CoV-2, the spread of the virus has reached a pandemic. The disease it causes has been announced as Coronavirus Disease 2019 (COVID-19) and is an emerging infectious disease estimated to have a significant consequence worldwide. In this review, the most recent evidence about COVID-19 in children is discussed to help in the management of pediatric cases (1).
2. Evidence Acquisition
The most recent evidence about the clinical features and potential reasons for the non-susceptibility of children to SARS-CoV-2 infection is provided in the present narrative review. In the primary stage, a systematic search was performed in some databases/search engines including ISI Web of Science, Scopus, PubMed, and Google Scholar. In the later stage, all selected documents were fully reviewed. The keywords utilized for finding related articles were severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), pediatric, COVID-19, treatment, angiotensin-converting enzyme 2 (ACE2), clinical feature, coronavirus, and pneumonia.
3. Results
3.1. Epidemiology of COVID-19 in Children
Once the COVID-19 outbreak originated in Wuhan, China, the new coronavirus named SARS-CoV-2 was detected in adults but not in children (World Health Organization. Report of the WHO-China joint mission on COVID-19, 16 - 24 February 2020. Geneva (Switzerland): World Health Organization; 2020). Subsequently, the pediatric infection rate was reported to increase from 2% to 13% rapidly. The potential explanation could be the lower exposure of children to SARS-CoV-2 in the early phase of the outbreak. Otherwise, due to mild symptoms in children, they were less likely to be tested in the early course of the outbreak (2).
There are limited data about the burden of COVID-19 in children. The currently available data show that COVID-19 infection is lower in children than in adults. A 10-year-old boy with recent travel history to Wuhan, China, was the first reported pediatric case (3). In addition, a retrospective study on 366 children (≤ 16 years of age), hospitalized with respiratory infections during the early stages of the outbreak, confirmed COVID-19 infection in 1.6% of cases. These results indicate the occurrence of COVID-19 infection in children since the early stage of the epidemic outbreak (4).
3.2. Clinical Features in Children
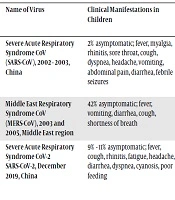
According to the currently reported epidemiological data, the incubation period of COVID-19 infection varies between 1 and 14 days, commonly ranging from three to seven days. The available information about children shows that the disease onset frequently occurs at the age of 1.5 months to 17 years. The majority of cases had a history of close contact with infected individuals or were family cluster cases (5) (Table 1). However, there are several reports about COVID-19 disease in neonates (6-8). Based on available data, the confirmed cases of COVID-19 infection in children tend to be infrequent and the symptoms are mostly mild with favorable prognosis. The fatality rate has been reported by 0% in children under 10 years of age (5). Several recent studies have suggested that children with COVID-19 frequently present with fever, cough, and fatigue accompanied by nasal stuffiness, rhinorrhea, sputum, diarrhea, and headache. Patients may also be afebrile or have a mild fever. Dyspnea can occur as a result of disease progression, which may be followed by systemic symptoms, including malaise and poor appetite. In the case of pneumonia, the disease progresses and can lead to respiratory failure. Furthermore, metabolic acidosis, septic shock, and irreversible bleeding, as well as coagulation dysfunction, have been reported in severe cases (9). A recent study reported fever, cough, sore throat, stuffy nose, and rhinorrhea as the most frequent symptoms in children with confirmed COVID-19 infection. The median patient age was also reported as 74 months (range, 3 - 131 months) (10). Another study described that among seven symptomatic infants, four had a fever, two had mild respiratory symptoms, and one was asymptomatic.
| Name of Virus | Clinical Manifestations in Children | Laboratory Findings in Children | Shedding Duration | Chest CT Findings in Children | Transmission in Children | Diagnosis in Children | Mortality Rate in Children, % |
|---|---|---|---|---|---|---|---|
| Severe Acute Respiratory Syndrome CoV (SARS-CoV), 2002 - 2003, China | 2% asymptomatic; fever, myalgia, rhinitis, sore throat, cough, dyspnea, headache, vomiting, abdominal pain, diarrhea, febrile seizures | Reduced neutrophil count; reduced lymphocyte count; thrombocytopenia; elevated alanine aminotransferase; elevated lactate dehydrogenase; deranged coagulation; elevated d-dimers in severe cases | Frequently after the onset of symptoms | GGO; consolidation | Household contact; nosocomial contact | RT-PCR or RNA sequencing on nasal and pharyngeal swabs, endotracheal aspirate or BAL, sputum | 0 |
| Middle East Respiratory Syndrome CoV (MERS-CoV), 2003 and 2005, Middle East region | 42% asymptomatic; fever, vomiting, diarrhea, cough, shortness of breath | Normal WBC, liver function tests, urea, and creatinine levels; thrombocytopenia | Frequently after the onset of symptoms | Bilateral airspace; consolidations | Household contact; nosocomial infection | RT-PCR or RNA sequencing on nasal and pharyngeal swabs, endotracheal aspirate or BAL, sputum | 6 |
| Severe Acute Respiratory Syndrome CoV-2 SARS-CoV-2, December 2019, China | 9% - 11% asymptomatic; fever, cough, rhinitis, fatigue, headache, diarrhea, dyspnea, cyanosis, poor feeding | Normal or reduced WBC; reduced neutrophil count; reduced lymphocyte count; normal CRP and PCT levels; irregular liver function tests; elevated lactate; dehydrogenase; elevated d-dimers in severe cases | Unknown | Bilateral multiple patchy nodular GGO; infiltrating shadows in the middle and outer regions of the lung | Household contact | RT-PCR or RNA sequencing on nasal and pharyngeal swabs, endotracheal aspirate or BAL; sputum; serology only when RT-PCR is not available | 0 |
Comparison of the Characteristics of Three Novel Coronaviruses Causing Epidemics in Recent Years in Children (11)
Moreover, no severe complications were noted (12). The first severe pediatric case described was a 13-month-old boy who was admitted with pneumonia, shock, Acute Respiratory Distress (ARDS), fever, dyspnea, oliguria, and renal failure. He had no known medical comorbidities. The patient received intensive care, including intubation and mechanical ventilation, and had been treated at a clinic for the past six days for intermittent diarrhea and vomiting but had no respiratory symptoms (13). Collectively, the large scale analysis of pediatric cases with confirmed COVID-19 reported that the most frequent symptoms were cough and fever, followed by other clinical symptoms such as myalgia, rhinorrhea, nasal congestion, nausea, headache, vomiting, dizziness, abdominal pain, and diarrhea, which commonly disappeared within a week (5).
3.3. Laboratory Findings
Complete blood counts were reported normal in the majority of cases. However, in the primary stage of the disease, normal or decreased white blood cell (WBC) count with reduced lymphocyte count is the most common laboratory finding (Table 1). In some cases, the levels of myohemoglobin, liver enzymes, and muscle enzymes are elevated. Increased levels of C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) are seen as common findings. Procalcitonin levels are normal. Increased levels of D-dimer and gradually decreased lymphocyte counts are the findings in severe cases. The real-time reverse transcriptase polymerase chain reaction (RT-PCR) assay of nasopharyngeal swab specimens, stool, and blood is also performed for the detection of viral RNA (14). In a recent study, only two cases showed reduced leukocyte counts, and one showed a slight reduction in the lymphocyte count was normal or temporarily elevated. The results of the classification of cases based on symptoms and chest radiology findings were as follows: 6.7% were asymptomatic with normal chest radiology results, 64.9% had mild symptoms with normal chest radiology results, 26.9% had pneumonia, and 1.5% experienced severe disease and were mechanically ventilated. Both of the cases with critical disease progression had underlying problems (5).
3.4. CT Findings
The reported chest computed tomography (CT) imaging features in children are similar to those in adults. Both suspected and confirmed individuals should carry out a chest X-ray analysis. A chest CT examination is performed when essential. The most frequent chest CT findings in children with COVID-19 are unilateral or bilateral subpleural ground‐glass opacities (GGO) and consolidations. Pulmonary consolidation occurs in severe disease, and pleural effusion is infrequent (15) (Table 1).
3.5. Differential Diagnosis
The other possibilities of diagnosis should be considered to differentiate SARS-CoV-2 infection from other recognized viral infections such as influenza and parainfluenza viruses, Respiratory Syncytial Virus (RSV), adenovirus, SARS coronavirus-1, and rhinovirus, as well as bacterial pneumonia. The coinfection of SARS-CoV-2 with other viruses and/or bacteria should also be evaluated (14).
3.6. Potential Reasons for Milder Presentation of Disease in Children than in Adults
The low prevalence of COVID-19 among children and mild symptoms of the disease in confirmed pediatric individuals have been reported from various countries, suggesting a lower risk of severe disease in children with no underlying conditions than in other age groups. The explanations for mild presentations of SARS-CoV-2 infection in pediatric patients remain unknown. However, several postulates exist (16).
3.6.1. Immunological Features
The difference in the composition and functional features of the immune systems of children and adults could be considered a possible explanation (17). It has been suggested that children have more functional and active innate immune responses and healthier respiratory tracts, as well as fewer underlying conditions (18). Moreover, maternal antibodies are important to protect infants within the initial months of life. However, these antibodies do not protect them against new viruses such as SARS-CoV-2 (19).
Age-related changes in T lymphocytes have been associated with the development and atrophy of the thymus. It has been described that SARS-CoV-2 is associated with a decrease in CD4 cells in older men. The CD4 counts decline rapidly in early childhood and then progressively decreases during the lifetime. The CD8 counts are maintained or show a small increase in elderly adults (20). Higher CD4 cell counts and lower CD8 T lymphocytes in children than in adults have been reported in different studies (21). Therefore, SARS-CoV-2 might predominantly affect lymphocytes, particularly T lymphocytes, as does SARS-CoV (22).
In addition, the recognition of pathogen-associated molecular patterns (PAMPs) is mediated by innate immune cells via various pattern recognition receptors (PRRs including toll-like receptors (TLRs) and other cytosolic pathogen sensors. PRRs stimulate downstream signaling cascade leading to the generation of interferons (IFNs) type I and type III, as well as other proinflammatory mediators. These result in the initiation of both innate and adaptive immune responses. Subsequently, type I IFNs stimulate the kinase (JAK)-signal transducer and activator of transcription (STAT) pathway, which has a significant role in the regulation of immune response. Moreover, IFNs can directly stimulate immunity by the stimulation of dendritic cells (DCs) and enhance the activity of both cytotoxic T cells and natural killer (NK) cells. The NK cells can move to the sites of infection and respond to viral pathogens through the generation of IFN-γ, killing the cells infected with the virus and increasing the adaptive immune response (23, 24). Both IFNs and cytokines are responsible for pulmonary damage during acute viral infection. High levels of innate pro-inflammatory cytokines and type I IFNs have been reported in severe cases of COVID-19 infection. Numerous studies have also reported enhanced neutrophil counts and decreased lymphocyte counts in COVID-19 cases with the initiation of the so-termed “cytokine storm”, suggesting the significance of innate immune response as a damaging and protective mechanism (25).
Furthermore, milder disease presentations in children seem to be associated with innate immune memory or “trained immunity”, which is formed by memory cells after antigen exposure (26). Lung innate lymphoid cells group 2 (ILC2) showed to remember their stimulation status by inhaled allergens (27). In the case of reinfection with cytomegalovirus (CMV) or influenza A, a more powerful secondary innate immune response mediated by NK cells is activated (28). Trained immunity memory provides cross-protection against different pathogenic agents and may also be stimulated by vaccines (26). There are increasing data on the decreased mortality rates in measles-vaccinated children and the reason is beyond the inhibition of deaths caused by measles (23).
In summary, trained immunity and a higher number of lymphocytes in children may describe the mild appearance of the disease in pediatric cases.
3.6.2. Virus-Virus Competition
The other possibility for milder disease in children could be the co-existence of other viral pathogens in the mucosal surfaces of the lungs and upper airways, which may contribute to the limitation of SARS-CoV-2 growth by direct virus-to-virus competition. This is in line with recent information about the current pandemic, which showed an association between the viral load (copies/mL) and the severity of SARS-CoV-2 infection (29).
3.6.3. Angiotensin-Converting Enzyme 2 (ACE2) Receptor
The SARS-CoV-2 virus employs ACE2 receptors for entrance into host cells and subsequent replication (30). A difference in the function and distribution of viral receptors can be the potential reason for age-related differences in infection occurrence. Former studies established that HCoV-NL63 infection is more frequent in adults than in children (31, 32). Therefore, differences in the ACE2 receptor expression could be the other explanation for the mild presentation of COVID-19 disease in children (33).
On the one hand, the correlation of ACE2 expression with the susceptibility to SARS-CoV-2 infection has been demonstrated previously (34, 35). Angiotensin-converting enzyme 2 is recognized as an important negative regulatory factor for the severity of pulmonary edema and lung failure. A considerably reduced pathologic modification in the lungs of ACE2 mutant mice was shown in a previous study. This study suggested that SARS-CoV infection leads to ACE2 downregulation through binding of the SARS-CoV Spike protein to the host cell ACE2 receptor. It was also shown that systemic treatment with recombinant ACE2 decreased lung damage (36).
On the other hand, ACE2 has been suggested to be involved in protective mechanisms of the lung. It can protect the lung against severe damage caused by viral infections (36, 37). It has been indicated that the circulating ACE2 enzyme protects against lung injury induced by influenza A (H7N9) (38). Furthermore, a former study showed that patients with higher levels of serum ACE2 indicated better outcomes, and the downregulation of the ACE2 gene in mice infected with H5N1 led to severe lung injury (38). Therefore, treatment with recombinant human ACE2 showed beneficial effects (39).
Generally, children have higher levels of ACE2 than adults (40). A study on rat lung revealed that the expression of ACE2 decreases with age (41). Moreover, a study on the transgenic mice model showed that AEC2 depletion was associated with both pulmonary fibrosis and increased mortality rate (42). Therefore, the explanation for the association between age and severity of COVID-19 might be related to the ACE2 plasma profile (43). These results may suggest that children are susceptible to SARS-CoV-2 infection but serum ACE2 may help them to present mild disease.
3.6.4. Bacille Calmette-Guérin Vaccine
Some epidemiological investigations have proposed a link between BCG vaccination and COVID-19 morbidity and mortality (44). The BCG vaccine has some off-target effects on the immune system, which leads to reduced viremia (45, 46). There are two ongoing randomized controlled clinical trials (NCT04327206, NCT04328441) in Australia and the Netherlands to evaluate whether BCG decreases the occurrence and severity of COVID-19 infection in health-care workers (47). According to WHO, as of now, there is no evidence that BCG vaccination protects individuals against SARS-CoV-2 infection (48). There would be a significant approach in response to future pandemics if the BCG vaccine is responsible for nonspecific protection against COVID-19 infection.
3.7. SARS-CoV-2 Transmission in Children
The transmission of SARS-CoV-2 in children predominantly occurs through direct contact with infected adults. On the contrary, direct transmission from a child to an adult has not been fully approved so far (5). A recent case series study emphasized the transmission of infection through familial exposure. The authors confirmed the transmission of SARS-CoV-2 infection from one child to his parents who developed symptomatic infection seven days after looking after the infant (10). Furthermore, an analysis of public disclosure data reported a link between three patients with the age range of 15 to 18 and three secondary cases (49). However, these studies are not reliable enough due to the lack of fully qualified designs and small sample sizes (50).
A non-peer-reviewed mathematical modeling study report described the high transmissibility of SARS-CoV-2 infection among adults ≥ 25 years of age and low transmissibility of this infection among children and individuals ≤ 14 years (51). The WHO-China Joint Mission expressed that children infected with SARS-CoV-2 are frequently recognized by contact tracing (50). Moreover, the emerging evidences has shown the potential transmission of COVID-19 from infected children to adults. In addition, an analysis of open access databases described that 35% of children and adults across 14 countries had an infected family member (52). However, they did not explain whether the infection was transmitted from children to adults. Collectively, the exact role of children in the transmission or spread of COVID-19 remains elusive yet (50).
There are limited data about the duration of viral shedding in children (Table 1). However, the prolonged detection of viral RNA in feces and nasopharyngeal/throat swabs of pediatric cases suggests the potential role of children as transmitters of the infection in the community (5). A case study reported an asymptomatic six-month-old infant whose nasopharyngeal sample was positive by PCR for SARS-CoV-2. His mother had previously confirmed COVID-19 pneumonia. Once the patient’s temperature reached 38.5°C, the PCR sample turned positive for the presence of SARS-CoV-2. In addition, a negative result was achieved on day 17 of hospitalization (53). In another study, test results for SARS-CoV-2 PCR on nasopharyngeal or throat swabs turned positive 4 to 48 hours after symptom onset, and negative conversion was observed at a median 12 days (6 - 22 days).
Furthermore, some patients had prolonged shedding of viral RNA in feces for 18 - 30 days (10). The current evidence shows that nucleic acid testing for SARS-CoV-2 in stools remain positive up to 8 - 20 days after the complete clearance of the viral RNA in respiratory specimens. Therefore, it has been suggested that fecal shedding of SARS-CoV-2 should not be ignored as one of the potential routes of community transmission (54). Children have a potential role in transmitting viruses, as they harbor large numbers of viruses without presentation of symptoms. Moreover, fecal samples in children remain positive for SARS–CoV-2 on RT-qPCR long after negative test results are obtained for nasopharyngeal secretions. Therefore, while children tend to present mild symptoms of infection, their role in community transmission should not be ignored.
3.7.1. Mother to Child Transmission (MTCT) or Vertical Transmission of SARS-CoV-2
According to the WHO-China Joint Mission on COVID-19, pregnant women are not at a higher risk for severe infection with SARS-CoV-2. Currently, the significance of COVID-19 during pregnancy is unidentified and it is supposed that pregnant women have already become infected. All kinds of pneumonia (viral or bacterial) can be the cause of morbidity and mortality among pregnant women and they have been considered the third most common cause of indirect maternal death (55). Pneumonia in pregnancy may cause adverse obstetrical consequences such as premature rupture of membranes, intrauterine growth restriction (IUGR), preterm labor, intrauterine fetal demise, and neonatal death (55).
Based on the previous experience with pregnant women who were infected with Middle East Respiratory Syndrome CoV (MERS-CoV) and SARS-CoV, the intrauterine maternal-fetal transmission has never been confirmed. In addition, viral infection through the birth canal during vaginal delivery or through post-partum breastfeeding would be extremely rare for a respiratory virus. A recent retrospective study on pregnant women with confirmed COVID-19 analyzed the possibility of MTCT of the virus. It was reported that none of the COVID-19 cases revealed severe pneumonia, needing ventilation, or expired of pneumonia. The most-reported complications after COVID-19 infection were fetal distress and premature rupture of membranes. Their findings suggested no evidence of MTCT in women with confirmed COVID-19 pneumonia in the third trimester of pregnancy (56). As a result, other transmission routes such as the inhalation of the pathogenic agent through aerosols produced by numerous sources in the hospital environment such as the coughing of the mother, relatives, or healthcare workers should also be considered. Additional clinical research is necessary to identify the potential risks and profits of novel treatments and vaccines in pregnancy (57).
3.8. Potential Treatment Options for COVID-19 in Children
The SARS‐CoV and MERS-CoV epidemics have provided comprehensive insights into how to overcome the SARS-CoV-2 pandemic (3) (Table 1). At present, there are no exact medications approved by the U.S. Food and Drug Administration (FDA) for COVID-19 treatment. The main currently available treatment strategy is symptomatic treatments, while effective antiviral therapy and vaccination are under evaluation (13, 17). The cases with hyperthermia should be dynamically controlled. Some antiviral agents such as lopinavir/ritonavir have been used for the treatment of adult cases, but their efficiency and safety in children remain to be investigated. Interferon-α decreases the load of the virus in the primary stage of infection and may be effective in alleviating symptoms. Intravenous immunoglobulin and systemic steroids can be administered in severe cases, but determining their effectiveness and adverse effects requires further assessment. Once hypoxia occurs, oxygen therapy should be undertaken. Nasal high-flow oxygen therapy or non-invasive therapy should be administered when necessary. Invasive ventilation may be used for severe cases with refractory hypoxemia (13, 17).
According to the Centers for Disease Control and Prevention (CDC), the decision to manage a pediatric COVID-19 case with mild to moderate infection in either outpatient or inpatient settings should be on a case-by-case basis. Moreover, the patient’s clinical presentation and underlying conditions should be considered. Numerous organizations have published guidelines related to the treatment of COVID-19 cases, including pediatric patients (58).
A treatment option recommended by the Zhejiang University School of Medicine is the combination of nebulized interferon-alpha-2b and oral lopinavir/ritonavir; in the case of complications such as encephalitis, hemophagocytic syndrome, ARDS, or septic shock, corticosteroids are also used. In addition, intravenous immunoglobulin has been recommended for severe cases (9, 11). A recent case series reported that 59% of COVID-19 cases were treated with lopinavir/ritonavir without receiving glucocorticoids or immunoglobulins (59). Moreover, monoclonal antibodies obtained from either convalescent human plasma or animal plasma against the glycoprotein S of SARS-CoV-2 have shown to block the entry of the virus into the target cells (60-66). Virus entry can also be prevented by inhibitors of related viral proteases (67-69). For instance, nelfinavir is an antiretroviral drug that selectively blocks the HIV protease and its inhibitory effect has been confirmed for SARS-CoV, suggesting the potential therapeutic effect for COVID-19 (70). Lopinavir/ritonavir combination is approved for HIV management in pediatric cases; however, there is no information about its effectiveness for the treatment of COVID-19 in children (71). Chloroquine/hydroxychloroquine is commonly used for the prevention and treatment of malaria and some autoimmune diseases such as lupus erythematosus. Known as a broad-spectrum antiviral medication, it inhibits virus-cell fusion and therefore blocks the virus entry (72).
However, the Canadian Pediatric Society recommended that chloroquine or hydroxychloroquine should not be used for suspected or confirmed COVID-19 children, except under the guidance of specialists, until a trial is approved by research ethics boards and Health Canada. However, children and adolescents who are receiving this drug for the treatment of chronic conditions such as lupus should continue the treatment under the direction of their physicians (73, 74). Moreover, a recent study suggested age-adjusted chloroquine dosing in pediatric COVID-19 cases to avoid suboptimal or toxic doses. In the mentioned study, the following total cumulative doses were proposed: 35, 47, 55, and 44 mg/kg (chloroquine base) for children aged 0 - 1 month, 1 - 6 months, 6 months - 12 years, and adolescents/adults, respectively. The given dose should not exceed 3,300 mg in any patient (75). Collectively, neither the WHO nor the US CDC recommends any specific treatment for adults or pediatric COVID-19 cases (97,163).
4. Conclusions
Coronavirus Disease 2019 is an emerging infectious disease, and its spread has recently reached a pandemic. There are limited data available on its prevalence and clinical features in pediatric patients. According to the currently available information, children comprise a small percentage of all COVID-19 cases globally and generally experience milder symptoms than adults. However, progression to severe disease has been reported. Several explanations have been suggested to explain the milder symptoms in children than in adults, including differences in immunity systems of children and adults and differences in ACE2 expression.
Moreover, newborns are susceptible to COVID-19 and viral RNA in their fecal samples remains detectable for a long time. Therefore, neonates could be a source of community transmission. The only treatment currently available for patients with COVID-19 mainly relies on symptomatic treatments, and effective antiviral therapy and vaccination are under evaluation. Additional investigation is required to evaluate the clinical features of the disease in children and their role in community transmission. This will provide important data about the protective mechanisms and targets for future treatments. Consequently, while children tend to present mild symptoms of infection, and due to the unclear role of them in the spread of the disease (as discussed before), their role in the pandemic should not be ignored. Therefore, certain measures, such as school closure can be considered an effective intervention to inhibit the spread of SARS-CoV-2.