1. Background
Sepsis is common among critically ill patients and is associated with significant mortality and morbidity (1, 2). Despite advances in clinical care and outcomes, existing epidemiologic studies suggest that sepsis remains a major health problem worldwide (3, 4). In 2017, more than 11 million sepsis-related deaths were reported, representing 19.7% of all global deaths (5). Sepsis is treatable, and infection-prevention efforts can reduce sepsis incidence and improve outcomes (6-8). Recently, it has been found that vitamin D supplements can improve prognosis in patients with sepsis (9, 10). Vitamin D plays an essential role in both the adaptive and innate immune systems for the ideal function of antimicrobial activity (11-13). Thus, it is a contributing factor to decreased sepsis incidence and severity (13). However, patients with severe infections, such as those withsepsis, usually have a high rate of vitamin D insufficiency (14-16). Furthermore, several studies on patients with sepsis have shown strong associations between decreased vitamin D levels and adverse outcomes (i.e. increased length of stay, acute kidney injury, and mortality) (17-19). Although the role of vitamin D in sepsis has been previously suggested, the association of vitamin D with increased morbidity and mortality has not yet been established (20-22). In Iran, a high proportion of vitamin D deficiency was observed in the general population (23). There is also an increasing number of patients with sepsis in healthcare settings (24).
2. Objectives
This study aimed to find the relationship between vitamin D levels and outcomes among patients with sepsis in Tehran, Iran.
3. Method
3.1. Settings and Patients
This cross-sectional study was conducted at a university hospital (Imam Hossein Hospital) in Tehran, Iran, from 1 February 2018 to 5 March 2019. Patients greater than 18 years old were included if they met the following criteria for sepsis: (1) Confirmed or suspected source of infection determined by the treating clinician and (2) having two or more criteria for the systemic inflammatory response syndrome (SIRS) (25). Patients were excluded if they experienced shock due to reasons other than sepsis, had neuroendocrine diseases, were in cardiac arrest on arrival, suffered from mental disorders, were under constant use of vitamin D, or were pregnant. Patient data, including demographics, comorbidities, and laboratories such as cultures and the suspected source of infection, were obtained through enrollment. The institutional human subjects committee approved the current study (IR.SBMU.MSP.REC.1397.126), and informed consent was obtained from the patients.
3.2. Vitamin D Status Measurements
Whole blood collected was placed into the tubes containing ethylenediaminetetraacetic acid as an anticoagulant. Within 1 hour of collection, each sample was centrifuged at 1500 rpm for 10 minutes. The plasma was directly aliquoted and stored at -20°C. After completion of samples, the concentration of serum 25-hydroxy vitamin D3 was measured by enzyme-linked immunosorbent assay (ELISA) (Immunoassays S.A., Belgium) according to the manufacturer's protocol. Vitamin D deficiency was defined as baseline serum 25-hydroxyvitamin D (25OHD) levels < 30 ng/mL (26).
3.3. Statistical Analysis
SPSS version 20.0 (IBM Corp., Armonk, NY, USA) was used for data analysis. Categorical and continuous variables were analyzed using the chi-square test, t-test, and analysis of variance (ANOVA). P-values less than 0.05 were considered statistically significant.
4. Results
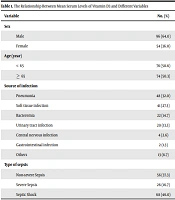
A total of 150 patients with a mean age of 70.8 ± 13.3 years were studied (males = 96 (64.0%) and females = 54 (36.0%)). In the studied patients, the mean serum level of vitamin D3 was 19.03 ± 13.08 ng/ml. If the serum levels of vitamin D3 were < 30 ng/mL, vitamin D deficiency was confirmed. Thus, the prevalence of vitamin D insufficiency in critically ill subjects with sepsis was 100% (150/150). Table 1 indicates the relationship between different variables and the mean serum level of vitamin D. Only sex (P = 0.01) showed a significant association with the mean serum level of vitamin D.
| Variable | No. (%) | Vitamin D3 | P-Value |
|---|---|---|---|
| Sex | 0.01 | ||
| Male | 96 (64.0) | 18 | |
| Female | 54 (36.0) | 19 | |
| Age (year) | 0.12 | ||
| < 65 | 76 (50.6) | 14 | |
| ≥ 65 | 74 (50.3) | 20 | |
| Source of infection | 0.14 | ||
| Pneumonia | 48 (32.0) | 20 | |
| Soft tissue infection | 41 (27.3) | 14 | |
| Bacteremia | 22 (14.7) | 16 | |
| Urinary tract infection | 20 (13.3) | 11 | |
| Central nervous infection | 4 (2.6) | 22 | |
| Gastrointestinal infection | 2 (1.3) | 7.6 | |
| Others | 13 (8.7) | 11 | |
| Type of sepsis | 0.07 | ||
| Non-severe Sepsis | 56 (37.3) | 18 | |
| Severe Sepsis | 26 (16.7) | 9 | |
| Septic Shock | 68 (46.0) | 13 |
The cases included 56 patients with non-severe sepsis, 26 with severe sepsis, and 68 with septic shock. The mean age of patients with septic shock was not statistically different from the other two groups. Likewise, the number of patients with underlying diseases in the septic shock group was comparable to those in other groups. Patients suffering from severe septic had lower levels of vitamin D compared to those with non-severe sepsis (P = 0.07).
5. Discussion
The present study indicated a high rate of vitamin D deficiency in critically ill patients. Although there were different cut-off points for the definition of vitamin D insufficiency, previous reports presented a high rate of vitamin D deficiency in patients with sepsis. In 2015, Alves and others reported that 98% of patients with sepsis had severe vitamin D deficiency (27). Likewise, a subsequent investigation by Shojaei et al. found that the median concentrations of vitamin D levels in patients with sepsis were 19 ng/mL, similar to the concentrations found in our study (24). Jeng and colleagues found significant differences in vitamin D concentrations in patients with sepsis in the intensive care unit compared to healthy individuals (13).
The results of previous studies revealed that vitamin D insufficiency in patients with sepsis was considerably associated with increased mortality (28, 29). The potential mechanisms for the observed investigation may be related to (1) the stimulatory effects of vitamin D on innate immunity, (2) the suppression of immune regulators, and (3) the negative effects on pathways serving to reduce potential inflammatory damage (26, 30-32).
Previous studies showed that vitamin D deficiency might increase the risk of bacterial and viral infections. Accordingly, in a randomized, placebo-controlled trial, patients with pulmonary tuberculosis who received vitamin D in their regimen showed a significant difference in sputum conversion compared to the control group (33). The evidence also proposed that vitamin D might act against influenza and invasive pneumococcal disease (34, 35).
Since vitamin D status plays an important role in the pathogenesis of sepsis and other critical diseases, limited treatment options exist to address this issue. The American Society for Parenteral and Enteral Nutrition (ASPEN) recommends 200 IU vitamin D daily for hospitalized patients (36). However, further investigations have estimated that doses of 400 or 500 IU daily remained in the insufficient range for most hospitalized patients (14, 37). Thus, these findings recommend that higher doses of vitamin D are likely needed to be used in patients with sepsis to correct vitamin D insufficiency.
There is some limitation to the current study. Due to the limited data obtained, we were unable to provide details on acute or chronic liver failure, premorbid health status, type of infection, use of vitamin D supplements, and data on the outcomes. Further studies are required to evaluate the effects of other potential medical comorbidities.
In conclusion, the present study indicated that critically ill patients had vitamin D insufficiency. In line with the biological evidence, we suggest that vitamin D deficiency may predispose patients to sepsis. It seems that the consumption of vitamin D supplements may help decrease the prevalence of sepsis. Further studies are needed to confirm the results and establish the causes and mechanisms underlying these observations.