1. Background
In developed countries, the majority of the children that are admitted to hospitals and all of those in the PICU or oncology wards have some kind of central venous line (CVL). The most common CVLs are peripherally inserted central catheters (PICC), which usually are put through the cephalic vein, and the tip of the catheter rests in the distal superior vena cava or right atrium. When the catheter is needed for a longer time, a tunneled catheter (like “Hickmans” or “Broviacs”) is used. The decision between Broviac rather than a port is usually based on the duration and frequency of use. A child that needs everyday venous access should have a line that exits the skin (like Broviac). Ports are more convenient for intermittent, short uses.
A port access catheter or, more correctly, "totally implantable venous access device” consists of three parts: The reservoir (portal) that has a silicone bubble for needle insertion, the catheter, and the catheter connector (which connects the catheter to the portal). The main benefit of CVL over the peripheral lines is the patient’s convenience, but its cost is more infection.
Infections of port catheters are divided into two clinical types. The first but less common form is Pocket infection with erythema, tenderness, induration, and drainage, over the pocket and subcutaneous tunnel with or without concomitant bloodstream infection. The second type of port infection is catheter-related blood stream infection (CRBSI), which often presents just with fever and no sign of pocket infection (1). The term CRBSI is reserved for bloodstream infections (BSI) that are demonstrated by the culture of CVC tip or other techniques caused by colonization of the device. On the other hand, the more general term central line–associated bloodstream infection (CLABSI) is often used for surveillance and refers to any BSI in patients with CVC, unless an alternative source is identified (2). The PAC infections are almost always due to high antibiotic-resistant pathogens (3).
In children’s hospitals of Mashhad, the frequency of CVL insertion for acutely ill children has largely increased during the past decade, and nowadays most PICU patients have CVL (mostly jugular or femoral), but unfortunately still most of the children of the oncology ward lack CVL.
2. Objectives
This research aimed to find the infection rate among children with PAC at Sheikh children’s hospital.
3. Methods
This is a retrospective descriptive study from 2006 - 2011, which was started by referring to the registry of the operation room of SCH, which is a 160-bed hospital with 21 ICU beds. At the time of the study, the SCH was the only children’s hospital in Mashhad and the only pediatric oncology, nephrology, and pediatric surgery center in the city. The traditional methods of blood culture and antibiogram were done using BHI (brain heart infusion) for culture and disc diffusion for antibiogram. We included all children with surgery for PAC implementation, and their data were extracted from the hospital files and completed by calling the parents. The files with incomplete data were excluded. Data were analyzed with SPSS 20. A Chi-square test was used to examine the significance of the association between categorical data. The probability values less than 0.05 were considered significant. The study was approved by the Research and Ethics Committees of the School of Medicine, Mashhad University of Medical Sciences, which is attached to the article and sent to the journal.
A case of PAC infection was defined as (1) fever and cellulitis and/or pus discharge along the subcutaneous pathway of the catheter (pocket infection) with or without positive blood culture, (2) fever during infusion from the catheter, and positive blood culture (peripheral or from the catheter), (3) persistent fever and positive blood culture (peripheral or from the catheter) and fulfillment of endocarditis criteria, and (4) fever during infusion from the catheter and resolution of fever with catheter removal and negative blood culture.
4. Results
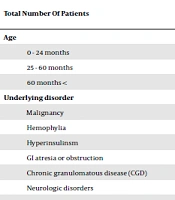
During six years, 70 children were registered for port CVC emplacement surgery in the SCH. The duration of having the catheter ranged from six months to six years at the time of the study. Table 1 demonstrates age distribution, underlying disorders, and the rate of infection in these children. As the table shows, 40% of all children with port catheters were younger than two years, and 71.4% had malignant disease. Catheter infection occurred in 16 (22.8%) cases. The rate of catheter infection was higher in children with hemophilia (P = 0.017), but it was not related to sex (P = 0.51), the age group of patients (P = 0.55), and the responsible surgeon (P= 0.98). The mean interval between the diagnosis of the primary disease and catheter emplacement was 10.8 and 6.1 months in those with and without catheter infection, respectively (P = 0.78).
Signs of catheter infection were fever and pocket infection in nine (56%) cases, chills and fever during catheter usage (without pocket infection) in six (37.5%) cases, and persistent fever in one (6.2%) patient (Table 2).
| All Children with Catheters, No.(%) | Children with Catheter Infection, No.(%) | |
|---|---|---|
| Total number of patients | 70 (100) | 16 (100) |
| Age | ||
| 0 - 24 months | 33 (47) | 6 |
| 25 - 60 months | 23 (33) | 7 |
| 60 months < | 14 (20) | 3 |
| Underlying disorder | ||
| Malignancy | 50 (71.4) | 12 (75) |
| Hemophylia | 4 (5.7) | 2 |
| Hyperinsulinsm | 4 (5.7) | 1 |
| GI atresia or obstruction | 4 (5.7) | 1 |
| Chronic granulomatous disease (CGD) | 2 | 0 |
| Neurologic disorders | 2 | 0 |
| Other singular cases | 4 | 0 |
| Nephrotic syndrome | ||
| Cystic Fibrosis | ||
| Lymphangectasis | ||
| Cystinosis |
| Clinical Presentations of Catheter Infections | Number of Patients: 16 (100%) | Cases with Positive Blood Culture: 12 |
|---|---|---|
| Fever | 16 (100%) | 12 |
| Pocket infection and fever | 9 | 6 |
| Chills and fever during catheter usage (without Pocket infection ) | 6 | 5 |
| Persistent fever (without Pocket infection ) | 1 | 1 |
The mean time between catheter emplacement and diagnosis of infection was 5.3 ± 5.5 months (range 1 - 22 months) in all 16 cases of catheter infection, but in the subgroup with pocket infection, it was 3.8 months (1-12 months). A great majority of catheter infections with pocket infection (77.7%) occurred during the first two months after catheter implementation, and in all of them (except one), the catheter was pulled out shortly after cellulitis.
Blood culture was positive in 75% (12 cases) of catheter infections. Gram-negative bacilli (five cases), Coagulase-negative staphylococci (CONS) (three cases), Candida (three cases), and group B streptococci (GBS) (one case) were the responsible organisms. In two patients, blood culture was positive for two organisms simultaneously (CONS and Candida; CONS and Enterobacter) (Table 3).
| Infections | Number of Positive Blood Cultures |
|---|---|
| Total | 12 (100%) |
| Gram-negative bacilli | 5 |
| Enterobacter agglomerans | 2 |
| Acinetobacter | 2 |
| Pseudomonas aeruginosa | 1 |
| Coagulase-negative staphylococci (CONS) | 3 |
| Candida | 3 |
| Group B Streptococci (GBS) | 1 |
The success rate of medical therapy alone was 68% in catheter infections. During the six years of the study of 70 patients with port CVC, the catheter was removed in 22 (31.4%) patients, which was due to infection in half of them. The mean time to removal was 15.3 months.
The parents of 92.8% of 70 children with port CVC were available by telephone call at the time of this study, and 79% of them were more satisfied with the port CVC than with the peripheral IV lines.
5. Discussion
This study is the first Iranian research that specifically refers to PAC infection in children. The rates of CLABSI and port infection (cellulitis over the catheter’s port) were 22.8% and 12.8%, respectively. According to the IDSA guideline, the definitive diagnosis of CRBSI needs clinical manifestations of infection (e.g., fever, chills, and/or hypotension) and growth of the same organism from both peripheral blood culture and the catheter tip, or two positive blood cultures by the same bacteria (one from the catheter and the other percutaneously) that meet CRBSI criteria for “Differential Time to Positivity” (DTP) (catheter blood culture is detected positive at least 2 h earlier than a simultaneously drawn peripheral blood of equal volume) or quantitative blood cultures (colony count of catheter blood culture is more than three times of simultaneously drawn peripheral blood of equal volume). Alternatively, when peripheral sampling is difficult, two quantitative blood cultures from different catheter lumens in which the colony count is at least three-fold greater in one of them is indicative of possible CRBSI (4).
Although quantitative blood culture is the most accurate test for diagnosis of CRBSI, it is not practical in most laboratories, even in the USA (5). The DTP has poor performance for many common enteric Gram-negative bacteria, and it has low sensitivity (42%) for S. aureus CRBSI despite high specificity (100%) (6, 7).
Peripheral sampling in infants and young children is more painful and difficult and thus is not routine practice when a CVC is available. Furthermore, peripheral blood culture sampling in infants uses smaller volumes (1 - 3 cc compared to 10 - 20 cc in adults), and the chance of positive culture drops below 70% in comparison with adults (8). Practically, for circumstances in which peripheral blood cultures are not obtained, positive blood cultures obtained through a catheter may be presumed to reflect true infection, in the absence of other identifiable sources of infection and in the presence of correlating clinical symptoms. Interestingly in almost all of the published research about PAC infections in children, this practical criterion is used for CRBSI, although for such cases, the term CLABSI is more appropriate (9).
After a PubMed search for English articles with keywords of “infection of port access catheters in children”, we found a few articles related to our research, which are summarized in Table 4. In Israel, the rate of CRBSI was 29.6% among 246 children with PAC in pediatric hematology-oncology ward. The rate of catheter extraction due to CRBSI was 61% (10). In Ankara, Turkey, the rate of pocket infection was 41.5% in 109 children with hematological diseases (with mean catheter duration of 379 days), in comparison with 12.8% in our case series. The rate of CRBSI was 98.1% in Ankara, while it was 22.8% in our group (11).
In Bengaluru, India, the rates of CRBSI and pocket infection among 209 children with cancer and PAC were 7.6% and 4.3%, respectively, and medical treatment without extraction was successful in 62.5% and 77% of them, respectively (12). In Singapore, pocket infection and CRBSI were reported in 2.9% and 18.9% of 175 children with cancer and port catheters. Coagulase-negative staphylococci (23.1%), Klebsiella (13.6%), Staphylococcus aureus (13.5%), and P. aeruginosa and Enterococcus faecalis (each 11.5%) were the most frequent causes of BSI. Medical therapy alone was effective in 69.6% of CRBSI (13).
In Switzerland, the overall catheter-related infection rate was 0.06% in a relatively old study (1987 - 1997) among 91 children with cancer and port catheter (14). In another study from Switzerland (Zurich), the rate of CRBSI was 16.5% among 155 children with cancer and PAC. Antibiotic alone without catheter removal was successful in 75%. The median time from implantation to catheter removal due to infection was 122 days. Coagulase-negative staphylococci were responsible for 91% of CRBSI, which led to catheter removal (15). In Bangalore, India, the rates of catheter-related bacteremia and pocket infection were 4.9% and 4.9% among 122 children with cancer and PAC (16). In Brazil, the rate of CRBSI was 50% among 188 children with cancer and port catheters. Gram-negative bacteria (46.8%) and Candida (12.8%) were the main causes of infection, and medical treatment without catheter extraction was successful in 97.9% (17).
This study has some limitations. The first one is its retrospective method. Because of the retrospective nature of the study, it was not possible to determine the place of blood sampling for each case; therefore, our definition of PAC infection was based on clinical signs and any positive blood culture. The second limitation is the method of reporting the rate of catheter infections. The best way to calculate the rate of CRBSI is based on the number of infections per 1000 catheter days. In this study, although we have reported the mean time between catheter implementation and diagnosis of infection (which was 5.3 ± 5.5 months), the rate of infections is based on the number of infections per number of catheters.
In summary, the rate of CLABSI at Sheikh hospital (22.8%) is in the middle range of other studies, but the pocket infection rate (12.8%) is higher than the other comparable reports (Table 4). This shows the importance of stricter infection control measures in operation rooms and during surgery because the port infection is more related to the implementation procedure, and CRBSI is related to the improper use of catheter during infusion and blood sampling.
| Setting of Study | Number of Patients | CLABSI (%) | Pocket Infection (%) | Rate of Catheter Extraction Because of Infection |
|---|---|---|---|---|
| Israel hematology | 246 | 29.6 | - | 61 |
| Brazil oncology | 188 | 50 | 2.1 | |
| Turkey hematology | 109 | 98.1 | 41.5 | - |
| India Bengaluru | 209 | 70.6 | 4.3 | 62.5 CRBSI; 77 POCKET |
| Singapore oncology | 175 | 18.9 | 2.9 | 30.4 |
| Switzerland oncology | 155 | 16.5 | - | 25 |
| India oncology | 122 | 4.9 | 4.9 | - |
| Iran (current study) mostly hematology- oncology | 70 | 22.8 | 12.8 | 32 |