1. Background
Urinary tract infections (UTIs) are one of the most common infectious diseases, affecting a large number of patients worldwide. UTIs increase the risk of morbidity and mortality among patients and impose great health costs on the affected patients and the community (1, 2). The incidence of UTIs varies with respect to age, sex, genetics, and underlying diseases (3).
Risk factors associated with UTIs are categorized into host factors, host behaviors, and bacterial characteristics, which expose the host to potential uropathogens, enhance colonization, or make the host respond to colonization (4). Colonized bacteria in the bowel and vaginal cavity can be easily transferred to women’s urinary tract, given the short distance, making sexually active women more susceptible to UTIs (4).
Escherichia coli (E. coli), a Gram-negative bacterium consisting of various subtypes, is the most common uropathogen causing UTIs (namely uropathogenic E. coli) (5-7). Therefore, treatment of UTIs is dependent on the administration of appropriate antibiotics. However, high resistance of E. coli to antibiotics has become a major treatment problem (8). Studies have reported the resistance of E. coli to various antibiotics, including β-lactam, cephalosporins, gentamicin, and fluoroquinolones (9, 10).
The high prevalence of resistance to trimethoprim-sulfamethoxazole (TMP/SMX) has led to the administration of fluoroquinolones, which are highly effective and convenient for persistent and recurrent UTIs caused by E. coli (11). However, previous review studies have reported an increasing trend in resistance to ciprofloxacin in recent years (12).
The mechanisms underlying this resistance in E. coli have been investigated in the literature. In 1990, Kato et al. first introduced topoisomerase IV, a homolog of DNA gyrase, composing of two parts, ie, parE and parC in E. coli (13).
Other studies later posited that quinolones stabilize the complex between DNA and DNA gyrase or topoisomerase IV through formation of reversible drug-enzyme-DNA complexes and inhibiting the progression of polymerase and DNA replications (14, 15). Therefore, mutations in parE and parC genes in E. coli strains could be involved in resistance to quinolones (16, 17). In addition, qnrB gene in interaction with E. coli DNA gyrase has been proposed as plasmid–encoded resistance to quinolones (18-21).
In general, better identification of the associated genes can help researchers and clinicians improve the treatment of various infections, including UTIs, with ciprofloxacin of fluoroquinolones. With this background in mind, we aimed to determine the role of parC, parE, and qnrB genes in ciprofloxacin–resistant E. coli isolates from urine samples of patients suffering from UTI.
2. Methods
2.1. Sample Size
The sample size was calculated, based on the frequency of E. coli isolates for UTIs, using the following formula: N= (1.96)2 (0.7) (0.3)/(0.1)2 = 80.6 ~ 80
2.2. Study Design and Bacterial Isolation
A total of 80 E. coli isolates from urine samples were collected from outpatients and inpatients, who were referred to Imam Khomeini Hospital for presumptive UTI during May-October 2014. All 124 urine samples were collected by midstream clean-catch method in sterile containers. Bacterial isolation was performed, based on standard bacteriological tests, such as catalase and oxidase tests, using MacConkey agar, triple sugar iron agar, and IMViC tests.
Patients were divided into adult (above 18 years) and young (below 17.9 years) groups. Also, the sex of the patients and the hospital wards were recorded. All E. coli isolates were maintained in tris-buffered saline culture, containing 15% glycerol at -70°C until they were transferred to the Research Laboratory of Shahid Beheshti University of Medical Sciences, Tehran, Iran for further analysis.
2.3. Antibiotic Sensitivity Test (AST)
The resistance/sensitivity of the isolates to 20 common antibiotics was investigated by standard disk diffusion technique (Kirby-Bauer method), according to the clinical and laboratory standards institute (CLSI) 2014 guidelines; all disks were provided from Rosco Company (Denmark). The antibiotics used for AST included ceftriaxone, piperacillin, amikacin, gentamicin, ciprofloxacin, amoxicillin, cefazolin, aztreonam, cefepime, trimethoprim/sulfamethoxazole, nitrofurantoin, chloramphenicol, imipenem, meropenem, ertapenem, ampicillin, ofloxacin, norfloxacin, tetracycline, and cefixime.
A bacterial lawn was prepared on the Mueller-Hinton agar, using a sterile cotton swab continuously. For this purpose, bacterial suspensions were prepared with turbidity equal to 0.5 MacFarland (1.5 × 108 CFU/mL). Afterwards, antibiotic disks were placed on the plate by sterile forceps. The plates were incubated at 37°C for 18 - 24 hours. Then, the diameter of the inhibition zone around the disks was measured by a millimeter ruler. The results were compared to the standard table and were reported as sensitive, intermediate, or resistant.
2.4. Minimum Inhibitory Concentration (MIC) of Ciprofloxacin by E-Test Method
Bacteria were considered resistant to the drug when there was no inhibition zone around them. The ciprofloxacin–resistant isolates in AST were assessed in terms of MIC by E-test strip method (Liofichem, Denmark). According to CLSI 2014 guidelines, MIC ≤ 1 was considered sensitive, MIC ≥ 4 was regarded as resistant, and MIC = 2 was considered intermediate.
2.5. DNA Extraction, PCR Method, and Sequencing
Chromosomal DNA of the bacteria was extracted by boiling method for further PCR of chromosomal parC and parE genes. The GeneJET Plasmid Miniprep kit (Thermo Scientific, Lithuania) was used to extract plasmid for further PCR analysis of qnrB gene. The Master Mix (Thermo Scientific, Lithuania) was used for the PCR mixture. Also, distilled water and E. coli ATCC 25922 were used as the negative and positive controls, respectively. The list of primers, PCR programs, and PCR products is mentioned in Tables 1 and 2.
| Genes | Primer Sequences | PCR Products, bp |
|---|---|---|
| parE | F5’TACCGAGCTGTTCCTTGTGG | 265 |
| R5’GGCAATGTGCAGACCATCAG (83) | ||
| qnrB | F 5'GGMATHGAAATTCGCCACTG | 268 |
| R 5' TTTGCYGYYCGCCAGTCGAA | ||
| M = A or C; H = A or C or T; Y = C or T (84) | ||
| parC | F5’CTGAATGCCAGCGCCAAATT | 389 |
| R5’TGCGGTGGAATATCGGTCGC (85) |
The Sequence of Primers Used in the Present Study
| Stages | Temperature, °C | Time (for parC and parE) | Time for qnrB Gene, min |
|---|---|---|---|
| Primary denaturation | 95 | 5 min | 10 |
| Denaturation | 95 | 40 s | 1 |
| Primer coupling | 56.2 | 40 s | 1 |
| Polymerization | 72 | 40 s | 1 |
| Final polymerization | 72 | 5 min | 10 |
The PCR Programs (35 Cycles)
PCR products of the studied genes were determined after electrophoresis on 1.5% agarose gel and visualized under UV radiation for gel documentation. Further sequencing was performed by Bioneer Company (Korea). The nucleotide sequences were analyzed with the Chromas 1.45 software and BLAST in NCBI. Afterwards, the genes were submitted to GenBank.
2.6. Statistical Analysis
In this descriptive-application study, the results are presented as frequency (percentage) for categorical variables with 95% confidence intervals (95% CI). For the statistical analysis, SPSS version 21.0 (SPSS Inc., Chicago, IL) was used. P value ≤ 0.05 was considered statistically significant.
3. Results
Among 80 E. coli strains, isolated from 124 urine samples (64.5%), 79% were from adults and 21% were from children. In terms of sex, 51% of the subjects were female, 28% were male, 13% were girls, and 8% were boys. Most of the urine samples were collected from inpatients: 35% from the gynecology ward, 15% from the urology ward, 14% from the neurology ward, and 10% from the miscellaneous wards. Also, 25% of the samples were collected from the outpatients.
The resistance level of E. coli to the studied antibiotics is demonstrated in Table 3. As shown, there was a high rate of resistance to piperacillin, ampicillin (85% and 83.8%, respectively), TMP/SMX, ciprofloxacin, and tetracycline (78.7%, 77.5%, and 75%, respectively). Also, 100% sensitivity to ertapenem, meropenem, and imipenem and 98.7% sensitivity to nitrofurantoin were detected.
| Antibiotics | Resistant | Intermediate | Sensitive |
|---|---|---|---|
| Piperacillin, 100 µg | 85 | 2.5 | 12.5 |
| Ampicillin, 10 µg | 16.2 | 27.5 | 56.3 |
| Amoxicillin-clavulanic, 20/10 µg | 77 | - | 23 |
| Cefazolin, 30 µg | 73.7 | 2.5 | 23.8 |
| Norfloxacin, 10 µg | 71.2 | 0 | 28.8 |
| Ceftriaxone, 30 µg | 72.5 | 1.3 | 26.2 |
| Cefepime, 30 µg | 42.5 | 21.3 | 36.2 |
| Gentamicin, 10 µg | 62.5 | 25 | 35 |
| Amikacin, 30 µg | 11.4 | 5.1 | 83.5 |
| Trimethoprim/sulfamethoxazole (SXT), 1.25/23.75 µg | 78.7 | 0 | 21.3 |
| Ciprofloxacin, 5 µg | 77.5 | 3.8 | 18.7 |
| Aztreonam, 30 µg | 68.7 | 3.8 | 27.5 |
| Nitrofurantoin, 300 µg | 1.3 | 0 | 98.7 |
| Ertapenem, 10 µg | 0 | 0 | 100 |
| Meropenem, 10 µg | 0 | 0 | 100 |
| Imipenem, 10 µg | 0 | 0 | 100 |
| Cefixime, 5 µg | 12.5 | 6.2 | 81.3 |
| Chloramphenicol, 30 µg | 22.5 | 5 | 72.5 |
| Tetracycline, 30 µg | 75 | 0 | 25 |
| Ofloxacin, 5 µg | 67.4 | 0 | 32.6 |
| Ampicillin, 10 µg | 83.8 | 6.2 | 10 |
The Frequency of E. coli Resistance to the Studied Antibioticsa
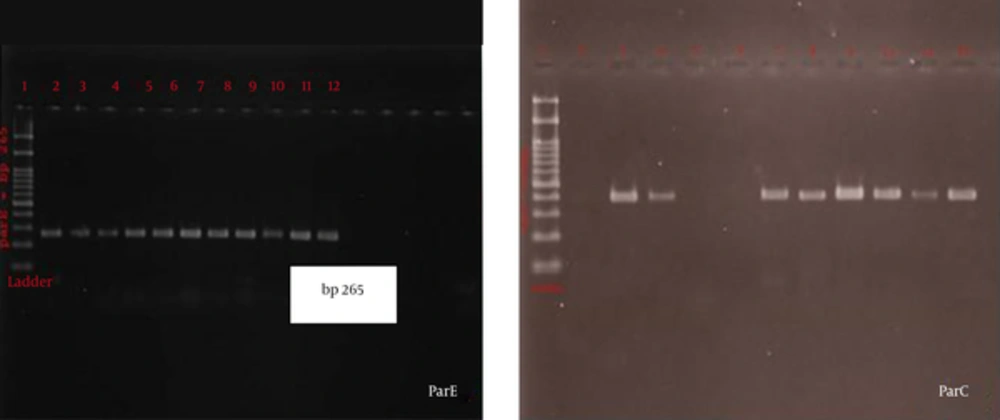
The intact genes of parC and parE were detected in 92.5% and 91.3% of the samples, respectively; the frequency of qnrB gene was negative in all the isolates. The size of PCR bands was 265 bp for parE, 389 bp for parC, and 268 bp for qnrB genes, as demonstrated in Figure 1. Comparison of the sequence of the extracted proteins with the protein sequence of ATCC 25922 is demonstrated in Figures 2 and 3.
In parE, from left to right: lane 1, ladder 100 bp; lane 2, positive control; and lane 3, negative control. Lanes 3-12 were positive for a band of 265 bp. In parC, from left to right: lane1, 100 bp ladder; lane 2, negative control; and lane 3, positive control. The rest of the samples were positive and had a band of 365 bp.
As observed, there was a change in the nucleotide sequence of ParE at 1316 (C to A), 1321 (A to T), 1360 (G to C), 1381 (G to C), and 1480 (C to T) and at 461 (V to L), 466 (V to G), 467 (A to S), 469 (Q to H), and 484 (K to L) of the protein nucleotide. Further submission of parC and parE genes in NCBI GenBank was performed with accession numbers, KT454384.1 and KT454385.1, respectively.
4. Discussion
This study was performed on 80 E. coli isolates from inpatients and outpatients with UTIs, who were referred to Imam Khomeini Hospital, Tehran, Iran during May-October 2014. Based on the AST results, 77.5% of the isolates were resistant to ciprofloxacin. In a study by Karlowsky et al. investigating a large number of urine samples, ciprofloxacin–resistant E. coli was reported in 10.8% of the samples (22). Also, a random investigation of 670 centers in the United States showed resistance to ciprofloxacin in less than 10% of the centers during 5 years (23).
Review studies have reported an increasing trend in fluoroquinolone-resistant E. coli (59% to 95%) (12). The resistance level in the present study was significantly higher than the mentioned reports, which indicates the high resistance level in the studied center. It can be concluded that ciprofloxacin is administered more frequently in Iran, compared to other countries.
According to a European study, the overall E. coli resistance to cephalosporin, nitrofurantoin, and gentamicin was less than 2%, while higher resistance levels for ampicillin, TMP/SMX, amoxicillin/clavulanic acid, and ciprofloxacin (0.5% - 7.6%) were reported with an increasing trend in resistance to quinolones and trimethoprim from 1999 - 2000 to 2007 - 2008 (9).
Similarly, in the present study, there was a high resistance level to ampicillin, TMP/SMX, and ciprofloxacin, whereas a low resistance level to nitrofurantoin was reported (although the rates were different). The discrepancy between studies performed in different countries can indicate that the prevalence of wide–spectrum antibiotic administration is more than needed in our country. Also, the increasing trend, suggested by the abovementioned studies, signifies that more caution should be taken with respect to the administration of the remaining sensitive antibiotics.
Khodadoost et al. from Iran reported 81.43% and 62.13% resistance rates to ampicillin and co-trimoxazole, respectively (24), which is close to the results of the present study. Mohajeri et al. also reported the resistance rate of E. coli isolates to ciprofloxacin as 43% (25), which was lower than the resistance rate reported in the present study (77.5%). This could be attributed to the increasing emergence of resistant bacteria to wide-spectrum antibiotics.
Moreover, Mohajeri et al. introduced piperacillin and ampicillin as resistant antibiotics and imipenem and nitrofurantoin as sensitive drugs (25). This finding was in line with the results of the present study; however, minor differences in the resistance level to other antibiotics were observed. The increasing rate of resistance to such antibiotics is a warning for physicians regarding the indiscriminate administration of antibiotics, especially in Iran.
In a review article by Dallhoff, with reference to a study by Sahm et al. a 3.7% resistance rate to ciprofloxacin was reported in UTI isolates of E. coli after 13 years of administration in the United States; this rate was twice higher in men than women and increased with age; they also reported a higher prevalence among inpatients (26). The mentioned study by Dallhoff was in line with the study by Karlowskey et al. from USA, which declared that the resistance of E. coli isolates to some antibiotics including ciprofloxacin is increasing. Also, ciprofloxacin was the only agent with a consistent increase in resistance from 0.7% to 2.5% during 1995 - 2001 (26).
Female dominance has been similarly reported in other studies (22). In the current research, women comprised 51% of adult patients and girls comprised 13% of the pediatric group of patients. The prevalence of E. coli in UTI in the present study was similar to previous research, reporting E. coli as the most prevalent pathogen causing UTIs (5-7, 27). In the present study, the frequency of the studied intact genes among ciprofloxacin–resistant isolates by PCR was as follows: 90.9% for parC, 97.67% for parE (intact chromosomal genes), and 0% for qnrB.
Linndgren et al. reported four mutations in parC (S80I, S80R, E84K, and E84G) and parE (I444F, S458T, D475E, and I529L) and detected mutations in parC among 83% of the resistant isolates. Also, none of the susceptible isolates showed mutations in parC gene. In addition, they postulated a significant genetic jump leading to a move from susceptibility to resistance (28). Various studies have similarly confirmed parC and parE mutations in quinolone-resistant bacterial isolates (29, 30).
In congruence with the present study, Warburg et al. identified a strong association between aac(6′)-Ib-cr gene and ciprofloxacin resistance, defined by the CLSI criteria, and found no qnrB genes (31). They also showed that the interaction between resistance to β‐lactamase and quinolones may result from the rise in the prescription of quinolones. Cattoir et al. also postulated that qnrB mutations do not play a role in resistance to β‐lactamase (32).
However, some other studies introduced the role of qnrB mutations in quinolone-resistance (33-35). Also, some studies detected an interaction between qnrB and topoisomerase IV and reported an increase in the MIC of ciprofloxacin in the presence of qnrB (20, 21, 36). This finding is inconsistent with the present results and demonstrated that the role of qnrB gene in ciprofloxacin resistance is low. Our previous study showed that 39% of E. coli isolates from UTIs included the qnrA gene. Also, coexistence of qnrA gene in extended-spectrum ß-lactamase-positive E. coli isolates was detected (37).
The current study had some limitations. We only investigated the patients referring to one center for sample collection, which limited the sample size of the study. Due to the absence of qnrB gene in ciprofloxacin–resistant E. coli isolates, we investigated other similar genes and found qnrA in 39% and aac(6′)-Ib-cr in 72% of resistant isolates (some of them are not published yet). Therefore, it is suggested that further studies consider the possible role of proton pump inhibitors in quinolone-resistance and investigate other wide-spectrum antibiotics.
In conclusion, the current study indicated that parC and parE gene mutations may play a more significant role in ciprofloxacin resistance in E. coli isolates, compared to other genes such as qnrB mutations. Also, it can be stated that AST must be performed according to CLSI protocols before prescribing any antibiotics in order to prevent resistance in some antibiotics to which E. coli is still sensitive.