1. Background
Hospital-acquired methicillin-resistant Staphylococcus aureus (HA-MRSA) was first isolated in hospitals in the United Kingdom in 1961 (1, 2). MRSA has since become widespread in hospitals (3, 4), and it now accounts for > 60% of S. aureus isolates from hospitals; moreover, it is one of the most prevalent agents causing nosocomial diseases in pediatric hospitals (5, 6). MRSA is increasingly resistant to typically used antibiotics, such as methicillin, dicloxacillin, oxacillin, nafcillin and vancomycin (7-10). Due to more widespread treatment with antibiotics and the increasing prevalence of antibiotic-resistant bacteria, use of bacteriophages and their endolysins could be an effective alternative in the treatment of infections. Bacteriophages (or phages) are natural bacterial viruses that attach specifically to their hosts and lyse them.
The first human phage therapy was reported in relation to the treatment of S. aureus skin infections. Phages have been reported to be effective in treating various bacterial diseases, such as skin infections caused by Staphylococcus, staphylococcal lung infections, neonatal sepsis, urinary tract infections, and diabetic foot as a complication of diabetes; bacteriophages have also been suggested as effective antibiofilm agents.
Treatment of MRSA using phages can be accomplished via local application for local infections, intraperitoneal administration for systemic infections, and the use of oral phage therapy to target MRSA in nurses who are carriers. Endolysins (or lysins) are produced by bacteriophages; they have been successfully used to control pathogenic antibiotic-resistant bacteria found on mucosal surfaces and in infected tissues with a low chance of bacterial resistance (11, 12).
Phages specifically recognize their hosts, and any region has specific phages; previous publications have demonstrated that any region has specific phages against MRSA and phages have specific hosts (13-15). Phages of S. aureus belong to the order Caudovirales and consist of double-stranded DNA located in the icosahedral capsid. According to the tail morphology, they can be classified into three families, namely Podoviridae, Siphoviridae, and Myoviridae (16). Isolations from wastewater have been carried out for phages against many bacteria, such as Escherichia coli, Pseudomonas aeruginosa, Acinetobacter sp., Klebsiella sp., Enterococcus faecalis, and Stenotrophomonas maltophilia, but phages against MRSA have not been isolated from wastewater at tertiary pediatric hospital (17-22).
Sewage is known to be rich sources of bacteriophages (23). Hospital sewage represents an extremely selective environment where high rates of resistant bacteria are evacuated in the natural environment or through animal fecal wastes; both hospital sewage and fecal wastes, can be environmental vectors for the horizontal transfer of antibiotic-resistant genes via transduction by bacteriophages (24, 25). Therefore, agents of antibiotic resistance are not only found in pathogenic bacteria but also in environmental organisms. More resistant bacteria are found in polluted habitats compared with unpolluted ones, indicating that environmental factors, such as sewage, have significantly contributed to the increased proportion of resistant bacteria occurring in the environment (26). The presence of phages in sewage is one element that can be used to justify the transfer of antibiotic resistance or virulence factors through transduction (27). Studies have demonstrated that prophages are integrated into the bacterial chromosomes and represent the most mobile genetic elements in S. aureus strains (28).
2. Objectives
Our objectives in this study were to characterize the structure and morphology of methicillin-resistant Staphylococcus aureus (MRSA) phages from a tertiary pediatric hospital.
3. Methods
3.1. Bacteriophage Isolation and Purification
Liquid samples of sewage at a volume of 200 cc were collected from the septic tank at the tertiary pediatric Bouali Sina hospital in Sari, Mazandaran province, northern Iran. The samples were transported to the laboratory and stored at 4°C. Ten milliliters of samples were centrifuged (11,100 × g, 5 minutes). Following this, the supernatant mixed with chloroform, centrifuged (11,100 × g, 5 minutes), and then filtered through a 0.22-μm filter. An equal volume of 2 X LB broths (QUELAB, United States) was added to the sample and cultured overnight with MRSA strains (29, 30) that were previously isolated from the blood of infant with septicemia at Buali Sina hospital.
3.2. Preparation of Bacterial Strains
Two isolated strains of MRSA were confirmed using conventional methods. The isolates were streaked for purity growth on a blood agar plate and incubated at 37˚C overnight. Microscopic examination of Gram stain smears from a Staphylococcus culture, as well as tube coagulase, catalase, DNase, and mannitol fermentation tests were performed on the specimens. For the detection of MRSA, the Kirby–Bauer disk diffusion technique was carried out using by methicillin and oxacillin antibiotic discs. MIC was carried out with E-test packages for vancomycin and linezolid (HiMedia, India) (31-33). The specimens were then incubated at 37°C and shaken overnight. The cultures were centrifuged at 4°C (10,000 × g; 10 minutes), and the supernatant was filtered using 0.22-μm (pore size) Millipore filters. To detect the presence of phages, the spot test was performed. The filtered phage lysate was titrated using the DLA technique (29, 30).
3.3. Determination of the Host Range
3.3.1. The Spot Test
An overnight culture of the potential indicator MRSA (100 μL) was mixed in a sterile tube with 3 mL of 0.4% soft agar; the contents were poured into petri dishes containing 15 mL of 1% bottom agar. The plate was placed on a bench until the soft agar had solidified, and then 5 μL of the sterile supernatant was poured over the solidified soft agar. After it had absorbed, the petri dishes were incubated upside up at 37°C overnight. The following day, the petri dishes were checked for zones of clearing (29, 30).
3.4. Double-Layer Plaque Assay (DLA Assay)
Phage stock and susceptible bacteria cell cultures were provided. Tenfold dilutions of the phage stock were added to 100 µl of bacterial suspensions. This solution was added to 3 mL of top agar and mixed gently, then poured into a petri dish containing 25 mL of bottom agar. The plates were incubated at 37°C overnight. The next day, the number of plaques was counted. Plaque-forming units (PFU) were calculated per milliliter by determining the number of plaques × 10 × the inverse of the dilution factor (29, 30).
3.5. Electron Microscope Observation of Phages
Bacteriophages were concentrated by centrifugation at 25,000 × g for 60 minutes using a high-speed centrifuge followed by two washes in 0.1 M neutral ammonium acetate. Purified bacteriophages were deposited on carbon-coated copper grids and stained with 2% uranyl acetate (pH = 4 - 4.5). After staining, phages were observed on a Philips CM 300 electron microscope at 150 kV (29, 30).
3.6. The Single-Step Growth Curve
Phage lysates were added to 100 μL of LB broth containing MRSA and preincubated for 10 minutes at 37°C for the adsorption of phages. After 10 minutes, this mixture was diluted in the LB broth medium (QUELAB, United States) and 100 µL of bacterial suspensions were added. Tubes were incubated from 10 minutes to 5 hours at 37°C. Samples were periodically removed from the mixture, and the number of free phages were counted using a plaque assay. The next day, the number of plaques was counted. PFU were calculated per milliliter as the number of plaques × 10 × the inverse of the dilution factor (29, 30).
4. Results
The results confirmed the isolation of MRSA from the blood of infants with septicemia.
4-1. Microscopic examination Gram stain smears showed a Gram-positive cluster of cocci.
4-2. Tests for catalase and coagulase were positive, exhibiting the formation of oxygen bubbles and clots, respectively (Table 1).
| Test | Result | |
|---|---|---|
| Gram staining | Gram-positive cocci | |
| Catalase test | Positive | |
| Coagulase test | Positive | |
| DNase test | Positive | |
| Mannitol fermentation test | Positive | |
| Oxacillin, 2 μg/mL | Methicillin, 5 μg/mL | |
| The Kirby–Bauer technique using the disk diffusion method | Resistant | Resistant |
| Determination of MIC with an Etest | Vancomycin | Linezolid |
| > 0.128 μg/mL | > 0.01 μg/mL |
4-3. The DNase test was found to be positive due to the formation of a transparent halo after HCL was added (Table 1).
4-4. The mannitol fermentation test was reported to be positive because the color of the culture was modified (Table 1).
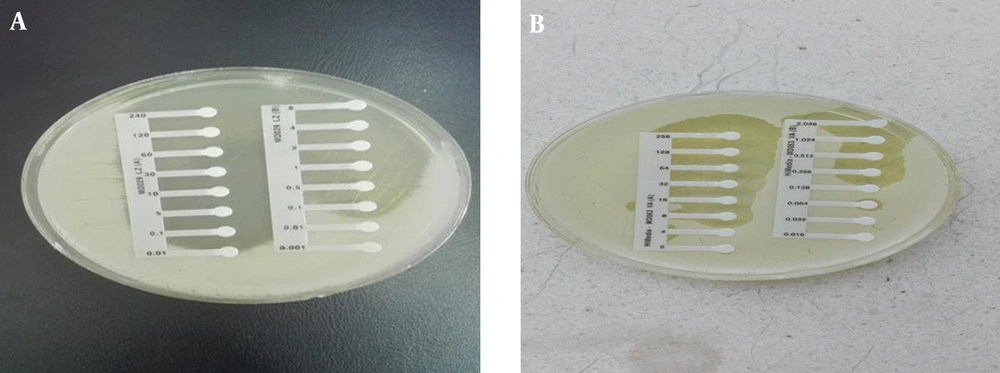
4-5. The Kirby-Bauer technique using the disk diffusion method with oxacillin (2 µg/mL) and methicillin (5 μg/mL) discs showed that the samples were quite resistant (Table 1; Figure 1).
4-6. The MIC was carried out with Etest packages for vancomycin and linezolid, and the results were > 0.128 μg/mL and > 0.01 μg/mL, respectively (Table 1; Figure 2).
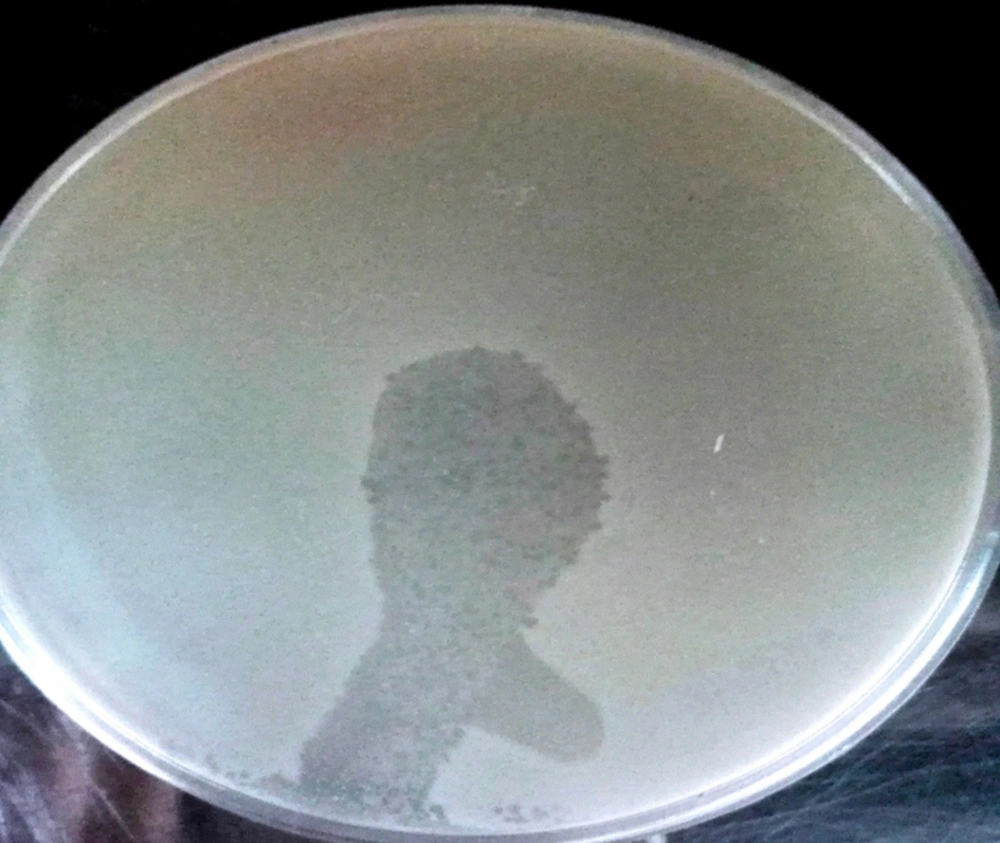
4-7. In the spot test, clear plaques observed on LB agar plates were covered with a bacterial lawn of MRSA; this demonstrated that the phages were isolated and had lytic activity (Figure 3).
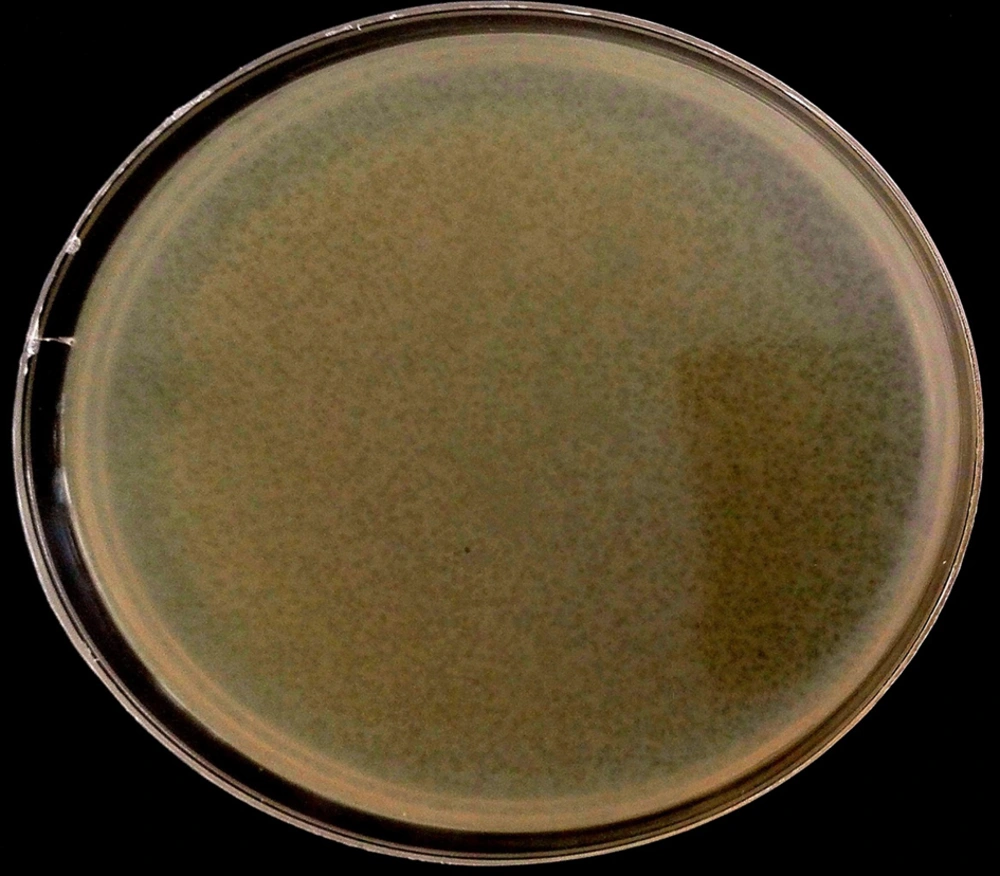
4-8. In the DLA technique, plaques of 9 - 14 μm in diameter formed, and the titer-isolated and purified phages were 3 × 109 PFU/mL-1 (Figure 4).
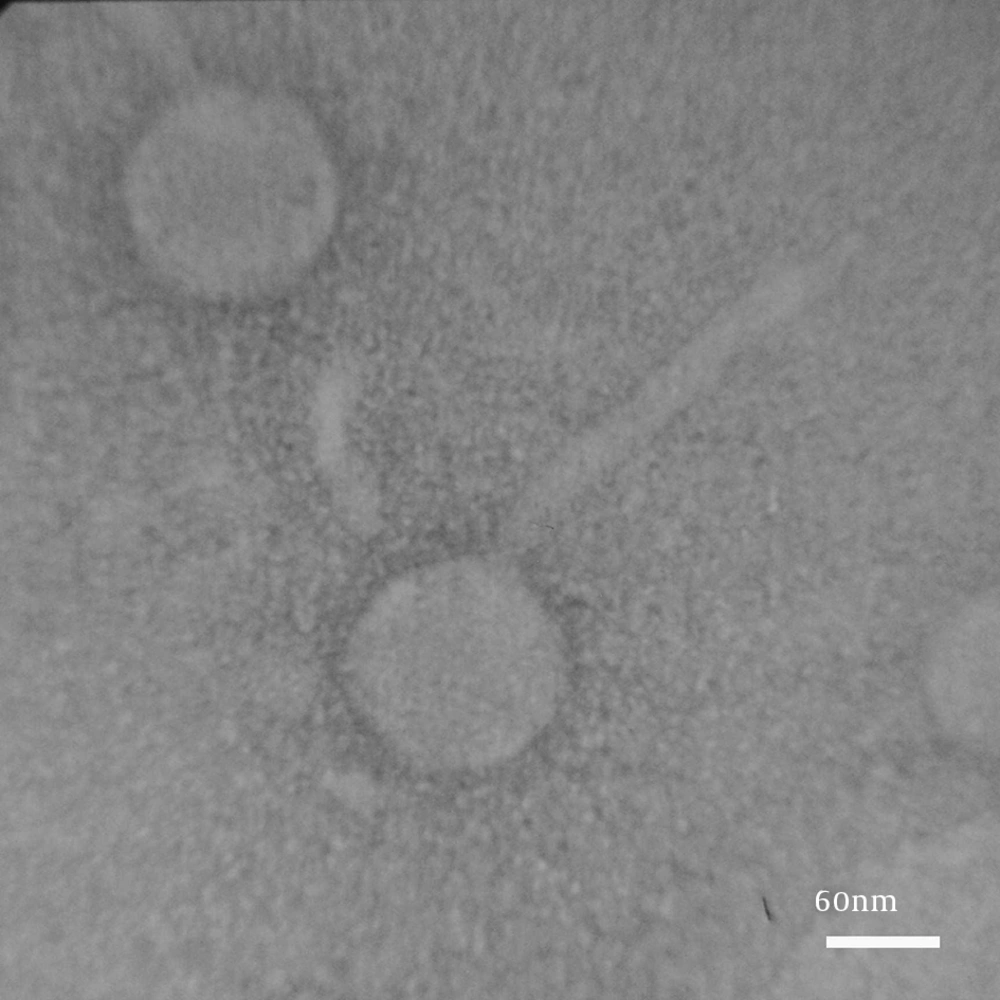
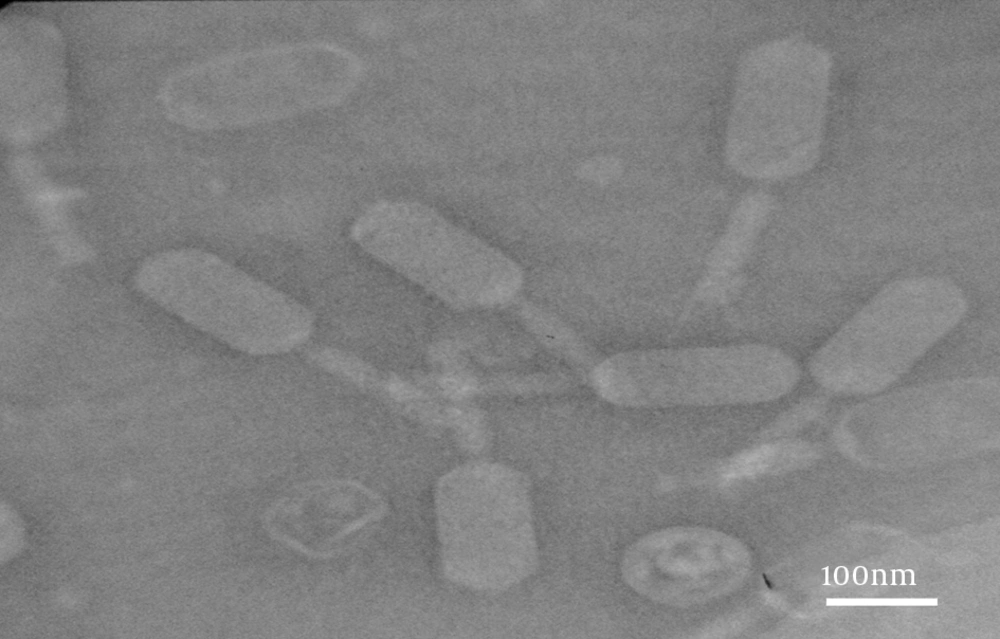
4-8. The result of electron microscopy showed that two phages were isolated. Group (I) had an icosahedral head and a long, noncontractile tail (400 nm; Figure 5). Group (II) had a distinct prolate head and a long, noncontractile tail (300 nm; Figure 6). Phages in Group (I) belonged to the family Siphoviridae (order Caudovirales), serogroup F, while phages in Group (II) belonged to the family Siphoviridae (order Caudovirales), serogroup A.
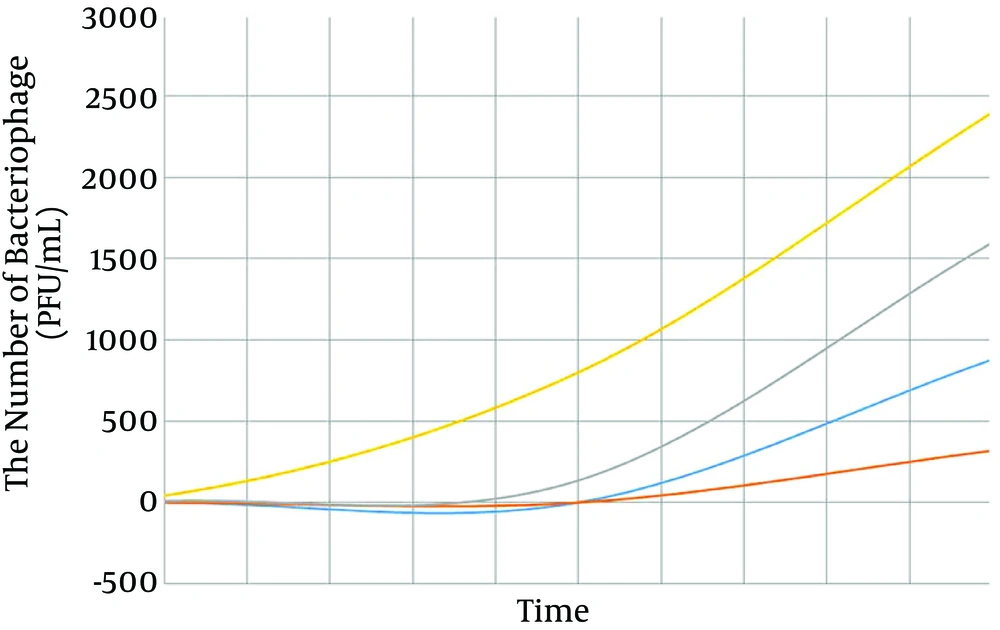
4-9. The single-step growth curve showed that the latent period of isolated bacteriophages was 70 minutes and the burst size was 2,400 PFU/infected host (Figure 7).
5. Discussion
In the current study, two phages against MRSA belonging to the family Siphoviridae were isolated from liquid sewage samples from a tertiary pediatric hospital. In previous studies, phages against MRSA have been isolated from different sources, such as cow’s milk, sewage/pond water, soil samples collected from poultry, and fecal samples from livestock farms; however, such phages have not been isolated from hospital wastewater (14, 16, 17, 34-36).
Isolation of phages is based on different sources; for example, in a study by Wommack et al. (2000) (37), phages were isolated from water samples from the TFF filtration apparatus used for the concentration of viruses from water samples. In a study by Dane et al. (2002) (38), phages from soil samples were extracted by centrifuging, sonication, and filtering the supernatant.
Phage isolation may not be an accessible or feasible method in the reference laboratory because such testing is sensitive and many variables can affect the results; for example, in our study, sewage samples were taken from different locations in the hospital, such as the aeration basin and vacuum pond, and those phages were not isolated. On the other hand, phages were isolated from sewage samples taken from the septic tank, and this process took a lot of time.
In Synnott et al.’s (2009) (9) study, phages against MRSA were isolated in liquefied sewage samples. Moreover, in a study by Chang et al. (2015) (35), phages against MRSA from skin were isolated; the results of electron microscopy showed that isolated phages belonged to the family Siphoviridae, serogroup B, and had a latent period time of 20 minutes. In a study by Kwiatek et al. (2011) (17), virulent bacteriophages (MSA6) were isolated from a cow with mastitis. The electron microscopy result of showed that the isolated phages belonged the family Myoviridae; they had a short latent period (15 minutes) and a relatively small burst size (23 PFU/cell). The isolated phages were capable infecting a wide spectrum of staphylococcal strains of both human and bovine origin. Moreover, Gupta et al. (2010) (14) used sewage/pond water samples and isolated three lytic phages (P-15/HP, P-27/HP and P-40/HP) against S. aureus host strains; phage P-27/HP showed a broad host spectrum that lysed to 17 S. aureus and coagulase-negative staphylococcus (CNS) isolates. In our study, phages isolated from hospital sewage belonged to the family Siphoviridae, serogroups A and F, with a latent period of 70 minutes; these phages exhibited lytic activity against coagulase-positive staphylococci.
In the current study, the lytic activity of phages was demonstrated through a spot test, and through DLA assay, the titer of phages was determined to be 3 × 109 PFU/mL, with formed plaques of 9 - 14 μm in diameter. In contrast, in research by Li et al. (2014) and Chhibber et al. (2013) (39, 40), phages were isolated from sewage samples and classified as members of the Myoviridae family; these produced clear plaques of a size of 2 - 3 mm in diameter with the titer of phages of more than 109 PFU/mL.
Antibiotic resistance, virulence factors, and toxins in bacteria strains are encoded by mobile genetic elements, such as prophages. These elements can be horizontally transferred by transduction. The presence of phages in hospital sewage may be one of the reasons for antibiotic resistance or virulence factors, as transduction may result in a nonvirulent strain of staphylococcus turning into a virulent one (41). Other research is still necessary for the full molecular characterization of bacteriophages, but some topics had to be neglected in the present research due to a lack of financial support (16-23). For instance, this study did not deal with the relation between antibiotic resistance in MRSA and the presence of phages, and this must be left for future work.
In this study, phages against MRSA belonging to the family Siphoviridae (order Caudovirales), serogroups A and F, were isolated from liquid sewage samples from a tertiary pediatric hospital. The presence of these phages may be one reason for transfer of antibiotic resistance or virulence factors through transduction in MRSA. Air-born phages can be transferred to the wards; thus, creating negative pressure in the sewage pipe at hospitals can prevent phages from entering wards via aerosols.