1. Background
Under normal conditions, bile is usually sterile (1), yet the isolation of bacteria from bile of patients with gallstones has derived researchers to study the microbiological epidemiology of gallstones. Though infection may occur in case of biliary tract obstruction, bacteria can access the biliary tract through either the papilla of vater or the portal circulation (2). In different regions (of both stone and bile), various types of bacteria have been found to be associated with gallstones. In a study of 100 stone samples from Colombo, 20% of the patients were positive for having bacteria. In this study, E. coli was found in the majority of cases followed by Klebsiella spp. and Enterobacter spp (3). So far, the most common bacteria isolated from gallstones are aerobes (3, 4), which include E. coli (5-7), Klebsiella spp. (6, 7), Enterobacter fecalis (8, 9), Streptococcus spp., Pseudomonas spp. (4, 8, 9) and Salmonella (10). However, anaerobes were also isolated in few cases (4) such as Clostridium spp., Bacteroids fragilis and Propioni bacteria (1, 11). Infection was found to increase with age (> 60 years) and certain diseases like acute cholecystitis, but sex and the number of common bile duct stones had no influence on positive cultures (11).
Maki et al. postulated that bacterial β-glucuronidase catalyzes the hydrolysis of bilirubin conjugates in bile (12), leading to increased amounts of unconjugated bilirubin, which precipitate as calcium bilirubinate (13-15). Another study, indicated that most biliary bacteria produce slime, and slime production had a greater correlation than beta-glucuronidase production, with pigment stone formation (16). Due to slime production, bacteria form biofilms, which makes them resistant to many antibiotics and this leads to greater severity of infection (17). Patients with primary Common Bile Duct stones and biliary tubes had the highest incidence of slime production. These findings suggest that bacterial slime are important in gallstone formation and the blockage of biliary tubes (18). In a SEM (scanning electron microscopy) study, bacteria were observed only within the calcium bilirubinate-protein matrix of brown pigment stones (P < 0.001). In comparison to black pigment stones, brown stones were more likely to contain calcium palmitate (P < 0.005) and cholesterol (P < 0.001) (19). Bacterial properties determine the path of gallstone formation, for instance, β-glucuronidase and urease activity are involved in calcium precipitation in cholesterol and pigmented stones, respectively. Role of bacteria in gallstone precipitation has been observed but the solidification of gallstones is still under study.
2. Objectives
To the best of our knowledge, this study is the first to report, the role of slime production activity on the solidification of gallstones. Previously, it has been postulated that slime production by biliary bacteria facilitates the adhesion factor of these bacteria on the gallstone surface (17).
3. Patients and Methods
3.1. Sample Collection
Sample of gallstone and bile were collected after cholecystectomy from a government medical college, Rajendra Institute of Medical Sciences, Ranchi, Jharkhand, India. The patients were aged between 30 and 60 years. A total of 526 cases of cholecystectomy were studied from September 2011 to March 2013. Of these 526 cases, 61 patients were asymptomatic and the rest had symptomatic stones. Gallstone samples were washed with sterile normal saline, broken into two halves and the internal surfaces were scoped out and inoculated in nutrient broth. An incubation period of 24 hours at 37°C was provided. The culture was then spread on nutrient agar plates to obtain pure isolated colonies. Colonies showing different morphological characteristics were picked up and identified using different biochemical tests (EMB, MacConkey, TSI (Triple Sugar Iron), Urease, Catalase, etc.).
3.2. Test for β-glucuronidase Activity
β-Glucuronidase production activity was identified via a previously developed method by Jackson et al. (20). Isolates were pour-plated on MacConkey agar containing 100 mg of 4-methylumbellifery-β-D-glucuronide/L. The plates were incubated at 37°C for 24 hours. When the plates were examined under ultraviolet light at 360 nm, the β-glucuronidase producing isolates gave a fluorescent appearance.
3.3. Urease Production Test
The isolated bacteria were grown in urease agar slant by stabbing in the butt and striking in the slant. Urease positive bacteria gave a pink color while a yellow color indicated urease negative bacteria.
3.4. Role of Urease Positive Bacteria in in Vitro Gallstone Formation
Isolates from gallstones with urease activity were inoculated in Brilliant Green Bile Broth (BGBB) supplemented with 10% cholesterol (HiMedia) and 0.5% calcium carbonate (HiMedia). The setup was incubated at 37°C and shaken at 80 rpm. Similar setup was established as a control while the only difference was that it was devoid of bacteria. The experiment was performed in triplicates.
3.5. Slime Formation Test
Slime formation was determined by a previously defined method by Freeman et al. (21). The medium composed of BHI (37 gms/L), sucrose (50 gms/L), agar (10 gms/L) and congo red stain (0.8 gms/L). Congo red was prepared separately from other medium constituents as a concentrated aqueous solution and autoclaved at 121°C for 15 minutes and was then added when the agar had cooled to 55°C. Plates were inoculated and incubated aerobically for 24 to 48 hours at 37°C. A positive test was indicated by black colonies with dry crystalline consistency and pink color indicated a negative test.
3.6. Role of Slime Producing Bacteria in in Vitro Gallstone Formation
Precipitation of calcium was observed in BGBB with bacteria having urease activity within seven days. In the same setup, the isolate from the gallstone with slime production activity, was inoculated along with 2 mL of fresh BGBB. The setup was again incubated at 37°C and shaken at 80 rpm.
3.7. Fourier Transform Infrared Spectroscopy Analysis of Gallstones
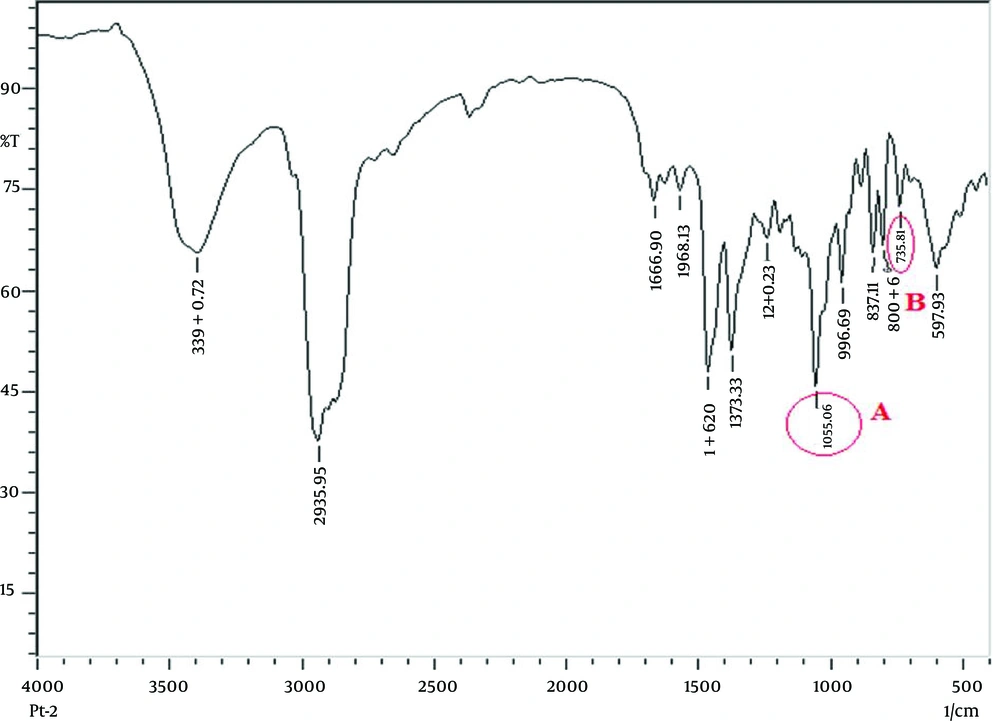
Stones were crushed into fine powder. Two milligrams of the finely powdered stone sample was used to make KBr (potassium bromide) discs. The Fourier transform infrared spectroscopy (FTIR) measurement was taken using the Scimadzu Corpn; Japan; IR-Prestige 21 instrument at a frequency range of 4000-500/cm.
3.8. Statistical Analysis
χ2 analysis was done to determine the relationship between types of stones and bacterial infection. One way ANOVA for interval data of beta-glucuronidase, urease and slime production activity and least significant difference (LSD) test were done for multiple comparisons. All the analysis was done using the statistical package for social sciences (SPSS) 16 software.
4. Results
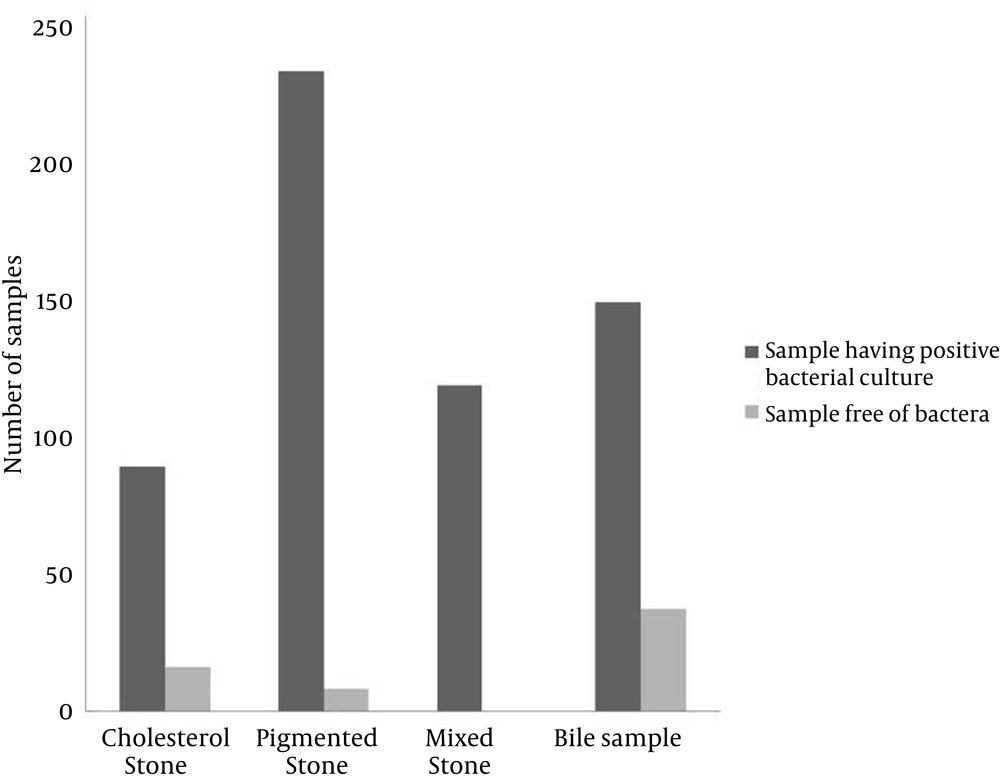
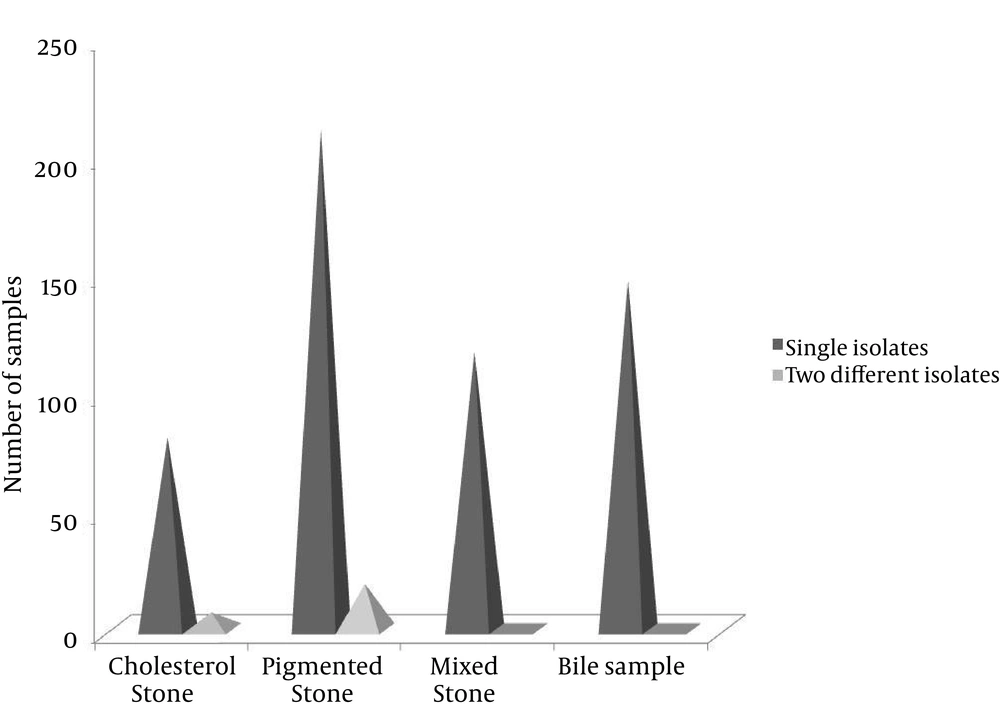
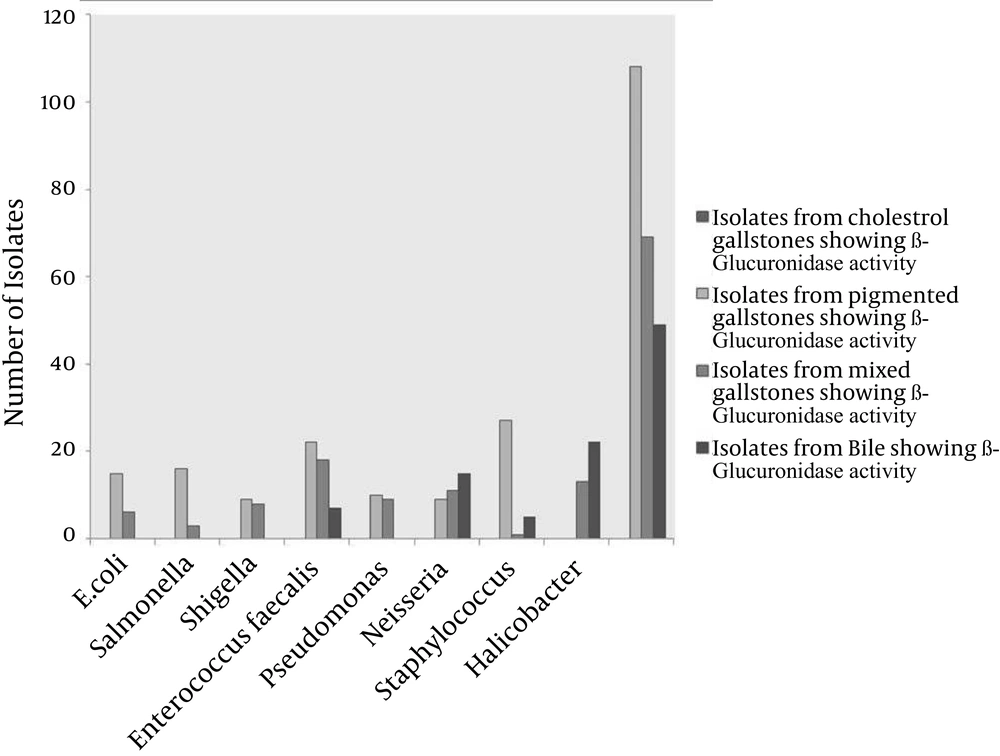
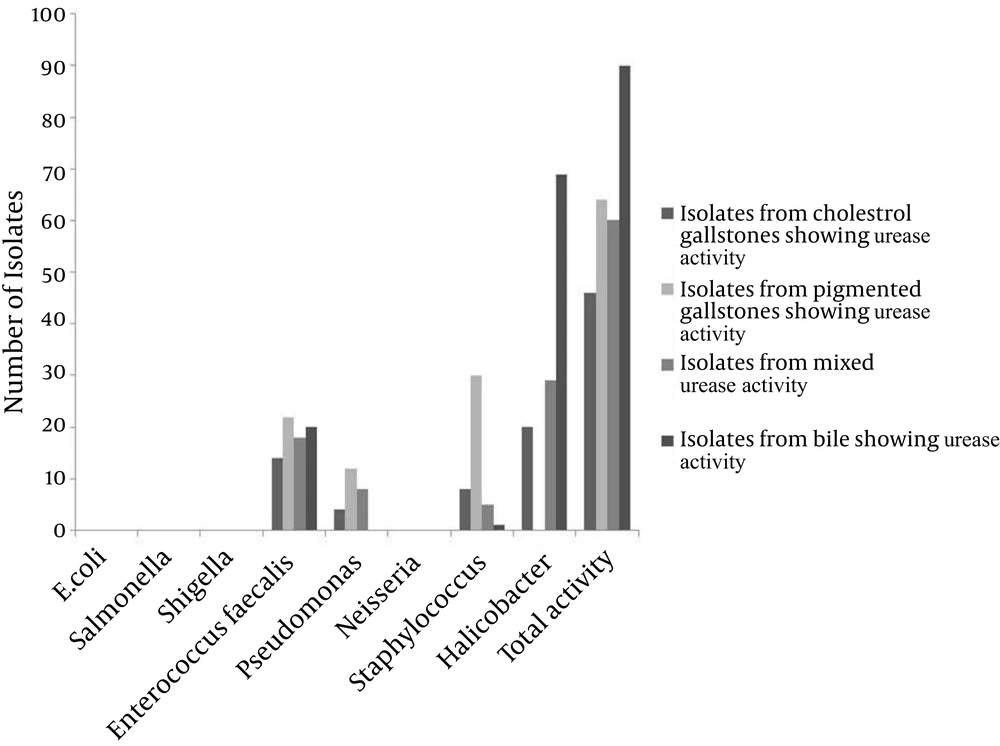
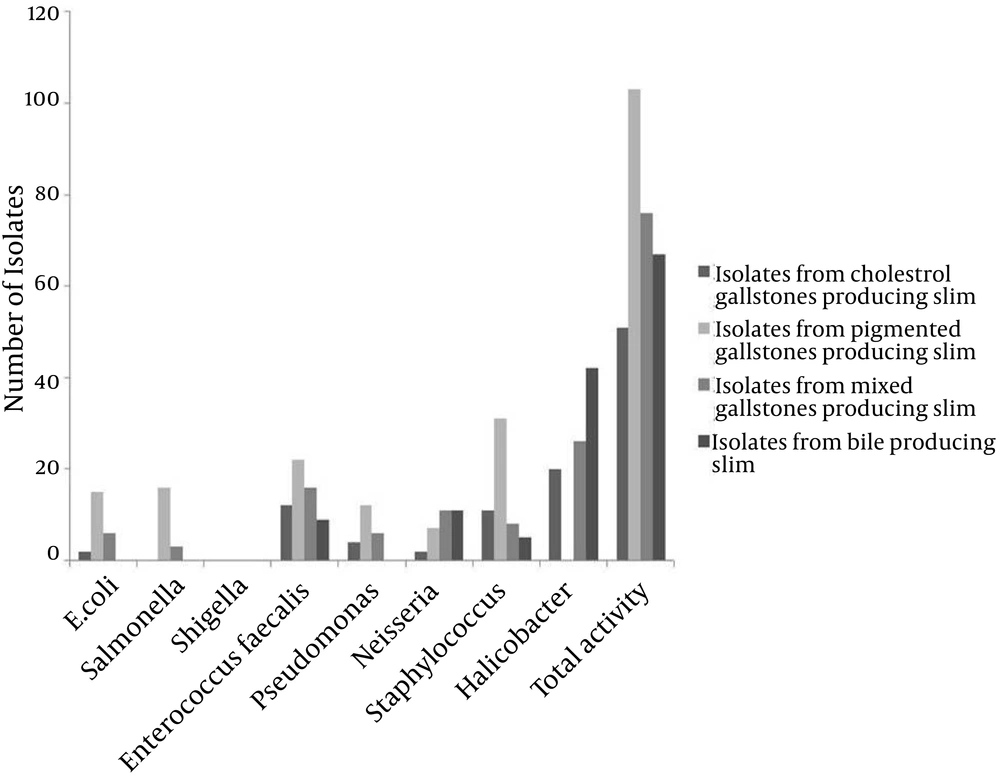
From the 526 cases of cholelithiasis examined between Sep 2011 - March 2013, 459 samples of gallstones and 184 bile samples were collected. The rest were either crushed during the operation or sucked along with the blood during the washing of blood. 435 out of 459 (94.77%) of the gallstones were infected and 27 stones were uninfected, which included 16 cholesterol and 8 pigmented stones (Figure 1). From 392 gallstones and 147 bile samples a single isolate was obtained from each while two different types of isolates were obtained from 26 gallstones; these included 7 cholesterol and 19 pigmented stones (Figure 2). A total of 461 and 147 isolates were obtained from gallstones and bile samples, respectively. Twenty-six isolated species from bile samples were similar to that of gallstones from the same patient. For the rest of the cases different types of species were obtained from bile and stone samples, even from the same patient. This shows that the presence of bacteria inside the gallstone is not via an external contamination rather the bacteria are playing a definite role during the mechanism of gallstone formation. Bacteria were present in 84.61% of 104 cholesterol stones, 96.63% of 238 pigmented gallstones and 100% of 117 mixed gallstones while 79.86% of the bile samples were infected. The types of gallstones are significantly associated with the type of bacteria (χ2 = 18.354, P < 0.0001). First, 300 gallstone isolates and all the bile isolates were selected for identification and microbial studies, and their association with gallstone formation was investigated. Mainly, facultative anaerobes are associated with gallstones, especially with pigmented stones (Table 1). β-glucuronidase activity was shown by isolates form pigmented and mixed stones only. None of the isolates from cholesterol stones showed β-glucuronidase activity whereas very few bacteria isolated from bile have shown this activity (Figure 3) (F = 3.380, P = 0.018; LSD-Cholesterol and bile vs pigmented and mixed) (Table 2). Urease activity is mainly associated with cholesterol gallstones (Figure 4), whereas slime production by the isolates was (Figure 5) not associated with the type of gallstone however bile isolates had a relatively higher slime production activity (Table 2).
| Genus Isolated | Isolates From Bile Sample of Gallstone Patients | Isolates From Cholesterol Gallstones | Isolates From Pigmented Gallstones | Isolates From Mixed Gallstones | Total Isolates | Facultative Anaerobes |
|---|---|---|---|---|---|---|
| E. coli | 0 | 2 | 15 | 6 | 23 | + |
| Salmonella | 0 | 0 | 16 | 3 | 19 | + |
| Shigella | 21 | 13 | 10 | 10 | 54 | + |
| Enterococcus faecalis | 21 | 14 | 22 | 18 | 75 | + |
| Pseudomonas | 0 | 4 | 12 | 8 | 24 | + |
| Neisseria | 31 | 5 | 30 | 11 | 77 | + |
| Staphylococcus | 5 | 11 | 31 | 8 | 55 | + |
| Halicobacter | 69 | 20 | 2 | 29 | 120 | - |
| Total isolates | 147 | 69 | 138 | 93 | 447 |
a LSD- Pigmented vs bile and cholesterol.
b LSD-Cholesterol and bile vs pigmented and mixed.
c LSD-Cholesterol and bile vs pigmented and mixed.
Urease activity and slime production were seen in calcium precipitation and solidification of gallstones, respectively. This has been proved by in vitro analysis by inoculating urease and slime producing bacteria in BGBB supplemented with cholesterol and calcium carbonate. Precipitation of calcium appeared within an average of 7 days after inoculation (Figure 6). After supplementing the same media with slime producing bacteria, the precipitate aggregated to form a small round stone-like structure after an average of 12 days from the day of urease bacteria inoculation. The liquid media were decanted and photographed (Figure 7). The experiment was repeated in triplicates.
5. Discussion
Role of bacteria in gallstone formation was proposed a long time ago. In our study, we found a predominant role for bacteria in gallstone formation as 94.77% of the gallstones were found to be infected with bacteria. In our study we obtained facultative anaerobic bacteria from our samples, excluding Halicobacter spp., which is aerobic in nature (Table 1). Facultative anaerobic nature of bacteria facilitates their survival inside the gallstones, although complete anaerobes were not seen.
The general mechanism involved in gallstone formation starts with nucleation (22). Bacteria also play a very important role in nucleation either by acting as a nidus itself, over which cholesterol deposition takes place or their enzyme activity may contribute to the formation of insoluble precipitates (23). Maki et al. was the first to note the importance of biliary bacterial infection in pigmented gallstone formation (12). He postulated the role of bacteria in coagulation of calcium bilirubinate stones, a major step in pigmented gallstone formation. Bilirubin, which is present in the blood, is esterified in the liver to form water-soluble glucuronides, before it is excreted into bile. In the presence of β-glucuronidase activity of bacteria, bilirubin glucuronide becomes hydrolysed to release free bilirubin, which combines with calcium to form calcium bilirubinate, a water insoluble compound. This insoluble calcium bilirubinate acts as a nidus for pigmented stone formation (24). At present, we have also isolated β-glucuronidase positive bacteria from pigmented and mixed stones while isolates from cholesterol stones did not show β-glucuronidase activity. Even the isolates of the same genus from cholesterol stones did not show β-glucuronidase activity. According to Stewart et al. β-glucuronidase activity is the major pigmentation factor in gallstones and this activity seemed to be more associated with mixed stones (16). However, in our study we did not find such an association; instead isolates form pigmented and mixed stones were almost equally probable of having β-glucuronidase activity. Until 1998, it was said that cholesterol stones are devoid of bacterial infection. However, bacteria were seen in SEM on the outer surface of cholesterol stones by Wu et al. (25). Kawai et al. found an association between Gram-positive cocci and pure cholesterol stones. Recently, bacteria DNA were also isolated from cholesterol stones (26). Nucleation of pigmented stones due to β-glucuronidase activity is very clear and the absence of β-glucuronidase activity in cholesterol stones has intrigued researchers to study the nucleation factor for cholesterol stones. It has been postulated that cholesterol stones nucleate when cholesterol supersaturates in the bile. Yet, bacteria are present in the cholesterol stone also, and might play some role in its formulation. Boquet et al. observed bacterial activity in CaCO3 precipitation, a finding which has also been supported by other (Table 3) (27).
Similar to calcium bilirubinate, the CaCO3 precipitate may act as a nidus in gallstone formation in cholesterol stones. Belzer et al. postulated that the urease activity of bacteria in calcium precipitation leads to nucleation for gallstone formation (33). We have experimentally established the role of urease in calcium precipitation and slime activity of bacteria in stone solidification under in vitro conditions. Bacteria having urease activity precipitated calcium carbonate in the added medium (Figure 6). Solidification of the precipitate resulted in the presence of bacterial slime activity (Figure 7).

Ureolytic bacteria generate carbonate from urea and precipitate CaCO3 to initiate crystal formation, as shown in Equation 1. In the FTIR study, we also found CaCO3 and vaterite (a calcium carbonate precipitate product) in all types of gallstones (Figure 8), which proved that urease activity might be involved in CaCO3 precipitation for initiation of gallstone formation. Nucleation factors for the gallstones had been already determined, yet until now, factors for stone solidification have not been explained. We are the first to postulate that slime producing bacteria may be involved in stone solidification. Slime basically consists of polysaccharides and minor amounts of proteins. This provides viscosity of up to 50000 cps, which act as a glue (34). The biopolymers of the slime crosslink with the precipitated calcium and may increase the strength of the gallstone leading to its solidification. Slime positivity was seen in 76.67% of the isolates and there is no association between slime production and the type of gallstone. Role of slime activity of bile isolates is still unclear, but it is hypothesized that they may provide adhesion factors for the gallstone surface and their cross linking activity might prevent the exposure of bacteria inside the gallstone, making them resistant over antibiotics. Thus, slime production may be one of the factors for stone solidification irrespective of its type (P < 0.0001).
β--glucuronidase activity of bacteria is mainly involved in pigmented gallstone formation. However, urease and slime production activity of bacteria help in nucleation and solidification of gallstones, respectively; irrespective of gallstone types. These two biliary bacterial factors do not determine the formation of different types of gallstones and may rather be involved in the general steps of gallstone formation.