1. Background
Systemic lupus erythematosus (SLE) is an autoimmune condition characterized by the presence of antinuclear antibodies (ANA) and the formation of immunological complexes, resulting in inflammatory damage to various organs (1). The management of SLE often involves immunosuppressive therapy to achieve disease remission. Due to an immunosuppressed state, SLE patients tend to exhibit more severe COVID-19 symptoms (2). Moreover, COVID-19 has been reported to be associated with symptom flares in SLE patients, manifested by arthritis, alopecia, rash, and pleurisy (2, 3). In light of that, the American College of Rheumatology and the European League Against Rheumatism (ACR/EULAR) strongly recommend prior immunization in SLE patients with approved COVID-19 vaccines (4, 5).
Nevertheless, hesitancy to get vaccinated against COVID-19 rose among SLE patients, primarily due to concerns about potential adverse effects (6); vaccine constituents--inactive viral components or weakened microbes–could trigger molecular mimicry or bystander activation in genetically vulnerable individuals. Also, adjuvants used to enhance immunity in some vaccines can lead to autoimmune reactions (7, 8).
2. Objectives
Existing evidence has predominantly focused on mRNA and adenoviral vector vaccines, leaving a knowledge gap regarding the safety and efficacy of inactivated virus-based vaccines in SLE patients. Additionally, while some studies reported flare-ups in SLE patients following COVID-19 vaccination, distinguishing flares from short-term vaccine-related adverse effects remains challenging. Hence, this study aimed to assess flare diagnoses using a robust approach, with evaluations conducted by medical specialists during clinic visits. The objective was to investigate the incidence and clinical characteristics of flare-ups in patients with SLE following inactivated SARS-CoV-2 vaccination.
3. Methods
3.1. Study Design and Ethics Approval
This cross-sectional study was conducted at the Rheumatology Clinic of Imam Hossein Hospital (Iran) from April 2021 to May 2022, involving SLE patients who received at least 1 dose of the Sinopharm BIBP COVID-19 vaccines. We used census sampling in collecting patient records, and the patients were followed for 3 months after receiving their last dose. The study was approved by the Ethics Committee of Shahid Beheshti University of Medical Sciences, Tehran, Iran (approval No. IR.SBMU.MSP.REC.1401.100), and the authors adhered to the principles of the Declaration of Helsinki.
3.2. Patient Selection
The patients were included in the study if they had a confirmed diagnosis of SLE by a rheumatologist and were in the remission phase within the last 3 months. Remission was defined as meeting the criteria of Lupus Low Disease Activity State (LLDAS): Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K) ≤4, absence of major organ system activity, no new features of disease activity compared to the previous evaluation, and being advised not to decrease or discontinue their daily glucocorticoid dose prior to receiving the vaccine (9). In this study, the SLEDAI-2K score in all participants was 0 (no activity). Additionally, all the participants had received at least 1 dose of Sinopharm inactivated BIBP COVID-19 vaccine. However, patients were excluded from the study if they had a COVID-19 infection within the last 6 months, had concurrent rheumatological diseases, such as rheumatoid arthritis or psoriatic arthritis, were undergoing high-dose therapeutic regimes, had received any other COVID-19 vaccines, such as mRNA or adenoviral vaccines, or had insufficient data with no visits to the clinic during the 3-month follow-up.
3.3. Data Collection
Patient demographics, total vaccine doses received, initial disease manifestations, interval of vaccination to flare-up, laboratory and clinical examination data, flare-up symptoms, and outcomes were extracted from their medical records. Precise assessment of SLE patients was carried out during a 3-month follow-up from each dose of vaccination. The flare-up was assessed using the SLEDAI-2K checklist by an internal medicine specialist and was defined as an increased lupus activity index post-vaccination compared to pre-vaccination.
3.4. Statistical Analysis
Statistical analyses were performed using the STATA v. 14.0 software. The continuous variables' normal distribution was assessed through Q-Q plots. The median and interquartile range (IQR) were used to describe continuous variables. Categorical variables were presented as frequency and percentage (%). If continuous variables did not follow a normal distribution, the Mann-Whitney U test was utilized to compare the means. Differences in the distribution of categorical variables were explored using the chi-square test or Fisher's exact test. All statistical analyses were 2-tailed tests, and significance was set to P < 0.05.
4. Results
4.1. Baseline Patient Characteristics
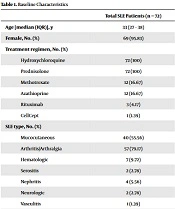
In this study, 72 SLE patients received the Sinopharm BIBP vaccine. The median age was 33 years (IQR: 27–38), and the majority of participants were women (95.83%). The most common organ involvements at diagnosis were arthritis/arthralgia (79.17%), mucocutaneous (55.56%), and hematologic (9.72%) involvements. All of the patients received prednisolone and hydroxychloroquine, and in addition to these drugs, some patients were prescribed immunosuppressive drugs, namely methotrexate (16.67%), azathioprine (16.67%), rituximab (4.17%), or mycophenolate mofetil (CellCept) (1.39%). All the participants had received the first dose, and 66 of them had received both doses (91.67%) (Table 1).
| Total SLE Patients (n = 72) | |
|---|---|
| Age [median (IQR)], y | 33 (27 – 38) |
| Female, No. (%) | 69 (95.83) |
| Treatment regimen, No. (%) | |
| Hydroxychloroquine | 72 (100) |
| Prednisolone | 72 (100) |
| Methotrexate | 12 (16.67) |
| Azathioprine | 12 (16.67) |
| Rituximab | 3 (4.17) |
| CellCept | 1 (1.39) |
| SLE type, No. (%) | |
| Mucocutaneous | 40 (55.56) |
| Arthritis/Arthralgia | 57 (79.17) |
| Hematologic | 7 (9.72) |
| Serositis | 2 (2.78) |
| Nephritis | 4 (5.56) |
| Neurologic | 2 (2.78) |
| Vasculitis | 1 (1.39) |
| Vaccine, No. (%) | |
| Single dose | 72 (100) |
| Both doses | 66 (91.67) |
Abbreviation: SLE, systemic lupus erythematosus.
4.2. Clinical Characteristics of SLE Patients with or Without Flare-ups After Inactivated SARS-Cov-2 Vaccination
Overall, 14 patients (19.44%) experienced a recurrence of SLE symptoms. After vaccination, the most common symptoms of flare-ups were arthritis (64.29%) and skin rash (21.43%). The median SLEDAI-2K score for all the patients was 4 (IQR = 4-4), minimum score = 1 (mild), and maximum score =16 (severe), indicating that most patients had mild disease activity. The median interval between vaccination and symptom recurrence onset was 3 (IQR = 2-6) days. Age, sex, initial lupus manifestations, and treatment medications did not significantly differ between those with and without symptom recurrence (P > 0.05) (Table 2).
| Flare-up Group (n = 14) | No flare-up Group (n = 58) | P-Value | |
|---|---|---|---|
| Age [median (IQR)], y | 30.5 (27 – 38) | 33.5 (27 – 38) | 0.653 |
| Female, No. (%) | 12 (85.71) | 57 (98.28) | 0.095 |
| Treatment regimen, No. (%) | |||
| Hydroxychloroquine | 14 (100) | 58 (100) | N/A |
| Prednisolone | 14 (100) | 58 (100) | N/A |
| Methotrexate | 4 (28.57) | 8 (13.79) | 0.231 |
| Azathioprine | 2 (14.29) | 10 (17.24) | >0.999 |
| Rituximab | 1 (7.14) | 2 (3.45) | 0.483 |
| CellCept | 1 (7.14) | 0 (0.00) | 0.194 |
| SLE type, No. (%) | |||
| Mucocutaneous | 7 (50.00) | 33 (56.90) | 0.641 |
| Arthritis/Arthralgia | 11 (78.57) | 46 (79.31) | >0.999 |
| Hematologic | 2 (14.29) | 5 (8.62) | 0.615 |
| Serositis | 0 (0.00) | 2 (3.45) | >0.999 |
| Nephritis | 1 (7.14) | 3 (5.17) | >0.999 |
| Neurologic | 0 (0.00) | 2 (3.45) | >0.999 |
| Vasculitis | 1 (7.14) | 0 (0.00) | 0.194 |
Abbreviations: N/A, not applicable; SLE, systemic lupus erythematosus.
Recurrence occurred in 6 patients (8.33%) after the first dose and in 8 (12.12%) after the second dose, indicating a higher recurrence rate with the second dose (P < 0.001). However, the severity of symptom recurrence, as measured by the SLEDAI-2K score, did not significantly differ between patients experiencing relapse after the first or second vaccine dose (P = 0.763). Furthermore, there was no significant difference regarding the interval between vaccine injection and the onset of relapse symptoms in the 2 groups (P = 0.075) (Table 3). All the patients experiencing symptom recurrence sought medical consultation, and the prednisolone dose was adjusted for symptom management. One patient was admitted to the hospital for 5 days and received rituximab treatment and high-dose steroid due to vasculitis flare, while another patient switched from azathioprine to CellCept based on the specialist's recommendation. Notably, none of the patients required hospitalization after symptom recurrence.
| Flare-up After 1 Dose (n = 6) | Flare-up After 2 Doses (n = 8) | Total Flare-up (n = 14) | P-Value | |
|---|---|---|---|---|
| Vaccination to flare-up interval [median (IQR)], d | 5.5 (5 – 6) | 3 (2 – 3) | 3 (2 – 6) | 0.075 |
| SLEDAI-2 K score [median (IQR)] | 4 (4 – 6) | 4 (4 – 4) | 4 (4 – 4) | 0.763 |
| Flare-up presentations, No. (%) | ||||
| Arthritis | 3 (50.00) | 6 (75.00) | 9 (64.29) | 0.580 |
| Rash | 3 (50.00) | 0 (0.00) | 3 (21.43) | 0.055 |
| Thrombocytopenia | 0 (0.00) | 1 (12.50) | 1 (7.14) | 1.000 |
| Vasculitis | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
| Raynaud | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
| Leukopenia | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
| Proteinuria | 0 (0.00) | 1 (12.50) | 1 (7.14) | 1.000 |
| Oral ulcer | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
| High DNA binding | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
| Fever | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
| Hospitalization (yes) | 1 (16.67) | 0 (0.00) | 1 (7.14) | 0.429 |
Abbreviation: IQR, interquartile range.
5. Discussion
This cross-sectional study aimed to investigate flare-ups in 72 patients with SLE who received the Sinopharm inactivated BIBP COVID-19 vaccine. Patients were followed for 3 months after the final vaccine dose. Among the participants, 14 patients (19.44%) experienced a recurrence of lupus symptoms following vaccination, with arthritis and skin rash being the most common manifestations. There was a significantly higher recurrence rate after the second vaccine dose compared to the first dose. However, the severity of symptom recurrence did not differ between patients experiencing relapse after the first or second vaccine dose. Hospitalization was not required for most of the patients, and adjustments in prednisolone dosage effectively controlled their symptoms in most cases. In 2 patients, the treatment regimen was changed, and the steroid dose was increased. One of them was admitted to the hospital for 5 days, and the other one was managed on an outpatient basis.
A previous meta-analysis of SARS-CoV-2 vaccination in rheumatic diseases (RDs) revealed an overall 7% post-vaccination flare rate (10). Among different RDs, SLE exhibited flare rates ranging from 0 to 27% (11, 12). In another systematic review of 13 studies involving 2,989 patients, 165 (5.5%) SLE patients experienced lupus flares after the COVID-19 vaccination (4). Our study aligns with the current literature, showing an incidence of 19.44% experiencing a recurrence of lupus symptoms following the COVID-19 vaccination. The variation in flare-up rates could be attributed to differences in the flare-up definitions used in various studies. For instance, Barbhaiya et al. relied on patient self-reports, while Tang et al. defined flares based on specific criteria involving medication dosage changes or new treatments in the presence of worsening symptoms or new organ involvements (13, 14). Additionally, variations in vaccine types and patients' baseline characteristics, as well as disease severity, might contribute to the observed differences in the flare-up prevalence.
Flares following COVID-19 vaccination in SLE patients were most frequently associated with musculoskeletal, dermatological, hematological, and renal symptoms. Additionally, various other uncommon manifestations were reported, such as lupus pneumonitis, leukopenia, myopericarditis, Raynaud's syndrome, and nasal ulcers (4, 10, 15). These flares typically presented quickly within the first week after vaccination, with an average time interval of 2.3 days, according to the study by Zavala-Flores et al. (11). In addition, most of the post-vaccination flares were mild or moderate, while only a small percentage of RD patients experienced severe flares according to another study (16). In our study, all flares were mild to moderate, except for 1 case that resulted in a vasculitis flare in a young woman, leading to hospitalization for 5 days.
The management of SLE often relies on immunosuppressive agents for disease control. While studies generally show no increased flare-up risk post-vaccination with these agents (2, 17, 18), Zavala-Flores et al. identified azathioprine, and Gerosa et al. suggested belimumab as potential risk factors (11, 15).
Regarding the assessment of risk factors for post-vaccination flare-ups, multiple studies have consistently reported that flares were not associated with age, SLE initial manifestations, or the use of corticosteroids, methotrexate, or CellCept. However, flares were found to be more likely in SLE patients with a pre-vaccination high disease activity score (disease severity) (11, 15). Our study aligns with these findings, as we did not observe any significant differences in flare-ups between patients with the aforementioned risk factors.
Patients with RDs are recommended to get protocolized 2-dose vaccination schedules due to poor immune responses after the first dose (19). Previous studies consistently showed a higher risk of flare after the second dose compared to the first dose in these patients (20, 21). Specifically, in the study by Zavala-Flores et al. focusing on SLE patients, similar results were observed with a higher incidence of flare after the second dose. Our study also aligns with these findings. The possible explanation is that the second COVID-19 vaccine dose might cause a stronger immune response, which could increase inflammation and disease activity in SLE patients.
Our study has several limitations. Firstly, the small sample size limited multivariate analyses of factors associated with post-vaccination flares. Secondly, recall bias may have affected the results as it was a post-vaccination survey. Besides, the focus on SLE patients in remission or with low disease activity, even though it aligns with current expert recommendations for COVID-19 vaccination in patients with SLE, limits the generalizability of our findings. Finally, the predominantly female sample might influence results and restrict generalization to male SLE patients.
5.1. Conclusions
The Sinopharm BIBP COVID-19 vaccine was associated with a low incidence of flare-ups in SLE patients in remission, with most being mild and not requiring hospitalization, except for 1 patient who was hospitalized and received rituximab due to vasculitis flare. These findings highlight vaccine safety and underscore the importance of close monitoring, especially after the second dose.