1. Introduction
At the end of 2019, Coronavirus disease 2019 (COVID-19) emerged in Wuhan, China, and quickly became a pandemic (1). The disease is caused by severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2), which produces an acute respiratory syndrome (2). The typical manifestations of COVID-19 include respiratory involvement (1), usually presenting with fever, cough, and shortness of breath (respiratory distress) (2). Gastrointestinal (GI) symptoms have also been reported as the first clinical manifestation (3), with a frequency varying as 3% - 50% of cases (4). The GI involvement can manifest with diarrhea, vomiting, and abdominal pain (2). In a broad study in China, nausea or vomiting was reported in 5% and diarrhea only in 3.8% (5). In other studies, the frequency of diarrhea was 2% - 10.1%, and nausea and vomiting were found in 1% - 1.1% of cases (2). Abdominal pain was less common and was published as 2.5% - 2.8% (2). Some authors have shown that RNA of coronavirus is found in feces (6). On the other hand, there are virus receptors (7) in small and large intestine biopsy specimens of the GI epithelial cells, and active viral proliferation was also noted (8). This fact indicates the ability of the virus to infect and replicate in the GI tract. The GI symptoms of COVID-19, including diarrhea, nausea, vomiting, abdominal pain, and GI bleeding, have been reported in previous studies, while the reports of dysentery in children with COVID-19 are very limited (9). Dysentery presents with high fever, abdominal pain, tenesmus, and the presence of blood and mucus in the stool. Moreover, there are white and red blood cells in the stool specimens. The mechanism of dysentery is epithelial destruction due to cytotoxin secretion following bacterial and amoebic infections or penetrating due to bacterial infection (10). However, in viral infections, dysentery has not been reported yet. In adults, cases of dysentery following coronavirus infection have been published and attributed to the symptoms of hemorrhagic colitis (4, 10, 11). The aim of presenting pediatric patients with dysentery is to depict this clinical manifestation as one of the signs of the GI involvement of novel coronavirus infection in children.
2. Case Presentation
We present 12 patients with COVID-19 with a clinical manifestation of dysentery. Half of these individuals were girls. The youngest was 10 months, and the eldest was 108 months. All patients had positive RT-PCR results. None had underlying illnesses or recent travel history. However, all children had contact with a first-degree relative with non-digestive COVID-19. In addition to diarrhea, we found nausea, vomiting, and fever. Obvious blood with diarrhea was observed in only three patients, accompanied by mild to moderate dehydration. In only four cases, mild abdominal pain was reported, and no children complained of respiratory symptoms.
A stool sample was collected to check for white and red blood cells, parasite ova, and stool culture (for Campylobacter, Salmonella, Shigella, Escherichia coli, and Yersinia, the producers of Shiga toxin) and was sent to the hospital laboratory. Another stool sample was sent to the Faculty of Health Laboratory of Tehran University of Medical Sciences for detecting Clostridium difficile toxins. All stool cultures were negative, and parasitic ova and toxins were also negative.
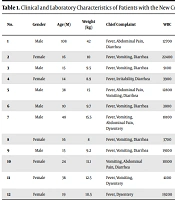
Antibiotics of the third-generation Cephalosporins were started at the beginning and before stool culture for relatively ill patients with high fever, considering the possible bacterial causes of dysentery. When the RT-PCR result was positive, and the stool cultures were negative, the antibiotics were discontinued (the maximum duration of antibiotic therapy was 72 h). Note that an anal swab was not performed for Coronavirus infection. Children received supportive care for dehydration and electrolyte disturbances during hospitalization with oral hydration therapy, serum therapy, zinc, and probiotics supplementation. They did not receive antiviral medications, systemic or topical corticosteroids, anticoagulants, or immunoglobulins. Vital signs and pulmonary monitoring, especially O2 saturation, were charted regularly. A chest X-ray was taken at the beginning of hospitalization despite the absence of respiratory symptoms and impaired oxygen flow, which were reported to be normal. However, no lung scan was performed. All children underwent abdominal and pelvic ultrasound and echocardiography to rule out multisystem inflammatory syndrome in children (MIS-C). All these tests were normal. Table 1 summarizes the clinical and laboratory characteristics of patients. All children were discharged with improved diarrhea, in good general condition, and without complications. In the follow-up after discharge, complete recovery was observed in all of them.
| No. | Gender | Age (M) | Weight (kg) | Chief Complaint | WBC | N % | L % | Hb | PLT | CRP | S/E | LDH | ALT | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WBC | RBC | |||||||||||||
| 1 | Male | 108 | 42 | Fever, abdominal pain, diarrhea | 12700 | 86 | 9 | 12 | 185000 | 65 | 10-15 | 20-25 | 341 | 14 |
| 2 | Female | 16 | 10 | Fever, vomiting, diarrhea | 22400 | 50 | 30 | 12.3 | 226000 | 38 | 30-35 | 15-20 | 397 | 28 |
| 3 | Male | 15 | 9.5 | Fever, vomiting, diarrhea | 9100 | 71 | 18 | 12.7 | 239000 | 5 | 10-15 | 20-25 | 623 | 38 |
| 4 | Female | 14 | 8.9 | Fever, irritability, diarrhea | 3900 | 51 | 41 | 11.2 | 153000 | 10 | 30-35 | Many | 350 | 29 |
| 5 | Male | 38 | 15 | Fever, abdominal pain, vomiting, diarrhea | 12800 | 28 | 45 | 12.9 | 338000 | 60 | 30-35 | Many | 280 | 25 |
| 6 | Male | 10 | 9.7 | Fever, vomiting, diarrhea | 3800 | 21 | 52 | 11.9 | 203000 | 100 | 3-4 | 30-35 | 48 | 40 |
| 7 | Male | 48 | 15.5 | Fever, vomiting, abdominal pain, dysentery | 18100 | 88 | 9 | 12.2 | 175000 | 30 | 10-15 | 30-45 | 480 | 20 |
| 8 | Female | 16 | 8 | Fever, vomiting, diarrhea | 3700 | 68 | 24 | 12.3 | 226000 | 98 | 30-35 | 15-20 | 397 | 32 |
| 9 | Male | 15 | 9.2 | Fever, vomiting, diarrhea | 19100 | 67 | 20 | 12.5 | 210000 | 7 | 20-25 | 10-15 | 623 | 38 |
| 10 | Female | 24 | 11.1 | Vomiting, abdominal pain, diarrhea | 10100 | 43 | 53 | 11.9 | 187000 | 56 | 10-15 | 10-15 | 340 | 28 |
| 11 | Female | 36 | 12.5 | Fever, vomiting, dysentery | 4100 | 55 | 39 | 11 | 210000 | 77 | 15-20 | Many | 280 | 32 |
| 12 | Female | 19 | 10.5 | Fever, dysentery | 19200 | 35 | 61 | 11.4 | 155000 | 88 | 30-45 | Many | 560 | 22 |
3. Discussion
SARS-CoV-2 tends to involve the GI tract with a lower prevalence of lung disease. The angiotensin-converting-enzyme receptor 2 is responsible for the entry of SARS-CoV-2 into cells and is abundantly expressed in the GI epithelium (9). There are cases of definitive COVID-19 in children who do not show obvious respiratory symptoms and represent other manifestations, such as GI involvement. Other differential diagnoses are made and, as a result, are identified later and lead to further spread of the infection in society (12). GI involvement has been reported in up to half of the cases of COVID-19 (4). The most common GI manifestations include nausea, vomiting, diarrhea, and abdominal pain (2). Only one dysentery case has been reported in children with this disease (9). Similar to other GI viral infections, COVID-19 may cause inflammation directly through cellular infection or indirectly by inflammatory cytokines (4).
In patients with dysentery, the common causes of infection, such as Campylobacter, Salmonella, Shigella, Yersinia, and E. coli, that produce Shiga toxin, should be ruled out tested negative in stool examination and culture. The possibility of developing pseudomembranous colitis secondary to C. difficile (toxin) was also ruled out by examining stool for the relevant toxin and the lack of a history of receiving antibiotics before the disease. The GI bleeding in patients with COVID-19 can be secondary to hypoxia and mucosal ischemia (13), mesenteric thrombosis (14), and stress ulcer or rectal ulcer secondary to the rectal tube (15). None of our patients had severe respiratory manifestations or hypoxia, and there was no mucosal ischemia. In the previous studies, mesenteric thrombosis was reported in patients with COVID-19, most of whom were old and had underlying diseases, including hypertension, diabetes, obesity, obstructive sleep apnea, anxiety, chronic bronchitis, heart transplantation, and a history of essential thrombocytosis. Thrombosis was reported in only one 9-year-old child with idiopathic medullar aplasia (14). However, thrombosis was one of the final manifestations of COVID-19 in hospitalized patients (14).
Stress ulcer was not considered because these children did not have a history of GI bleeding, had stable vital signs, and did not have a noticeable decline in hemoglobin. No patients needed a rectal tube, and the possibility of a rectal ulcer was also ruled out. Because dysentery started acutely and improved in 2 weeks (no criteria for chronic dysentery), inflammatory bowel disease diagnosis was not raised. None of the studied patients had clinical or laboratory evidence of liver involvement, and the levels of aminotransferases were reported to be normal, as shown by Sattar et al. (4). In addition, none of our children met the diagnostic criteria for MIS-C. Endoscopy and colonoscopy could pose a risk to medical staff and, in general, had to be limited to patients with life-threatening GI bleeding or esophageal obstruction due to a foreign body (15, 16). Our patients did not have life-threatening symptoms or severe bleeding. Therefore, they did not undergo endoscopy.
Finally, COVID-19 can represent non-pulmonary manifestations, such as dysentery. Symptoms were usually self-limiting and did not require invasive diagnosis or antiviral therapy. However, it should be noted that this manifestation of the new coronavirus infection is unlike other viral intestinal infections. In viral infections, dysentery is not reported. We aimed to inform physicians about this symptom of COVID-19 to allow rapid detection and early preventive measures for disease spread. It is recommended to avoid unnecessary antibiotics due to the viral cause.