1. Background
Thanks to the advancement of technology and promotion of global health standards, the prevalence of new viruses decreased, but in late 2019, a series of unusual pneumonia was reported in Wuhan, Hubei Province, China (1, 2), and following the analysis of various respiratory samples, researchers found that the etiologic agent was a newly emerged coronavirus called SARS-CoV-2, hence, WHO officially named the disease coronavirus disease 2019 (COVID-19) (3, 4). A common feature of emerging infectious diseases is the rapid increase in incidence, which strains and potentially overwhelms medical services. The current pandemic placed a significant physical and psychological burden on patients, health care workers (HCWs), and certain risk groups.
The results of epidemiological studies have revealed that the mortality rate of COVID-19 varies in different age groups and affects older people more frequently (4). Fortunately, this rate is less reported in children (5, 6). Some studies showed that women are less susceptible than men, probably due to factors such as sex hormones and the high expression of coronavirus receptors (ACE2) in men, as well as lifestyle (women smoke and drink alcohol less than men), so they are safer against the disease (7, 8). Immunity is also an important factor in determining the severity of the disease, so people with underlying diseases such as diabetes, hypertension, heart disease, and cancer due to weak immune systems are more vulnerable to the disease, and strengthening their immune systems is helpful. A healthy lifestyle, proper diet and nutrition, a balanced weight, and the intake of the required vitamins B, C, D, E, and minerals, can help strengthen the immune system and, thus, reduce the severity of COVID-19 (9-11). Another group with high exposure to the infection of COVID-19 is HCWs. HCWs are at the front line of the fight against the disease that makes up a high percentage of patients developing SARS-CoV-2 infections due to confirmed or intimate physical contact with suspected cases. As a result of factors such as high workload, stress, and confronting the risks of infection for themselves and their families, there has been an increased psychological burden that may exacerbate the disease symptoms (12, 13). By characterizing and limiting risk factors, making preventive decisions, and taking action to reduce exposure, this important goal, i.e., protecting HCWs as the most valuable health resource, is possible.
2. Objectives
Considering the importance of the outbreak of Covid-19 among the HCWs of hospitals as a large group in close contact with the general population and the outbreak of this infection in them, leading to an increase of the infection in the community, as well as limited research in this area, the present study aimed to determine the prevalence of coronavirus in HCWs in Abu Ali Sina Transplant Hospital (AASTH) and Amir al-Momenin Burn Injury Hospitals (AABIH) in Shiraz, southern Iran and to evaluate the risk of SARS-CoV-2 transmission in these hospitals to with immunocompromised hospitalized patients.
3. Methods
3.1. Data Collection
In the present cross-sectional study, nearly all HCWs (1232) employed in the two specialist hospitals in Shiraz, AASTH and AABIH, for whom qPCR assays for SARS-CoV-2 were performed between April 11, 2020, and June 16, 2021, were enrolled. All the oro- and nasopharyngeal samples were sent and tested to the Corona Laboratory of Microbiology Research Center (CMRC), Shiraz University of Medical Sciences, Shiraz, southern Iran. By using a questionnaire, data were collected regarding age, sex, working location (medical and service personnel), risks of exposure to COVID-19 patients (low, medium, and high) (14), pregnancy, the presence and types of underlying diseases, such as diabetes, cardiovascular disease, chronic lung disease, chronic neuromuscular disease, kidney disease, and liver disease, and the need for hospitalization due to COVID-19. The sources of the data were obtained from the respective internet site of http://coronalab.sums.ac.ir/. The high-risk group was HCWs, including physicians, nurses, and service personnel with direct exposure to COVID-19 patients; the medium-risk group included HCWs with indirect contact with the patients, and the low-risk group was HCWs, such as administrative personnel with no exposure to the patients. The study design and proposal were approved by the ethics committee of Shiraz University of Medical Sciences (code: IR.SUMS.REC.1399.1319).
3.2. Laboratory Procedures
Samples were prepared using two sterile dacron separate swabs from the throat and nose of HCWs with suspected SARS-CoV-2 infection. The clinical samples were transferred using a sterile virus transport medium containing antifungal and antibacterial supplements. We avoided repeated freezing and thawing of the clinical samples. Viral genomes were extracted from 200 μL of each sample by Sina Pure kits (SinaClone Co. -Iran); afterward, the extracted samples were subjected to viral genome quantification with multiplex TaqMan real-time PCR assay kits (Pishtaz Teb Zaman, Iran). For each sample, the final volume of each PCR reaction was 20 μL. The assay simultaneously targeted specific regions of RdRp (FAM-labeled) and N (HEX-labeled) of the viral genome and internal control of RNaseP (ROX-labeled) using the Applied Biosystems StepOne Plus Real-Time PCR instrument (ABI, Thermo Fisher Sci).
3.3. Statistical Analysis
In this study, we identified the study group using descriptive statistics classified by SARS-COV-2 PCR results. Classified variables were reported as numbers and percentages. There were no missing data. The chi-square test was employed to determine statistically significant differences between categorical variables. Data were analyzed using SPSS software version 26 at a significant P < 0.05.
4. Results
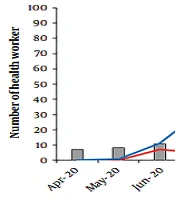
One thousand two hundred thirty-two samples were obtained from eligible HCWs, including 967 and 265 from AABIH and AASTH. Some HCWs had repeated sampling (ranging between 1 - 8 times) (Tables 1 and 2). The study group comprised 538 males (44%) and 694 females (56%). The mean age ± SD was 32.35 years, with an age range between 20 and 69 years and a majority of those in the 20 - 40 age range (88%) (Table 3). In total, 715 (58%) of the HCWs were in a high-risk group of exposure, 242 (20%) in the medium-risk group, and 275 (22%) in a low-risk group. As revealed, 373 (30%) HCWs in both hospitals were infected with COVID-19 during 14 months post-outbreak of the SARS-CoV-2 pandemic; 24% and 6% from AASTH and AAMBIH, respectively. The infection rates among HCWs in each of the two hospitals were more prevalent in June (2020), October (2020), November (2020), and April (2021) than in other months. While in the first months of SARS-CoV-2 emergences in Iran, the lowest monthly incidence was reported among the HCWs of these two hospitals, it increased from June to November 2020. In December 2020, thanks to stricter control of the disease, the prevalence and number of infected cases reduced, while the rate remained the same for the following three months. In April 2021, the infection trend shifted and rose again, which was consistent with the statistics in the general Iranian population (Figure 1) (15).
| Characteristics | All Staff | High-risk Group | Medium-risk Group | Low-risk Group |
|---|---|---|---|---|
| No. (%) | 265 (100) | 83 (31) | 119 (45) | 63 (24) |
| Age range (y) | 20 - 65 | 20 - 52 | 22 - 53 | 24 - 65 |
| Sex | ||||
| Men | 131 (49) | 42 (51) | 55 (46) | 34 (54) |
| Women | 134 (51) | 41 (49) | 64 (54) | 29 (46) |
| Concomitant diseases | 3 (1) | 0 | 2 (2) | 1 (2) |
| Diabetes | 6 (2) | 1 (1) | 4 (3) | 1 (2) |
| Cardiovascular disease | 6 (2) | 1 (1) | 4 (3) | 1 (2) |
| kidney disease | 2 (1) | 0 | 2 (1) | 0 |
| Liver disease | 0 | 0 | 0 | 0 |
| Chronic obstructive pulmonary disease | 1 (0.3) | 1 (1) | 0 | 0 |
| Chronic neurological disease | 1 (0.4) | 0 | 0 | 1 (2) |
| Immunosuppressive disease | 0 | 0 | 0 | 0 |
| Malignancy | 1 (0.4) | 0 | 1 (1) | 0 |
| Characteristics | All Staff | High-risk Group | Medium-risk Group | Low-risk Group |
|---|---|---|---|---|
| No. (%) | 967 (100) | 632 (65) | 123 (13) | 212 (22) |
| Age range (y) | 20 - 69 | 20 - 62 | 20 - 56 | 22 - 69 |
| Sex | ||||
| Men | 407 (42) | 188 (30) | 64 (52) | 155 (73) |
| Women | 560 (58) | 444 (70) | 59 (48) | 57 (27) |
| Concomitant diseases | 3 (0.3) | 2 (0.3) | 0 | 1 (0.5) |
| Diabetes | 7 (0.7) | 5 (0.8) | 0 | 2 (1) |
| Cardiovascular disease | 13 | 9 (1.4) | 1 (0.8) | 3 (1.4) |
| Kidney disease | 0 | 0 | 0 | 0 |
| Liver disease | 1 (0.1) | 1 (0.2) | 0 | 0 |
| Chronic obstructive pulmonary disease | 3 (0.3) | 2 (0.3) | 0 | 1 (0.5) |
| Chronic neurological disease | 4 (0.4) | 3 (0.5) | 1 (0.8) | 0 |
| Immunosuppressive disease | 1 (0.1) | 1 (0.2) | 0 | 0 |
| Malignancy | 1 (0.1) | 0 | 1 (0.8) | 0 |
| Age Range | Percentage (%) | Female (n = 694) | Male (n = 538) | P Value |
|---|---|---|---|---|
| (Pos/Neg) | (Pos/Neg) | |||
| 20 - 30 | 52 | 135/255 (0.53) | 83/169 (0.49) | 0.661 |
| 30 - 40 | 36 | 67/161 (0.42) | 50/160 (0.31) | 0.188 |
| 40 - 50 | 9 | 19/44 (0.43) | 10/42 (0.24) | 0.179 |
| 50 - 60 | 2 | 2/10 (0.2) | 4/14 (0.28) | 0.545 |
| 60 ≤ | 1 | 0/1 (0) | 3/3 (1) | 0.571 |
The infection rates among HCWs in AABIH and AASTH during different months compared with the general population in Iran (15).
218 (58%) of the total HCWs infected with COVID-19 were within the 20 - 30 year age range, of which 135 (62%) were female, and 83 (38%) were male (P < 0.0001). Likewise, the female-to-male ratio of suspected patients was significantly higher in other age groups, except for the groups over 50 years of age. Meanwhile, no statistically significant differences between males and females in the positivity rate were observed in any age group (Table 3). According to Figure 2 and Table 4, the PCR positivity rate was significantly higher among symptomatic HCWs than in asymptomatic ones.
| Groups and Total (N= 373) | P Value | AABIH (N= 71) | P Value | AASTH (N= 302) | P Value |
|---|---|---|---|---|---|
| High-risk | < 0.0001 | 0.371 | < 0.0001 | ||
| Sym: 175 | Sym: 11 | Sym: 164 | |||
| Asym: 55 | Asym: 7 | Asym: 48 | |||
| Medium-risk | < 0.0001 | 0.002 | 0.0052 | ||
| Sym: 52 | Sym: 32 | Sym: 20 | |||
| Asym: 12 | Asym: 9 | Asym: 3 | |||
| Low-risk | < 0.0001 | 0.587 | 0.0001 | ||
| Sym: 59 | Sym: 7 | Sym: 52 | |||
| Asym: 20 | Asym: 5 | Asym: 15 |
Abbreviations: Sym, symptomatic; Asym, asymptomatic.
Signs and symptoms associated with COVID-were present in 50 out of 71 (70%) definite cases among the personnel of AABIH and 164 out of 302 (54%) personnel of AASTH. No symptoms were reported in 69 cases (6%) of 1232 HCWs of both hospitals, but due to lack of access to information or negligence in collecting data, they could not be classified as asymptomatic.
Respiratory symptoms such as cough, sore throat, runny nose, shortness of breath, and general signs such as fever, headache, fatigue, dizziness, myalgia, and general weakness were significantly higher in PCR-positive patients (PCR-positive P ≤ 001). Gastrointestinal symptoms such as abdominal pain, nausea, and diarrhea were also significantly more common in PCR-positive patients (P ≤ 01). Mild pneumonia, severe pneumonia, and respiratory distress syndrome were the symptoms with no difference between positive and negative PCR patients (Figure 3). In the present study, we observed a limited number of underlying diseases in HCWs, including 13 patients (1%) with diabetes, 19 patients (2%) with cardiovascular diseases, four patients (0.5%) with chronic pulmonary disease, two patients (0.2%) with kidney disease, one patient (0.08%) with liver disease, five patients (0.4%) with chronic neurological disease, two patients (0.2%) with malignancy, and one patient (0.08%) with the weakened immune system. Among the HCWs with the above-mentioned underlying diseases, 8 cases with cardiovascular disease, 6 with diabetes, and 2 with lung disease had positive PCR test results. Also, among the personnel, 1 case with chronic kidney and 1 with a neurological disease were reported with positive PCR test results. Fortunately, none of the personnel needed to receive mechanical therapy.
5. Discussion
This study aimed to determine the prevalence of COVID-19 infection among health care workers in Shiraz, southern Iran, given the high risk of COVID-19 disease among patients in these two hospitals. Due to the special conditions of patients hospitalized in these two hospitals, it is very critical to know the level of infection among the HCWs. All necessary actions were taken to reduce the infection and transmission among the HCWs. Considering the weakening of the immune system and skin damage during burns and transplants, the patients of these two hospitals are among the groups at higher risk of infection with SARS-CoV-2. In the case of burns, the burned patient is prone to various infections due to skin damage and the weakening of the immune system. During burns, T cell activity of the inflammatory cytokines is decreased, eventually leading to a general defect in host defense (16). Transplant patients, like other immunocompromised patients, exhibit weakened resistance to infections, atypical clinical symptoms, and higher morbidity and mortality rates (17). In addition, most of the published articles have highlighted the severe and rapid progression of COVID-19 in immunocompromised patients such as transplant recipients (10, 18-21). Therefore, ensuring that the staff of these hospitals are aware of the prevalence of COVID-19 among them as well as ensuring that the SARS-CoV-2 infection chain is broken in these hospitals, is crucial to reducing the incidence of the disease and consequentially the mortality in hospitalized patients.
In our study, from 1232 HCWs entered this study, 373 (30%) had positive SARS-CoV-2 PCR test results, which indicates further infection of HCWs in the high-risk group. The findings of this study were in agreement with the research of Buonafine et al., conducted in Brazil, with a statistical population consisting of 295 symptomatic HCWs. Out of 125 definite cases of COVID-19, among 295 symptomatic HCWs, the highest infection rates were reported in physicians and nurses who had direct contact with the patients, and only 6.4% of the other groups contracted COVID-19 infection (22). Similarly, in a study by Barrett et al., in two New Jersey hospitals, the prevalence of infection among HCWs who had direct contact with COVID-19 was reported higher than in other groups. The infection of COVID-19 among the participants of these two hospitals was 7.3% of the HCWs, with the highest infection rate reported among the nurses because they had the highest rate of contact with SARS-CoV-2 infected patients (23). Of the 9648 employees of Tongji Wuhan Hospital in China, 110 employees had definite COVID-19, including 26 (23.6%) physicians, 62 (56.4%) nurses, and 22 (20%) health care assistants (24). In another study conducted in Italy by Lahner et al., HCWs were evaluated by PCR test, and 58 (3%) were positive, consisting of 53 people (91.4%) with direct contact with COVID-19 patients and 5 (8.6%) were from other hospital personnel (25). In addition to the results of PCR-based research, the results of serological research also support a higher risk of health care personnel in direct contact with COVID-19 patients. For example, in a study conducted in Denmark, out of 28,792 healthcare workers who underwent corona antibody screening, 1163 (4%) were positive. The prevalence of serum antibodies was higher among healthcare personnel (physician, nurse, and assistant nurse) with direct contact with the patients than in the other groups (26). Among the definite cases, 58% and 14% of the personnel had direct or indirect contact (radiographer, midwife, laboratory personnel, medical and paramedical students), respectively, and 28% were administrative and other personnel. In another study, 249 HCWs in Nashville, Tennessee, including 105 nurses, 86 service providers (physicians and advanced practice providers), 17 radiologists, and 41 other health care personnel, were examined for one month, of which 19 (8%) were positive, and the serum prevalence of antibodies among this personnel was reported as 5 (26.3%), 8 (42.1%), 5 (26.3%) and 1 (5.3%), respectively (27). The study by Korth et al. (28) in Germany examined 316 healthcare personnel.
Participants in this study were classified into three groups based on the number of encounters with COVID-19 patients: (1) high-risk group: Daily contact with the patients in designated wards and intensive care units; (2) moderate risk group: Daily contact with the non-COVID-19 patients; and (3) low-risk group: No daily contact with the patients. As reported, 1.6% were diagnosed with SARS-CoV-2-IgG antibodies, of which 1.2% belonged to the high-risk group, and 5.4% were in the moderate-risk group (28). Our findings contradict observations in the UK, as in a study by Hunter et al. (29), 15% had positive PCR test results among the 81% of personnel who had direct contact with the patients. In addition, among 86 (8%) personnel who had indirect contact with the patients, 14 (16%) and among 109 (11%) administrative and other personnel, 2 (18%) had positive PCR test results, indicating that there is a significant difference in the rate of infection among different groups of personnel. Due to the placement of HCWs in the first line of the fight against COVID-19 disease, the prevalence of the disease, and the rate of death, personnel are more vulnerable to psychological disorders and anxiety. A review of the results of Liu et al. study on Chinese medical personnel shows that out of 512 personnel, about 32.03% had direct contact with patients infected with COVID-19. This study showed that people who had direct contact with patients infected with COVID-19 had a higher anxiety index than those without direct contact with these patients (30). Another study of Chinese healthcare workers found that HCWs experienced more symptoms, including insomnia, anxiety, depression, and obsessive-compulsive disorder, than non-medical healthcare workers (31).
It is noteworthy that based on the results of this study, among the personnel who had direct contact with infected patients, women were with a higher rate of psychological symptoms.
In the present study, out of 373 definite cases, 60% were women, and 40% were men, indicating a higher correlation between the infection rate and psychological symptoms in women. Our findings are also consistent with the research by Lai et al., reporting that psychological symptoms were more frequent among women and those at the forefront fight against the infection, suggesting the need for special attention (32).
In reviewing the recorded symptoms, in addition to HCWs with positive test results, some with negative PCR test results reported several symptoms, which could have been to an improper sampling of personnel, processing, transfer, low sample concentrations, improper storage conditions of solutions, or contaminants created during sample processing, that ultimately leading to the occurrence of false-negative results. We found that symptoms like fever, cough, sore throat, myalgia and fatigue and general weakness of the body, etc., are more common among the cases with positive SARS-CoV-2 test versus the cases with negative COVID-19 SARS-CoV-2 test. Our results are also consistent with Stubblefield et al., the incidence of symptoms like fever, cough, myalgia, and sore throat among personnel with positive SARS-CoV-2 test was higher than in personnel with negative test results (27). Furthermore, in a study by Mani et al., the incidence of three symptoms, fever, headache, and myalgia/fatigue, reported among HCWs with positive PCR test results were higher than in those with negative results (33).
The first limitation of the study was that the results were based on self-reports. Second, details for getting exposed levels to patients were shortened. For example, we did not ask for experience level, type of PPE used (mask, respirator, air purifying respirator, etc.), or PPE training received and frequency of exposure to SARS-CoV-2 infected patients.
5.1. Conclusions
We observed a high prevalence of COVID-19 among HCWs with direct contact with the patients compared to other personnel and a higher infection among women. To protect the medical and service personnel and at the same time reduce the transmission of the disease to the patients, it is necessary to identify the deficiencies and eliminate them.