The worldwide emergence of MRSA is a remarkable challenge for public health (16 - 18) Based on centers for disease control (CDC) reports, 1% of all Staphylococcal infections and 50% of healthcare-associated Staphylococcal infections are caused by MRSA (
3).
In this examination of 229 samples, 21 (12%) MRSA strains were detected. Similarly with our results, in trials conducted in Germany, in 2007, (
13) and west of Iran, in 2013 (
14), the prevalence of MRSA isolates among health care providers carriers was reported 11.3% and 17.57%, respectively.
Compared to studies in Germany (6.5%), The Netherlands (1.4%), Shiraz, Iran (5.3%), Pediatric Infections Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran (3.2%), Switzerland (3.3%), the USA (3.4%), France (6.6%) and the UK (6.7%), the prevalence of MRSA strains were lower than in our study (
9,
15-
21).
Rezaei et al. considered colonization with methicillin resistant and methicillin sensitive
Staphylococcus aureus subtypes in patients with atopic dermatitis. They found a higher rate (33%) of MRSA colonization in the nasal cavity. The MRSA was one of the most frequent organisms that was found on their skin (
22).
The high percentage of MRSA in health care providers, especially who do not exhibit any symptoms or signs of severe disease, is very dangerous, mainly because they can cause epidemic diseases, raise the occurrence of severe diseases among patients, and enhance mortality rate by transfer of the strains to patients (
22).
Linezolid is one of the most effective oral medications used for outpatient treatment of MRSA infections that are resistant to other antibiotics. In this study, there was no resistance against linezolid, in both groups (
23).
Resistances to antibiotics among the MRSA isolates were higher compared to MSSA, although without no significant difference was between them. The MRSA isolates showed variable resistance to clindamycin, ceftriaxone, cefpodoxime, azithromycine, and erythromycin (
23). Resistance to penicillin and clindamycin (
23,
24) was similar with the other studies. Moderate resistance to other conventional antibiotics (such as azithromycin, erythromycin, clindamycin, cefpodoxim, ceftriaxone was detected in MRSA (
22).
By definition, all MRSA isolates carry the
mecA gene, which confers resistance to all beta-lactam antibiotics, including cephalosporins and carbapenems. In our study and in similar studies, several MRSA are susceptible to a number of beta lactams, such as cephalosporins (
25-
27).
Several additional auxiliary factors increase MRSA susceptibility to beta-lactams or other clinically used antibiotics. These auxiliary genes, including femX (fmhB), murE, pbp2, SAV1220, SAV175 and femD (glmM), were regularly identified to give back beta-lactam susceptibility of MRSA strain context. Supplementary essential genes contain the majority of the mur and pbp genes and involve synthesizing the peptidoglycan precursor that shares this phenotype by antisense incorporation, although to different extents among several beta-lactam antibiotic classes. Genes not formerly known to change MRSA beta-lactam susceptibility, including those involved in cell division (ftsA, ftsW, ftsZ), transcription (hu), secretion (spsB), cell wall teichoic acid biosynthesis (tarL) and SAV1892, are susceptible to interfere in cell wall synthesis or repairing (
28).
In every region, rating of the resistance or sensitivity of MRSA against conventionally antibiotics is different. When antimicrobials are considered for therapy, susceptibility testing for antibiotics for every isolate of MRSA should be done. This study showed that all MRSA isolates were significantly less sensitive to antibiotics, compared with MSSA isolates (
24).
A remarkable result in this study was the high percentage of MRSA in health care providers. Unfortunately, it is considered that the rate of MRSA in health care providers carriers is lower than in clinical samples. Therefore, MRSA screening in these persons is not often performed in Iran. The best program for the monitoring of MRSA spread and infection remains to debate formally. However, studies have consistently indicated that screening is advantageous in high-risk units, to discover the reservoir and to begin contact cautions. Therefore, management programs may be useful to decrease MRSA infection in health care providers (
29). Current studies reveal an important change of carrier rate, ranging from 0% to 29% (
23-
25,
30-
34).
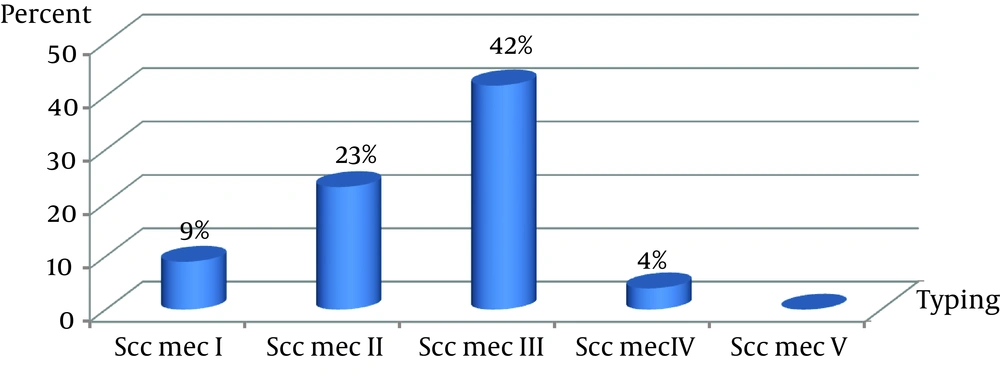
One of the most important molecular methods available for knowing the epidemiology and clonal strain relatedness of MRSA is SCCmec typing.
The SCCmec, a 21- to 67-kb mobile genetic element, is very important, because it harbors the methicillin resistance (mecA) gene and other antibiotic resistance markers that can transfer these characteristics to other bacteria, such as Enterococci. If MRSA propagates, it will become a major nosocomial pathogen worldwide.
Usually, SCC
mec types I‒IV are prevalent among healthcare providers’ isolates, whereas the type IV SCC
mec is the dominant element in community associated MRSA (CA-MRSA) (
7,
18). Although we detected SCC
mec types I‒IV, no SCC
mec type V was identified.
There is a significant relation between the SCC
mec types I, II and III and the hospital-acquired MRSA (HA-MRSA). However, the SCC
mec type IV in
S. aureus isolates has several different genetic backgrounds. It is smaller than the other SCC
mec types, because it does not carry any additional resistance genes, which may facilitate its mobility. The SCC
mec type IV carries functional recombinases and it has been found in multiple clones, even in the HA-MRSA, which is more mobile than the other types (
9,
35-
37).
As we determined the SCC
mec types of isolates, we identified the SCC
mec type IV element in our isolates. Usually, HA-MRSA strains carry SCC
mec types I, II, and III, while the type IV element is generally carried by CA-MRSA (
9,
37-
39). Eventually, strains with several types of SCC
mec have resulted from the genetic context, with additional virulence characteristics and the ability to spread (
9,
40).
With attention to control infection, isolates harboring this SCC
mec element should be treated as MRSA. Likewise, they are determined to be
mecA negative by PCR. Therefore, when isolates with unclarified resistance phenotypes are detected, they require further characterization and should be transferred to specialized laboratories, until updated routine assays are accessible (
39,
41).
Conventional methods for MRSA screening need to be reconsidered and only using phenotypic approaches for detection should be abandoned. Given the high rates of MRSA in health care providers in this study, detecting the carriers and decolonizing them, to reduce transmission of S. aureus in the hospital, is important. Annual screening of these persons, along with patients, is recommended.