1. Background
Enteric viral infections are a significant worry for public health (1). In 2016, diarrhea caused more than 1.6 million deaths worldwide and was the fifth leading cause of death (446,000 deaths) among those younger than five years (2). Nevertheless, the role of acute diarrheal disease in the morbidity and mortality of pediatric patients under five years of age is still significant, particularly in low-income populations living in areas with little or no infrastructure (3). Diarrhea has a notable influence on the quality of life of pediatric patients and their families (4). Viruses account for up to 40% of all severe cases of diarrhea among children in some emerging economies (5). Adenoviruses, rotaviruses, and noroviruses are the leading causes of viral gastroenteritis, especially in young children (6-8). Rotavirus is the primary etiologic agent of acute, severe gastroenteritis in infants worldwide, causing an estimated 128,515 deaths per annum (2). Rotavirus vaccination has not yet been introduced in many developing countries, including Iran.
The detection and identification of viruses are not routinely performed in Iran's hospitals and medical diagnostic laboratories. No comprehensive studies have been conducted to determine the role of viral agents in pediatric diarrhea in the central part of the country. Detailed investigations into the causal agents of diarrhea can provide valuable understanding for planning control strategies to reduce the incidence of this significant public health concern, mitigate its financial burden, and help improve the quality of health care facilities (9).
2. Objectives
The purpose of this study was to evaluate the frequency of adenoviruses, rotaviruses, and noroviruses in children in central Iran using PCR and reverse transcription (RT)-PCR methods.
3. Methods
3.1. Sample Collection
This study was confirmed by the Ethics Committee of Arak University of Medical Sciences, numbered 93-176-10. This descriptive cross-sectional study collected 173 out of 658 stool samples from under 16-year-old pediatric patients with diarrhea. The patients were referred to a Pediatric’s Educational-Therapeutic Diagnostic Center affiliated with the Arak University of Medical Sciences from the beginning of May 2015 to the end of May 2016. The medical center admits 1,500 children a year on average, of whom 268 are hospitalized in the infectious ward and 140 are hospitalized with infectious gastroenteritis. The pediatric diarrheal samples in this study were collected during all four seasons and were assessed immediately after receipt by the laboratory. Written informed consent was obtained, and questionnaires were filled in by the guardians or parents of participants under 16-years-old (Amir Kabir Hospital). Diarrhea was defined as at least three liquid or loose stools in a 24-h period accompanied by at least one of the following symptoms: abdominal pain, vomiting, fever, watery diarrhea, or dehydration.
Clinical signs and symptoms were recorded. All participants had diarrhea, dehydration, and fever at the time of referral, and none had any other illness. Each 5-mL fecal sample was immediately added to a phosphate-buffered saline (PBS) medium, placed on ice, and transported to the molecular laboratory in less than 40 minutes. Deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) were then either immediately extracted from the samples or after the samples were placed at -70°C for later extraction.
3.2. DNA and RNA Extraction
The RNA and DNA were extracted directly from stool samples using the QIAamp DNA Stool Mini Kit and QIAamp Viral RNA Kit (Qiagen GmbH, Hilden, Germany) following the manufacturer’s protocols. The quantity and purity of the extracted RNA and DNA were measured with a NanoDrop machine (Thermo Fisher Scientific, Waltham. MA, USA).
3.3. PCR and RT-PCR Amplification
We used PCR and RT-PCR assays to investigate the presence of pathogenic viruses (Table 1). All the primers and assays were developed and validated at the Virology Laboratory of Arak University of Medical Sciences. The analytical sensitivity and limit of detection (LOD) of the assays had already been determined by the laboratory with dilution series of the viruses using a commercial control for adenoviruses (Argene, Verniolle, France) and in vitro transcribed RNA for rota and noroviruses. For each control, dilutions of 100, 200, 500, 103, and 104 copies/mL of the materials were analyzed on three different days in six replicates. The LOD for adenovirus, rotavirus, and norovirus PCR tests was 212, 518, and 388 copies/mL, respectively.
| Pathogen | Primer | Size | Sequence 5' → 3' | References |
|---|---|---|---|---|
| Adenovirus | hexon | 169 bp | F: 5-CCCMTTYAACCACCACCG -3 | (8) |
| R:5- ACATCCTTBCKGAAGTTCCA-3 | ||||
| Rotavirus | GCMP | 205 bp | F: 5-CAAATGATTCAGAATCTATTG -3 | (6) |
| R: 5-GTTTCTGTACTAGCTGGTGAA -3 | ||||
| Genogroup I Norovirus | Mon431 | 213 bp | F: 5-TGGACICGYGGICCYAAYCA -3 | (7) |
| Mon434 | R:5- GAASCGCATCCARCGGAACAT -3 | |||
| Genogroup II Norovirus | Mon431 | 213 bp | F: 5-TGGACIAGRGGICCYAAYCA -3 | (7) |
| Mon433 | R:5-GAAYCTCATCCAYCTGAACAT -3 |
Primers Used in This Study
The final volume of the PCR was 25 µL, including 2 µL of DNA template (5 ng), 8 µL of double-distilled water, 0.5 µL of Taq DNA polymerase (2.5 units), 12.5 µL of master mix (1X), and 1 µL each of the forward and reverse primers (10 pM) (all bought from Yekta Tajhiz Company, Iran). The final volume of the RT-PCR was 20 µL and included 10 µL of master mix (1X), 5 µL of RNA template (5 ng), 0.5 µL each of the forward and reverse primers (10 pM), 0.4 µL of RT-Enzyme, and 3.6 µL of RNase-free water (all purchased from Yekta Tajhiz Company, Iran). A single positive sample for each pathogen was used for sequencing (Gene Fanavaran Company, Tehran, Iran), and the resulting sequences were confirmed using Basic Local Alignment Search Tool (BLAST) analysis. Positive controls of adenovirus, rotavirus, and genogroup I, II norovirus were obtained from the Virology Laboratory of Arak University of Medical Sciences.
4. Results
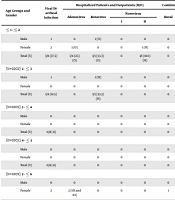
Of the 173 samples of pediatric diarrhea, eight (4.6%) were positive for diarrheal viruses, including four (2.3%) with adenoviruses, three (1.7%) with rotaviruses, and one (0.6%) with a genogroup II norovirus. Four (50%) male and four (50%) female patients were diagnosed with the viral infection. Of the participants, 129 (74.5%) were hospitalized, and 44 (25.4%) were outpatients. The hospitalization period ranged from three to five days (Table 2).
| Age Groups and Gender | Viral Diarrheal Infection | Hospitalized Patients and Outpatients (H/O) | Combining Population | Watery Diarrhea | Dehydration | Abdominal Pain | Fever | Bloody Stool | Vomiting | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenovirus | Rotavirus | Norovirus | Rural | Urban | |||||||||
| I | II | ||||||||||||
| ≤ 1 - ≤ 2 | |||||||||||||
| Male | 1 | 0 | 1 (O) | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 |
| Female | 2 | 1 (O) | 0 | 0 | 1 (H) | 0 | 2 | 2 | 2 | 2 | 2 | 1 | 1 |
| Total (%) | 3/8 (37.5) | 1/4 (25) (O) | 1/3 (33.3) (O) | 0 | 1/1 (100) (H) | 0 | 3/5 (60) | 3/8 (37.5) | 3/8 (37.5) | 3/8 (37.5) | 3/8 (37.5) | 1/8 (12.5) | 1/8 (12.5) |
| > 2 - ≤ 3 | |||||||||||||
| Male | 1 | 0 | 1 (H) | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 |
| Female | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total (%) | 1/8 (12.5) | 0 | 1/3 (33.3) (H) | 0 | 0 | 0 | 1/5 (20) | 1/8 (12.5) | 1/8 (12.5) | 1/8 (12.5) | 1/8 (12.5) | 0/8 (0) | 1/8 (12.5) |
| > 3 - ≤ 4 | |||||||||||||
| Male | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Female | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total (%) | 0/8 (0) | 0 | 0 | 0 | 0 | 0 | 0 | 0/8 (0) | 0/8 (0) | 0/8 (0) | 0/8 (0) | 0/8 (0) | 0/8 (0) |
| > 4 - ≤ 5 | |||||||||||||
| Male | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Female | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total (%) | 0/8 (0) | 0 | 0 | 0 | 0 | 0 | 0 | 0/8 (0) | 0/8 (0) | 0/8 (0) | 0/8 (0) | 0/8 (0) | 0/8 (0) |
| > 5 - ≤ 6 | |||||||||||||
| Male | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Female | 2 | 2 (1H and 1O) | 0 | 0 | 0 | 1 | 1 | 2 | 2 | 2 | 2 | 1 | 2 |
| Total (%) | 2/8 (25) | 2/4 (50) (1H and 1O) | 0 | 0 | 0 | 1/3 (33.3) | 1/5 (20) | 2/8 (25) | 2/8 (25) | 2/8 (25) | 2/8 (25) | 1/8 (12.5) | 2/8 (25) |
| > 6 - ≤ 7 | |||||||||||||
| Male | 1 | 1 (H) | 1 (H) | 0 | 0 | 2 | 0 | 1 | 1 | 1 | 1 | 1 | 1 |
| Female | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 |
| Total (%) | 2/8 (25) | 1/4 (25) (H) | 1/3 (33.3) (H) | 0 | 0 | 2/3 (66.6) | 0 | 2/8 (25) | 2/8 (25) | 2/8 (25) | 2/8 (25) | 1/8 (12.5) | 1/8 (12.5) |
| Final total (%) | 8/173 (4.6) | 4/8 (50) (2O and 2H) | 3/8 (37.5) (1O and 2H) | 0 | 1/8 (12.5) (1H) | 3/8 (37.5) | 5/8 (62.5) | 8/8 (100) | 8/8 (100) | 8/8 (100) | 8/8 (100) | 3/8 (37.5) | 5/8 (62.5) |
Age, Demographic Information, Number of Hospitalized Patients and Outpatients, and Clinical Symptoms of Pediatric Patients Infected With Viral Diarrhea
4.1. Adenovirus
One (25%) male and three (75%) female patients were diagnosed with the adenoviral infection. The average age of the infectious patients was four years and eight months. The youngest patient was a 12-month-old girl, and the oldest was a seven-year-old boy. The clinical symptoms of pediatric patients with adenovirus infection included abdominal pain, vomiting, fever, watery diarrhea, dehydration (100%), and bloody stool (75%).
4.2. Rotavirus
Of three (1.7%) patients infected with rotavirus, all (100%) were male, giving a male-to-female infection ratio of 1: 0. The average age of these patients was three years and six months. The clinical symptoms of the pediatric patients with rotavirus infection included abdominal pain, fever, watery diarrhea, dehydration (100%), vomiting (33.3%), and bloody stool (0%).
4.3. Norovirus
A one-year-old girl was infected with a genogroup II norovirus. The clinical symptoms of the pediatric patients with norovirus infection included abdominal pain, vomiting, fever, watery diarrhea, and dehydration (100%). Coinfection with adenoviruses and rotaviruses was observed in a seven-year-old boy (0.5%). Gastroenteritis severity was measured using the Vesikari Scoring System (Table 3).
| Severity | Total | Adenovirus | Rotavirus | Norovirus |
|---|---|---|---|---|
| Mild (%) (score < 7) | 0 | 0 | 0 | 0 |
| Moderate (%) (score 7 - 10) | 3/8 (37.5) | 2/4 (50) | 1/3 (33.3) | 0 |
| Severe (%) (score ≥ 11) | 5/8 (62.5) | 2/4 (50) | 2/3 (66.6) | 1/1 (100) |
Diarrheal Severity Distribution
5. Discussion
The pathophysiological role of most enteric viruses in gastroenteric disease remains uncertain, underlining the requirement for studying the epidemiology of these viruses (1). Viral gastroenteritis mainly occurs in the winter worldwide, with transmission chiefly through the fecal-oral route (10). This is the first report to estimate the frequency of adenoviral, noroviral, and rotaviral diseases in central Iran, which suggested that the frequency of these three most important species in the samples of pediatric patients was lower than that of bacterial agents (11-13). All samples were simultaneously tested for viruses, bacteria, and parasites, and the frequencies of DEC (35.1%), Campylobacter jejuni (19.5%), Shigella (8.2%), and Salmonella (9.1%) were higher than those of viral and parasitic agents and other bacterial agents (11-17). The samples were taken during DEC and campylobacteriosis outbreaks, which may explain the observed lower frequencies of viral enteropathogens (11-13).
In similar studies in Iran, adenoviral frequency in pediatric diarrheal stool samples was reported to be 2.3% in northern Iran and 9% in Shiraz (18, 19). Adenoviral frequency was 1.5% in Thailand and about 14% in Turkey (20, 21). The PCR test for diagnosing adenoviral infections is considered the gold standard (22). There are great hardships in organizing the diagnosis of adenoviral diarrheal infections, causing the prevalence of these viral infections to be underestimated (22).
In this study, the frequency of rotavirus in pediatric diarrheal patients was 1.7% (three cases). In similar studies in Iran, the rotaviral frequency in pediatric diarrheal stool samples was reported to be 35% in Ahvaz and 42% in Shiraz (19, 23). In other countries, the rotaviral frequency was 6.1% in Japan and 21% in France (20, 24). The differences between the results may be due to the duration of the studies, age of patients, different seasons of study, and the methods used for detecting rotavirus (25). This study was conducted during the hot and cold seasons of the year. The outbreaks of rotaviral gastroenteritis vary with season (19). Seventy-eight percent of rotaviral gastroenteritis occurred during winter, while only 2% of this infection was seen among diarrheal patients in summer. This pattern is generally seen in temperate climate areas, but it does not apply to all climate conditions. Another study mentioned that the seasonal pattern of rotaviral gastroenteritis was not global, and in countries within 10º of the equator, infections happened year-round (18).
In similar studies, the peaks of rotaviral infections in Iran (Tehran) and Latin America were in the autumn and winter (26, 27). Rotavirus outbreaks have been previously reported from different geographical locations in Europe and the USA (28). In Europe, rotavirus is widespread during January-March (29). Nevertheless, in tropical countries like Malaysia, a seasonal pattern of rotavirus frequency was not found (29). The annual seasonality of viral enteritis depends on climate globally. In colder climates, a higher frequency of rotavirus was found, and relatively low humidity and dry climates were detected in some reports (10). The higher prevalence in the present study may be due to the time family members stayed indoors in cold weather, leading to enhanced close contact transmission, as dry conditions encourage the aerosol formation of virus-laden particles from patients’ feces (10). Improvements in sanitation and the availability of clean water have not reduced the number of rotavirus diarrheal cases (30). Although the frequency of rotavirus in this study was low, more studies are needed to confirm the results before deciding on the need for vaccination in the central region of Iran.
In this study, the frequency of norovirus was 0.5% (one case). In similar studies in Iran, the noroviral frequency in pediatric diarrheal stool samples was reported to be 0.6% in Tabriz and 8.8% in Tehran (26, 31). The noroviral frequency was 5.8% in China and 14.8% in Japan (32, 33). The worldwide reported prevalence of norovirus in the fecal samples of pediatric patients with sporadic acute gastroenteritis has a broad range, depending on whether patients have been hospitalized or not, the age of children, and the study methodology (10).
This study observed coinfection with adenoviruses and rotaviruses in a seven-year-old boy (0.5%). In similar studies, adenovirus-rotavirus coinfection frequency in pediatric diarrheal stool samples was 1.3% in Italy and 8% in Turkey (21, 34). Adenovirus and rotavirus coinfections have been found between 1.2 and 8.2% in some other reports (10). An eight-percent coinfection with different rotaviruses and noroviruses was reported in China in 2011 (35).
Clinically, it would be challenging to separate viral gastroenteritis from gastroenteritis caused by other microorganisms, particularly bacterial gastroenteritis. Molecular techniques increased the overall diagnostic efficacy by 10% and by 2.5% for each adenovirus and rotavirus (10). Studies detecting adenoviruses using PCR have commonly reported higher frequencies (36).
The weaknesses of this study include the small number of samples (due to financial constraints) from a limited area. Samples from all over the country will provide more robust results. This study's strengths include standardized sampling, transportation, and storage methods. The DNA/RNA extraction and PCR/RT-PCR test were also carefully performed.
5.1. Conclusion
Adenovirus, norovirus, and rotavirus were rarely found as viral agents responsible for gastroenteritis among children in central Iran. Although the viral gastroenteritis frequency has been lower in this area than bacterial gastroenteritis frequency (11-13), the long-term monitoring of all enteropathogenic agents should continue to screen for the unexpected occurrence of outbreaks and get better visibility on the precise seasonality of viral and bacterial diarrhea. Doing so will help better understand the precise epidemiology of this severe disease and improve appropriate medical diagnosis and management of diarrhea in pediatric patients.