1. Background
Hospital acquired foodborne diseases are considered as life-threatening complications among hospitalized patients (1). The role of medical foods in transmission of pathogenic bacteria and occurrence of gastrointestinal diseases are established by several reports (2, 3). While foodborne transmission of microbes responsible for nosocomial outbreaks encompass a small portion of these events, the importance of some bacterial pathogens including Klebsiella pneumonia, K. oxytoca, Salmonella spp., Clostridium perfringens, Coliforms, Bacillus cereus, and Staphylococcus aureus was illustrated in foodborne outbreaks in different hospitals (4-7). Medical foods provide essential energetic metabolites for the growth of pathogenic microorganisms. Also, they could provide necessary conditions to produce toxins with serious impact on human health. Preparation of some of these foods, such as the foods for enteral tube feeding, requires a great deal of processing that could increase the risk of microbial infection among them. Some risk factors such as the existence of weak immune system among the patients, changes of natural microbial community in their gastrointestinal tract (GIT), or their natural performance of the body defense system could boost their susceptibility to foodborne diseases in the hospital setting.
Poor hand hygiene of hospital kitchen staff could serve as the main risk factor in contamination of foods. Resident microbial flora of cooking staffs and usage of contaminated utensils, together with impaired cooking process are the main putative risk factors in the food microbial contamination in hospitals (8-11). Food staff can also indirectly involve in nosocomial infections through introducing new resistant bacterial strains into hospital kitchens or the environment.
Despite the high number of studies conducted on hospital-acquired infections (HAIs) in Iranian hospitals, only a few studies investigated the possible role of foods in this regard. A surveillance study provides reliable data about these pathogens and their transmission patterns. According to the priority of preventing foodborne outbreaks in the hospitals that provide homemade food products, assessing the role of medical foods in the occurrence of HAIs and transmission of pathogenic bacteria into the hospital environments are of great importance. Characterization of pathogenic bacteria, their antibiotic resistance profiles, and similarity of their molecular patterns among the isolates of food, food staff, cooking instruments, and patients’ clinical samples were aimed in the current study to analyze their possible transmission patterns in a teaching hospital in Tehran, Iran.
2. Methods
2.1. Setting and Sampling
The current cross sectional study was performed in a hospital in Tehran, Iran from April 2011 to January 2013. Accordingly, simple random sampling of raw and cooked food materials, swab samples of cooking utensils, and hands and noses of food staff were used in the current study. The sample size was calculated based on a power of 80%. The clinical samples (urine and blood samples of patients with urinary tract and blood stream infections), environmental swab samples of intensive care unit (ICU), and hands of health care workers were collected from the same hospital by the standard sampling method as described by Mackowiak et al. at the same time scale (12). The samples were collected from all food preparation steps while the involved staff was unaware of the sampling. All prepacked samples and food materials were excluded from the study. All the samples were immediately transferred to laboratory for further processing and microbiological analyses. In the case of swab samples, all of the specimens were cultured on selective (mannitol salt agar and MacConkey agar) and non-selective media (blood agar). In the case of food samples, defined amounts of the specimens were suspended in normal-saline solution and homogenized in a STOMACHER system (Seward stomacher) (13). Defined amounts of each food suspension were cultured on the culture media. Colony count of each bacterial isolate was determined after 24 hours incubation at 37°C under ambient atmosphere. The current study protocol was approved by the ethical committee of research institute for gastroenterology and liver diseases (RIGLD-688).
2.2. Bacterial Identification
Characterization of the grown bacterial isolates was done based on conventional biochemical tests (14). Briefly, staphylococcal isolates were characterized based on their colony morphology on mannitol salt agar and blood agar, microscopic examination, catalase, DNase, and coagulase activities. To confirm the identity of S. aureus isolates, polymerase chain reaction (PCR) was performed for 2 species of specific gene loci nuc and femA (Section 3.1). In the case of the grown Gram-negative bacteria on MacConkey agar, patterns of their fermentation in triple sugar iron (TSI) agar and results of their IMViC and lysine decarboxylase reactions, utilization of amino acids, and sugars were used.
2.3. DNA Extraction and PCR
In order to do molecular fingerprinting experiments, total DNA of E. coli (as a marker of fecal contamination) and S. aureus (a common skin pathogen) isolates were extracted from freshly prepared overnight cultures on blood agar medium. In the case of S. aureus isolates, the bacterial colonies were resuspended in 45 µL of Tris-EDTA buffer (pH 8) and 5 µL of lysostaphin solution (Sigma-Aldrich, USA) (100 µg/mL) was added. The samples were incubated at 37°C for 10 minutes. After the incubation, 5 µL of proteinase K solution (100 µg/mL) and 150 µL of 0.1 M Tris-HCl (pH 7.5) were added to the suspension. The samples were incubated for 10 minutes and heated for 5 minutes at 100°C. The DNA samples were collected after 10 minutes centrifugation in 13,000 g. A DNA purification kit was used to extract DNA from the E. coli isolates (DNP™ kit, Cinagen, Iran).
2.4. PCR for nucA and femA
PCR was utilized to detect S. aureus isolates with species specific primers that detect nucA gene, nuc-F: 5′-CTGGCATATGTATGGCAATTGTT-3′ and nuc-R: 5′-TATTGACCTGAATCAGCGTTGTCT-3′, and femA gene, femA-F: 5′-AAAAAAGCACATAACAGCG-3′ and femA- R: 5′- GATAAAGAAGAAACCAGCAG-3′. In brief, a total of 1 µL DNA template was added to 24 µL of PCR mixture containing 2.5 µL Taq DNA polymerase buffer, 0.75 µL MgCl2, 0.5 µL dNTPs (Gene Fanavaran, IR.IRAN), 1.25 µL of each primer, and 0.15 µL of Taq DNA polymerase (Gene Fanavaran, IR.IRAN). The amplification was carried out on a thermal cycler under the following conditions: first, denaturation at 95°C for 3 minutes followed by 30 cycles of denaturation at 95°C for 40seconds, annealing at 53°C for 40 seconds (for nuc gene) and 47°C for 40 seconds (for femA gene), and extension at 72°C for 40 seconds. The final extension was performed at 72°C for 5 minutes. The amplified products were separated by electrophoresis in 1.2% agarose gel at 90 V for 90 minutes.
2.5. Antimicrobial Susceptibility Testing
All the isolates were tested for susceptibility to commonly used antibiotics to treat infections caused by different bacteria. The assay was performed by Kirby-Bauer disk diffusion method on Mueller-Hinton agar medium according to the CLSI (the Clinical and Laboratory Standard Institute) guidelines (15).
2.6. RAPD-PCR Fingerprinting
PCR was carried out in 25 µL reaction mixture containing 7 µL of genomic DNA, 1.5 mM MgCl2, 2.5 µL PCR buffer, 1.2 µL dNTPs (Gene Fanavaran, IR.IRAN), 2.5 µL primer, and 0.2 µL of Taq DNA polymerase (Gene Fanavaran, IR.IRAN). The cycling program when using 10-nt primer 1283 included initial denaturation at 94°C for 4 minutes, first annealing at 36°C for 4 minutes and first extension at 72°C for 4 minutes followed by 4 cycles of the second denaturation at 94°C for 30 seconds, annealing at 36°C for 1 minute, and final elongation at 72°C for 2 minutes. Electrophoresis in 1.8% agarose gel was applied to separate the amplicon fragments according to their sizes. The agarose gels were stained with 1% ethidium bromide and analyzed under UV transilluminator (Gel Documentation System, Syngene). Similarity of all RAPD banding profiles was analyzed by GelCompar II Software. Polymorphisms of ≤ 2 and > 2 RAPD bands were considered as definitive criteria to detect related and different strains, respectively.
2.7. Statistical Analysis
The data were analyzed using SPSS software version 19; the chi-square test was applied wherever applicable. In addition, chi-square and the Fisher exact tests were used to find the statistical correlation between the frequency of bacteria from hospital kitchen and ICU isolates. Statistical analyses were done to detect the association among drug resistance, types of pathogens isolated from the samples, similarity of phenetic data, and similarity of the strains in biotyping compared with those of the molecular typing methods. Significance was defined as a P value < 0.05.
3. Results
Frequency of bacteria in hospital kitchen and ICU:
Out of 200 kitchen samples used in the current study (foods = 44, utensils = 65, food handlers = 91), five common bacteria including S. aureus, E. coli, Acinetobacter spp., Pseudomonas spp., and Enterococcus spp. were isolated in frequencies of 15.5%, 8%, 2.5%, 0.5%, and 0.5%, respectively (Table 1). Total rate of the contamination in the kitchen samples was 27.5% (foods 5/44 (11.3%), utensils 26/65 (40%), and food handlers 24/91 (26.3%). Among the clinical samples, S. aureus (15.1%), E. coli (9.43%), Enterococcus spp. (11.33%), Pseudomonas spp. (5.6%), and Acinetobacter spp. (9.4%) were identified. While only S. aureus (5.8%) and Enterococcus (0.5%) were isolated from the health care workers, all the noted bacteria were isolated from the environmental samples of the studied ICU in frequencies shown in Table 1.
| Kitchen Samples | Hospital Samples | |||||||
|---|---|---|---|---|---|---|---|---|
| Bacteria | Food a (N = 44) | Food Staffs (N = 91) | Utensils (N = 65) | Total (N = 200) | Clinical Samples (N = 53) | ICU Environment (N = 90) | Health Care Workers (N = 179) | Total (N = 322) |
| S. aureus | 2 (4.5) | 19 (20.8) | 11 (16.9) | 32 (15.5) | 8 (15.1) | 6 (6.6) | 10 (5.58) | 24 (7.4) |
| E. coli | 3 (6.8) | 2 (2.1) | 11 (16.9) | 16 (8) | 5 (9.4) | 0 | 0 | 5 (1.5) |
| Acinetobacter spp. | 0 | 2 (2.1) | 3 (4.6) | 5 (2.5) | 5 (9.4) | 6 (6.6) | 0 | 11 (3.4) |
| Pseudomonas spp. | 0 | 0 | 1 (1.5) | 1 (0.5) | 3 (5.6) | 1 (5.8) | 0 | 4 (1.2) |
| Enterococcus spp. | 0 | 1 (1.1) | 0 | 1 (0.5) | 6 (11.32) | 3 (3.3) | 1 (0.55) | 10 (3.1) |
aN, total number of samples was collected in each category; N, number of each bacterial genus isolated from each type of sample.
bValues are expressed as No. (%).
3.1. Molecular Detection of S. aureus Strains
Identification of the biochemically characterized S. aureus isolates was followed by molecular methods. Accordingly, their identity was confirmed in all cases by PCR for nuc and femA genes.
Resistance profile and multidrug resistance phenotype of the bacterial isolates in hospital kitchen and ICU
The frequency of antibiotic resistance among the strains isolated from the hospital kitchen and ICU was summarized in Table 2. In general, bacterial isolates from the ICU showed higher rates of resistance, compared with those isolated from the kitchen. However, resistance to azithromycin in Acinetobacter spp. strains obtained from the hospital kitchen was significantly higher than those isolated from the ICU (P = 0.0001). Also, resistance to cefepime among the P. aeruginosa strains from the utensils was higher (100%) than those isolated from the clinical samples (0%). Multiple drug resistant-methicillin resistant strains of S. aureus (MDR-MRSA) comprised over 18.75% (6/32) of all the isolates among the samples of the hospital kitchen and 91.6% (22/24) of the ICU. The MDR-MRSA strains were related to foods, 16.66% (7/44), food handlers, 33.33% (30/91), and utensils, 50% (32/65). Most of these strains were resistant to amoxicillin-clavulanate and cefepime. Frequency of MDR patterns among the E. coli strains in the kitchen was 52.94%, and the highest MDR phenotype was observed among the isolates from the utensils. No other bacterial species showed MDR pattern in the studied kitchen.
| Hospital Kitchen (N) % | ICU (N) % | |||||
|---|---|---|---|---|---|---|
| Foods | Utensils | Staffs | Environment | Clinical Samples | Staffs | |
| Staphylococcus aureus | 2 | 11 | 19 | 6 | 8 | 10 |
| Penicillin | (2) 100 | (11) 100 | (19) 100 | (6) 100 | (8) 100 | (10) 100 |
| Gentamicin | 0 | 0 | 0 | (6) 100 | (7) 87 | (5) 50 |
| Chloramphenicol | 0 | 0 | 0 | 0 | 0 | (1) 10 |
| Amoxicillin clavulanic acid | (1) 50 | (4) 36.3 | (10) 52.6 | (5) 83.3 | (8) 100 | (8) 80 |
| Ciprofloxacin | 0 | 0 | 0 | (4) 66.6 | (8) 100 | (4) 40 |
| Erythromycin | (2) 100 | 0 | (1) 5.2 | (6) 100 | (8) 100 | (7) 70 |
| Cefepime | (2) 100 | (11) 100 | (16) 84.2 | (6) 100 | (8) 100 | (8) 80 |
| Cefotaxime | 0 | 0 | 0 | (5) 83.3 | (8) 100 | (6) 60 |
| Cefoxitin | (1) 50 | (3) 27.2 | (5) 26.3 | (6) 100 | (8) 100 | (8) 80 |
| Trimethoprim/sulfamethoxazole | 0 | 0 | 0 | (3) 50 | 0 | (1) 10 |
| Imipenem | 0 | 0 | 0 | (5) 83.3 | (8)100 | (4) 40 |
| Escherichia coli | 3 | 11 | 2 | 0 | 5 | 0 |
| Ceftriaxone | 0 | 0 | (2) 100 | - | (4) 80 | - |
| Ceftazidime | 0 | 0 | (2) 100 | - | (5)100 | - |
| Chloramphenicol | 0 | 0 | 0 | - | (2) 40 | - |
| Ciprofloxacin | 0 | 0 | 0 | - | (2) 40 | - |
| Trimethoprim/sulfamethoxazole | 0 | 0 | 0 | - | (2) 40 | - |
| Ampicillin | (3) 100 | (9) 81.8 | (1) 50 | - | 0 | - |
| Azithromycin | (1) 33.3 | (2) 18.1 | (1) 50 | - | (4) 80 | - |
| Cefepime | 0 | (2) 18.1 | 0 | - | (4) 80 | - |
| Amoxicillin Clavulanic acid- | (3) 100 | (2) 18.1 | (11) 57.8 | - | (5) 100 | - |
| Nalidixic acid | 0 | (1) 9 | 0 | - | (3) 60 | |
| Tetracycline | 0 | 0 | 0 | - | (5) 100 | - |
| Cefalotin | (2) 66.6 | (8) 72.7 | 1 (50) | - | (5) 100 | - |
| Gentamicin | 0 | 0 | 0 | - | (3) 60 | - |
| Imipenem | 0 | 0 | 0 | - | (4) 80 | - |
| Acinetobacter spp. | 0 | 3 | 2 | 6 | 5 | 0 |
| Imipenem | - | 0 | 0 | (6) 100 | (5) 100 | - |
| Piperacillin/tazobactam | - | (2) 66.6 | 0 | (6) 100 | (5) 100 | - |
| Azithromycin | - | (2) 66.6 | 0 | (10) 16.6 | (3) 60 | - |
| Cefepime | - | (3) 100 | (2) 100 | (6) 100 | (5) 100 | - |
| Ceftazidime | - | (3) 100 | (2) 100 | (6) 100 | (5) 100 | - |
| Ciprofloxacin | - | 0 | 0 | (6) 100 | (5) 100 | - |
| Ofloxacin | - | 0 | 0 | (6) 100 | (5) 100 | - |
| Gentamicin | - | 0 | 0 | (5) 83.33 | (2) 40 | - |
| Tobramycin | - | 0 | 0 | (6) 100 | (2) 40 | - |
| Trimethoprim/sulfamethoxazole | - | 0 | 0 | (6) 100 | (5) 100 | - |
| Ceftriaxone | - | (3) 100 | (2) 100% | (6) 100 | (5) 100 | - |
| Pseudomonas spp. | 0 | 1 | 0 | 1 | 3 | 0 |
| Imipenem | - | 0 | - | 0 | 0 | - |
| Piperacillin/tazobactam | - | 0 | - | 0 | 0 | - |
| Azithromycin | - | 0 | - | (1) 100 | (3) 100 | - |
| Cefepime | - | (1) 100 | - | 0 | 0 | - |
| Ceftazidime | - | 0 | - | (1) 100 | (3) 100 | - |
| Ciprofloxacin | - | 0 | - | 0 | 0 | - |
| Ofloxacin | - | 0 | - | 0 | 0 | - |
| Gentamicin | - | 0 | - | 0 | 0 | - |
| Tobramycin | - | 0 | - | 0 | 0 | - |
| Trimethoprim/sulfamethoxazole | - | 0 | - | (1) 100 | (2) 66.6 | - |
| Ceftriaxone | - | 0 | - | (1) 100 | (2) 66.6 | - |
| Enterococcus spp. | 0 | 0 | 1 | 3 | 6 | 1 |
| Gentamicin | - | - | 0 | (1) 33.3 | (5) 83.3 | 0 |
| Ampicillin | - | - | 0 | (1) 33.3 | (6) 100 | 0 |
| Vancomycin | - | - | (1) 100 | (2) 66.6 | (4) 66.6 | (1) 100 |
| Penicillin | - | - | (1) 100 | (3) 100 | (6) 100 | (1) 100 |
| Ciprofloxacin | - | - | 0 | (1) 33.3 | (6) 100 | 0 |
| Nitrofurantoin | - | - | 0 | (2) 66.6 | (4) 66.6 | (1) 100 |
| Chloramphenicol | - | - | 0 | (1) 33.3 | (1) 16.6 | (1) 100 |
Comparison of the frequency of resistance to the studied antibiotics in either ICU or kitchen samples did not propose homology of the bacterial isolates for most of the genera between the 2 environments (Table 2). Similarity of the resistance patterns among the S. aureus strains from the kitchen or those isolated from the ICU was high; however, this similarity was not the case between the 2 groups. The current study results also showed dissimilarity of the E. coli strains between the 2 places, since all the isolates from the hospital kitchen were put in separate groups.
3.2. S. aureus and E. coli Phylogenetic Dendrogram:
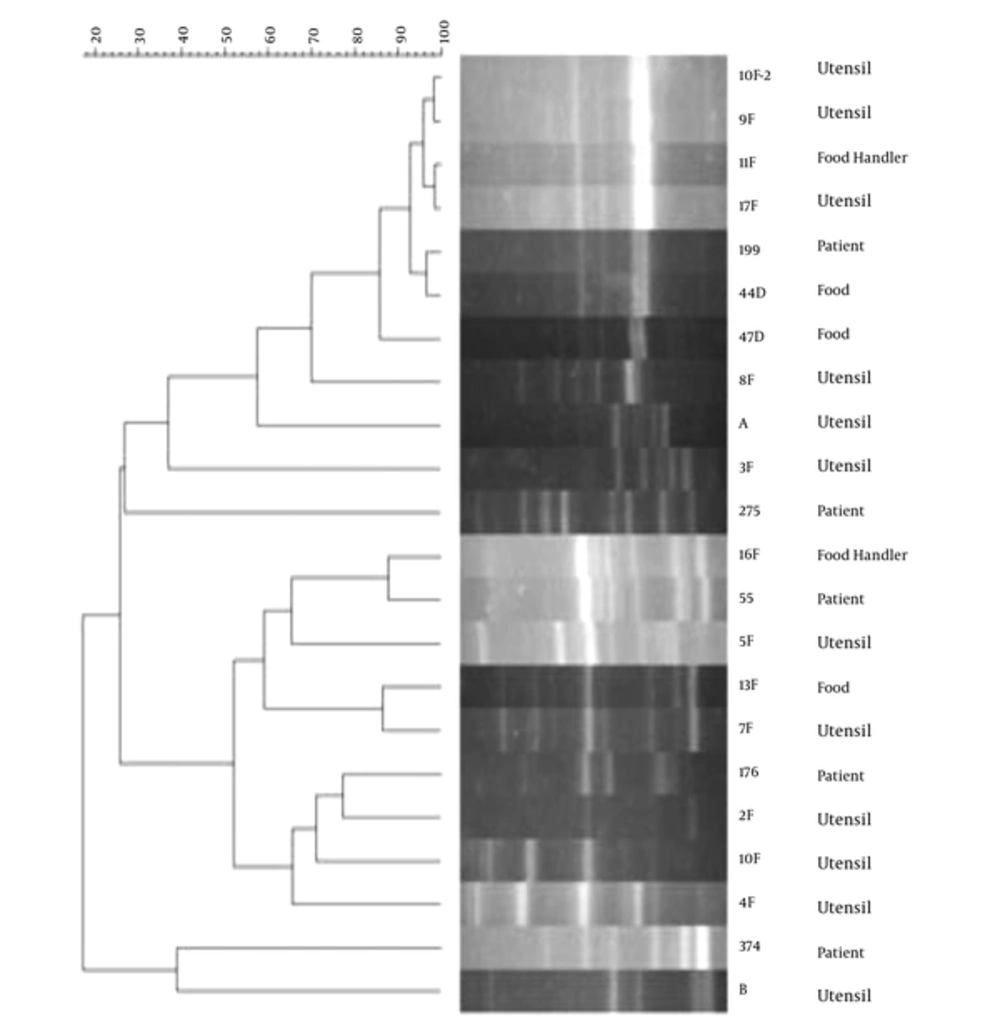
The phylogenetic analysis of S. aureus RAPD patterns did not show significant relationship between the isolates of the ICU and hospital kitchen. However, as shown in Figure 1, different strains of this bacterium from the food staff (Strains 1A-1C) or food staff and utensils (strains 27D-4D and 12A-21A) showed identical patterns. In the ICU samples, similarity of the S. aureus RAPD patterns was detected among the strains isolated from the patients (strains 215-2, 64-2, 65-2) and those isolated from the ICU environment (strains 192-2 and 373), individually. These analyses also presented the relationship between the E. coli strains isolated from the studied foods and utensils (strains 9A-17A-44D-8F). Two strains of the patients showed similar patterns with one strain isolated from the food handlers (strains 55 and 16F, respectively) and 7 strains isolated from the kitchen samples; however their diversity in resistance patterns refused this correlation (Figure 2).
4. Discussion
Hospital kitchen appears to be a source of food contamination and occurrence of foodborne outbreaks in hospitalized patients in different countries (16-19). Utensils, such as industrial blender and meat grinder, regardless of the contamination of raw food materials, could be the sources of pathogenic bacteria, because their cleaning and disinfection cannot accompany completely due to their designed structure (20, 21). Food handlers, through their weak health, are also suspects of common sources of bacterial contamination in such outbreaks. Incomplete cooking of the contaminated foods can introduce important bacterial pathogens involved in gastrointestinal and hospital acquired infections (22, 23). Results of the current study showed utensils as the most contaminated samples in the studied kitchen. S. aureus and E. coli, as known members of the skin and faecal microbiota, were equally detected in these samples with the highest frequency (16.9%). This finding was in agreement to a recent study in Italy that established the presence of skin associated bacteria, including Staphylococcus, Streptococcus, Corynebacterium, and Propionibacterium spp., in cooking and processing tools in a hospital cooking center (24). The high rate of contamination with coliform bacteria in the food utensils could be explained by weak hygiene of the food handlers or using contaminated food staff (2, 25). Given the lower level of E. coli contamination in the studied food samples (6.8%), food handlers seem to be the main sources of this contamination in this hospital. This finding was supported by the results of molecular and phenetic typing methods, which provided evidences of the occurrence of cross contamination between the staff of the hospital kitchen, foods, and utensils. In a study by Anderton and Aidoo, the effect of handling procedures on the level of microbial contamination of enteral feeds was reported (26). They found no contamination in foodstuffs collected from systems assembled wearing sterile gloves, while the contamination was detected when non-sterile disposable gloves were used by the food handlers. In a study by Borges et al. in Brazil, 36% of hospital food handlers harbored S. aureus on their nails and/or hands, which was higher than that of the current study rates (20.87%) (27). Aycicek H. et al., in Turkey showed a frequency of 70% (S. aureus) and 7.8% (E. coli) contamination on the hands of food handlers that was due to poor hand hygiene and improper glove use (28). These differences could be explained by factors that influence safety of food materials including socioeconomic conditions, geographic region, and performance of surveillance programs in each country. Transmission of pathogenic bacteria from contaminated utensils and/or food handlers to medical foods and hospital environment is problematic in clinical settings. In the current experiment, this type of cross contamination was limited to the food handlers and utensils, patient to patient, and samples of the hospital environment. However, involvement of food handlers in contamination of medical foods and occurrence of hospital foodborne outbreaks was previously reported in some countries (29-33). In the cases of E. coli and S. aureus, Borges. et al. and Kluytmans J. et al. established involvement of food handlers in the occurrence of foodborne outbreaks through contamination of medical foods (27, 34). While the current study results did not support involvement of the bacterial isolates in HAIs, transmission of these bacteria to patients’ foods and also their survival after the cooking procedure proposed them as possible sources of intestinal and extra-intestinal infections in hospitalized patients consuming them. Resistance of these bacteria to multiple drug families was also considered as a risk factor in this hospital. Spread of MDR bacteria between utensils/food handlers and foods is a disturbing thread, because these bacteria are involved in most of the infections acquired from hospitals. Although results of the susceptibility testing showed the presence of MDR patterns among different strains of E. coli and S. aureus in the hospital foods and utensils, spread of other MDR bacteria, such as Acinetobacter, Pseudomonas, and Enterococcus spp., through hospital kitchen was not confirmed in these foods and the studied kitchen during the current study.
4.1. Conclusions
Results of the current study indicated E. coli and S. aureus as the most common bacteria isolated from foods, utensils, and food staffs in the studied hospital kitchen. However, detection of other bacteria, including Acinetobacter, Pseudomonas, and Enterococcus spp. was established in the current study. Although results of the antimicrobial susceptibility testing showed low frequency of MDR phenotypes among these isolates in this hospital kitchen, characterization of the MDR E. coli and MDR-MRSA strains proposed their risk of transmission into hospital environment or patients consuming them. Results of the molecular- and the resistance biotyping experiments suggested the occurrence of cross-contamination between the food staff/utensils and food samples. In this regard, food staff and utensils were considered as the main sources of S. aureus and E. coli transmission, respectively. Poor sanitary practices for food processing, weak health conditions of food handlers and inappropriate cooking practice were postulated as the main risk factors for bacterial contamination of the medical foods, which strongly suggests that efforts should be made to improve health status in this hospital. While simultaneous analysis of bacterial contamination of cooking instruments, food stuff, and food handlers and their transmission throughout the food chain in a hospital was the main strength of the current study, limited numbers of samples used in the current study was its main weakness. Further studies in different hospitals are needed to determine the main risk factors associated with foodborne diseases in hospital settings.