1. Background
2. Objectives
3. Methods
3.1. Isolation and Identification of Staphylococcus aureus
3.2. Antibiotic Susceptibility Testing
3.3. Agar Screening Methods
3.4. DNA Extraction
3.5. Identification of nuc, mecA and pvl genes by the Polymerase Chain Reaction
| Primer | Sequence (5′→3′) | Amplicon Size, bp | Target Gene | References |
|---|---|---|---|---|
| nuc/F | GCGATTGATGGTGATACGGTT | 267 | nuc | 24 |
| nuc/R | AGCCAAGCCTTGACGAACTAAAGC | |||
| mecA/F | GTAGAAATGACTGAACGTCCGATAA | 310 | mecA | 7 |
| mecA/R | CCAATTCCACATTGTTTCGGTCTAA | |||
| pvl/F | ATCATTAGGTAAAATGTCTGGACATGATCCA | 433 | pvl | 12 |
| pvl/R | GCATCAASTGTATTGGATAGCAAAAGC | |||
| β | ATTGCCTTGATAATAGCCYTCT | 937 | SCCmec type II, IV | 7.26 |
| 3α | TAAAGGCATCAATGCACAAACACT | |||
| ccrC/F | CGTCTATTACAAGATGTTAAGGATAAT | 518 | SCCmec type III, V | 7.26 |
| ccrC/R | CCTTTATAGACTGGATTATTCAAAATAT | |||
| 1272F1 | GCCACTCATAACATATGGAA | 415 | SCCmec type I, IV | 7.26 |
| 1272R1 | CATCCGAGTGAAACCCAAA | |||
| 5RmecA | TATACCAAACCCGACAACTAC | 359 | SCCmec type V | 7.26 |
| 5R431 | CGGCTACAGTGATAACATCC | |||
| IVa-F | GCCTTATTCGAAGAAACCG | 776 | SCCmec IVa | 27 |
| IVa-R | CTACTCTTCTGAAAAGCGTCG | |||
| IVb-F | TCTGGAATTACTTCAGCTGC | 493 | SCCmec IVb | 27 |
| IVb-R | AAACAATATTGCTCTCCCTC | |||
| IVc-F | ACAATATTTGTATTATCGGAGAGC | 200 | SCCmec IVc | 27 |
| IVc-R | TTGGTATGAGGTATTGCTGG | |||
| IVd-F5 | CTCAAAATACGGACCCCAATACA | 881 | SCCmec IVd | 27 |
| IVd-R6 | TGCTCCAGTAATTGCTAAAG | |||
| (GTG) | GTGGTGGTGGTGGTG | - | Rep | 18 |
Abbreviations: F, forward; R, reverse.
3.6. Methicillin Resistant Staphylococcus aureus Typing
3.6.1. Amplification of SCCmec by Multiplex PCR
3.6.2. Methicillin Resistant Staphylococcus aureus Typing by Rep-Polymerase Chain Reaction
3.7. Control Strains Used in this Study
3.8. Statistical Analyses
4. Results
4.1. Bacterial Isolation and Confirmation
| Type of Specimen | No. of S. aureus Isolates (%) | No. of MRSA Isolates (%)b | MDR (%) in MRSA Isolates |
|---|---|---|---|
| Wound | 80 (37.2) | 40 (50) | 36 (90) |
| Blood | 38 (16.7) | 20 (52.63) | 19 (95) |
| Urine | 30 (14) | 8 (26.67) | 6 (75) |
| Abscess | 20 (9.3) | 4 (20) | 2 (50) |
| Sputum | 13 (6) | 8 (61.54) | 8 (100) |
| CSF | 4 (1.9) | 3 (75) | 3 (100) |
| Other body fluids | 30 (14) | 4 (13.34) | 3 (75) |
| Total | 215 (100) | 87 (40.5) | 77 (88.50) |
Abbreviation: MRSA, methicillin-resistant Staphylococcus aureus.
aMulti-drug resistant (isolates which were resistant to 3 and more antimicrobial agents, with 88.50% of the MRSA isolates being MDR).
bThe rate of MRSA isolates among 215 Staphylococcus aureus isolates was 40.5%.
4.2. Phenotypic Tests
4.2.1. Antimicrobial Resistance
| Resistance Patterns | No. (Identity of Isolates) | Antibiotic Resistance Patterns | MDRa or Non-MDR |
|---|---|---|---|
| R1 | 4 (14, 75, 111, 119) | P, OX, FOX | Non-MDR |
| R2 | 2 (132, 135) | P, OX, FOX, GM, CC, SXT | MDR |
| R3 | 39 (2 - 5, 8, 15, 25, 37, 41, 42, 44, 47, 61-63, 91, 98, 107, 114, 117, 118, 123, 124, 136, 140, 151, 152, 154, 156-158, 160, 162, 173, 175, 176-178, 209) | P, OX, FOX, GM, CC, E, CP, SXT | MDR |
| R4 | 26 (10, 11, 13, 16, 19, 22, 28, 40, 48, 57, 76, 83, 96, 97, 105, 153, 155, 159, 161, 174, 181, 182, 201-203, 206) | P, OX, FOX, GM, CC, E, CP, R | MDR |
| R5 | 6 (60, 68, 74, 78, 125, 179) | P, OX, FOX, GM, CC, E, CP, SXT, R | MDR |
| R6 | 1 (192) | P, OX, FOX, CC, E, CP, R | MDR |
| R7 | 1 (7) | P, FOX, CC, E, CP, SXT | MDR |
| R8 | 2 (20, 147) | P, FOX, CC, E | MDR |
| R9 | 3 (109, 115, 150) | P, FOX | Non-MDR |
| R10 | 3 (39, 50, 51) | P | Non-MDR |
Abbreviation: CC, clindamycin; CP, ciprofloxacin; FOX, cefoxitin; GM, gentamicin; E, erythromycin; OX, oxacillin; P, penicillin; R, rifampin; SXT, trimethoprim-sulfamethoxazole.
aMultidrug-resistant (isolates which were resistant to 3 and more antimicrobial agents, with 88.50% of the MRSA isolates being MDR).
4.2.2. Agar Screening Methods
4.2.3. Minimum Inhibitory Concentration (by E-Test)
4.3. Methicillin Resistant Staphylococcus aureus Detection
4.4. Molecular Tests
4.4.1. Identification of nuc, mecA and pvl Genes
4.4.2. Methicillin Resistant Staphylococcus aureus Typing
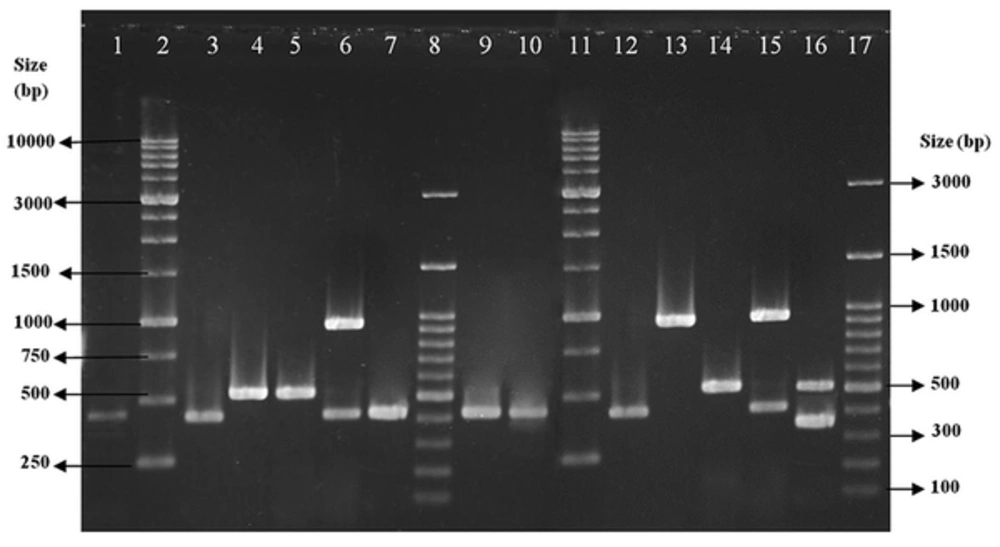
4.4.2.1. SCCmec Typing
Lanes 1, 3, 7, 9 and 10, clinical isolates with SCCmec type I; lanes 2 and 11, size markers (1 kb DNA Ladder); lanes 4 and 5, clinical isolates with SCCmec type III; lane 6, clinical isolate with SCCmec type IV; lanes 8 and 17, size markers (100 bp DNA ladder Plus); and lanes 12 - 16, control strains (types I - V).