1. Background
Globally, a new case of dementia (the loss of cognitive functioning) occurs every four seconds (1). Alzheimer's disease (AD), the most common cause of dementia among older adults, is currently ranked as the sixth leading cause of death in the United States but appears to be the third-ranked cause of death in the elderly (1). Alzheimer’s disease is a neurodegenerative brain cell disorder characterized by neuropathological changes that cause a progressive impairment of recent memory, semantic aphasia, agnosia, apraxia, and a decrease in executive function (2).
With AD having tremendous influences on public health and economics, approaches have to be proposed and implemented to prevent and/or delay the progression of AD (3). In this regard, prevention and the slowing of AD progression should be directed towards the recognition of modifiable risk factors. In order to achieve this, interventions need to be implemented that improve cognitive engagement, improve nutrition, increase antihypertensive drug use, and/or increase participation in physical exercise (4). Early detection and treatment of AD will allow for more focus on improving the quality of life (QOL) of the patient suffering from AD, as well as the patient’s caregiver, to decrease the family burden and delay the point of placing the patient in a care facility (5, 6).
The advancement of AD may be prevented or delayed via the use of exercise due to its ability to enhance cognitive functioning and brain plasticity and improving neuronal health (3, 7). It has been proposed that patients at all stages of AD can benefit from regular exercise due to exercise’s ability to maintain overall motor function and its proposed positive outcomes on cognitive function, behavioral problems, sleep, and overall well-being (3, 8). However, for exercise to be an effective treatment method in AD, the patient with AD must remain engaged in and committed to the exercise-training program. This is problematic since it is a generally accepted challenge to keep patients with AD engaged over a short-term and long-term period (9). This lack of adherence on the part of patients with AD is a result of various influencing factors, such as task value and difficulty, practicality, motivation, personality traits, and companionship (10). These factors can certainly be overcome by implementing the best practice and methods. In this regard, there is an obvious benefit for exercising in a group (10). Such benefits include; group exercise requiring simple portable equipment and social support for involvement in the program (10). Importantly for the patient with AD, it has previously been demonstrated that these factors are able to effectively discriminate between adherence and dropout (10). Furthermore, group exercise has also proven to result in an enhanced ability for individuals to develop recreational skills, to want to go out with friends, to satisfy curiosity, to release competitive drive, and/or to develop social relationships (10). Individuals participating in group exercise have also reported receiving more encouragement for involvement from their friends and work supervisors than individual exercisers (10).
2. Objectives
This study aimed to determine the effect of low-cost, group-based exercise on mental outcomes in patients with mild to moderate AD.
3. Methods
3.1. Participants
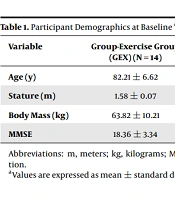
Participants were volunteers with mild to moderate AD based in AD care facilities based in Pretoria, South Africa. The criteria for inclusion in the study required that participants should: have mild to moderate AD as diagnosed by the National Institute of Neurological and Communication Disorders and Stroke and the Alzheimer disease and related disorders association (NINCDS-ADRDA) criteria (11); be cleared for participation in the present study by their supervising physician, and have had no relative or absolute contraindications to exercise and/or testing (12). Participants and/or legal guardians were informed about the characteristics of the study and gave written informed consent. The protocol was designed according to the ethical norms set out in the 1961 Helsinki declaration (modified in Edinburgh in 2000) and was approved by the University of Johannesburg’s Institutional Review Board (REC-01-247-2015). The 40 participants that met the initial criteria were divided at random, using a random numbers table, into one of two groups: a non-exercising control (CON) group or a group-exercise (GEX) group (n = 20 each). Table 1 provides a summary of the mean demographic baseline values of each group. During the eight-week study period, the participants and their caregivers were told to keep to their daily routines, medication usage, and eating habits and not to begin any new exercise regimen. The CON group participants continued with their normal daily routines without participating in any exercise.
| Variable | Group-Exercise Group (GEX) (N = 14) | Non-Exercising Control Group (CON) (N = 20) |
|---|---|---|
| Age (y) | 82.21 ± 6.62 | 78.5 ± 6.69 |
| Stature (m) | 1.58 ± 0.07 | 1.63 ± 0.07 |
| Body Mass (kg) | 63.82 ± 10.21 | 66.64 ± 13.73 |
| MMSE | 18.36 ± 3.34 | 18.8 ± 5.77 |
Abbreviations: m, meters; kg, kilograms; MMSE, Mini-Mental State Examination.
aValues are expressed as mean ± standard deviation
3.2. Procedures
Principal continual measures of mental function using standardized assessment scales were utilized to evaluate the participants at baseline, at four-weeks, and at the eight-week follow-up. The specific intervals used for evaluation were due to the progressive nature of AD that increases the probability of cognitive deterioration as the duration of the intervention progresses (13). The eight-week training intervention was implemented to determine the effects of group-based exercise on cognitive function, abilities to perform activities of daily living (ADL’s), and QOL in patients with AD.
Baseline, mid-, and post-assessment of cognitive function was performed using the Mini-Mental State Examination (MMSE) (14). The MMSE measures mental status and cognitive impairment and uses a score out of 30. The MMSE has 11 tasks that are performed, and a score higher than 23 on this assessment indicates better cognitive function, with 30 being the maximum achievable score. It is utilized to assess orientation, attention, registration, calculation, recall, and language (15, 16).
The Alzheimer’s Disease Cooperative Study Activities of Daily Living (ADCS-ADL) Scale was used to assess each participant’s ability to perform ADLs (17). The ADCS-ADL was administered in questionnaire format. All responses were related to the four weeks prior to the time of rating. The six basic ADL items each select an ADL and provide descriptions of level of competence, with the rater selecting the most appropriate option. The ADCS-ADL uses a score out of 78 and consists of 23 basic and instrumental ADL’s used in mild to moderate AD (18).
Quality of life was assessed by using the Quality of Life-Alzheimer’s Disease (QOL-AD) Scale (19). This scale has 13 items measured using a four-point scale, with one being poor and four being excellent. Total scores range from 13 to 52. This scale was specifically designed to be used on individuals with dementia and measures aspects related to QOL. The QOL-AD assesses aspects concerned with the individual's distress about finances, relationships with friends, and family, physical condition, and mood (19).
3.3. Intervention
All participants participated in a similar eight-week exercise training program that consisted of approximately 45 minutes of exercise performed non-consecutively for three days per week (20). The sessions for the GEX began with a five-minute warm-up period involving five gentle range of motion exercises, such as trunk rotations, performed for three sets of 10 repetitions each, followed by 15 minutes of resistance training using 12 exercises, such as straight leg raises, performed with elastic bands and Pilates balls for 3 sets of 10 repetitions each (21, 22). The participants then went on to perform nine aerobic exercises, such as marching on the spot, for approximately 15 minutes, followed by five minutes of flexibility exercises using five exercises, such as a neck/levator scapulae stretch, followed by a final one-minute cool-down consisting of inhalation and exhalation accompanied by relaxing arm movements (21). All intensities for the resistance, aerobic, and flexibility portions of the program were self-selected, which allowed for individual adaptation and progressive overload (20, 21). This multidimensional exercise intervention program was implemented in order to improve the components of functional capacity and frontal functional cognition (23). In each training session, the caregivers and/or legal guardians were required to be on standby but were not required to be present. Each session was fully supervised by the same researcher.
3.4. Statistical Analysis
Data were analyzed using the statistical package for social sciences (SPSS) Version 25.0 for Windows (IBM Corporation, Armonk, NY) and included the calculation of the descriptive statistics, including the means and standard deviations of the data obtained. The normality of the distribution of the data for each group and variable studied (including pre-and post-test [prepost] differences) was evaluated by the Shapiro-Wilk test, and equal variance was determined by the Levene's test. When normality was found in the measured variable, parametric tests were performed to assess within-group and between-group differences. Differences in prepost evolution for each group and variable were examined using Student’s t-test for paired samples. Alpha levels were set at P ≤ 0.05 for statistical significance.
4. Results
The adherence rate to the GEX intervention was 93%. Of the 40 volunteers who fulfilled the initial criteria, six were eventually excluded from the study, with the specifics being: participant 15 (2% adherence) and 16 (2.9% adherence) dropped-out due to lack of interest, participant 17 (100% adherence) dropped out due to admission to hospital with pneumonia, participant 18 (67% adherence) dropped out due to bronchitis and possible pneumonia, participant 19 (58% adherence) and 20 (67% adherence) dropped out due to a severe reduction in functionality and increased weakness not related to the study in the last three weeks.
The GEX and CON were found to be homogeneous (P > 0.05) for their MMSE (P = 0.144) and QOL-AD (P = 0.388) scores at pre-test, but heterogeneous for their ADCS-ADL scores (P = 0.047) (Table 2). The exercise intervention resulted in a significant (P ≤ 0.05) 10.89% increase in MMSE scores across the eight-week intervention period (P = 0.023), with no change in MMSE scores observed in the CON (P = 0.315). While the eight-week exercise intervention failed to elicit any change in the ADCS-ADL scores (P = 0.574), the CON demonstrated a significant, but deleterious 13.77% decrease in their ADCS-ADL scores from pre- to mid-test (P = 0.023) and from pre- to post-test (P = 0.038). Both the GEX and CON were found to have no significant (P > 0.05) changes in their QOL-AD scores across the entire eight-week study period.
| Variable | Group-Exercise Group (GEX), N = 14 | Non-Exercising Control Group (CON), N = 20 | ||||
|---|---|---|---|---|---|---|
| Pre-Test | Mid-Test | Post-Test | Pre-Test | Mid-Test | Post-Test | |
| MMSE | 18.36 ± 3.34 | 19.43 ± 3.86 | 20.36 ± 4.05*** | 18.8 ± 5.77 | 19.1 ± 6.28 | 18.3 ± 6.26 |
| ADCS-ADL | 33.86 ± 10.03 | 33.64 ± 9.98 | 32.5 ± 8.51 | 38.5 ± 17.26* | 32.05 ± 14.76 | 33.2 ± 13.66*** |
| QOL-AD | 39.5 ± 5.30 | 39.79 ± 3.38 | 40.93 ± 3.93 | 38.55 ± 5.88 | 38.8 ± 6.01 | 38.95 ± 5.23 |
Abbreviations: MMSE, Mini-Mental State Examination; ADCS-ADL, Alzheimer’s Disease Cooperative Study Activities of Daily Living; QOL-AD, Quality of Life-Alzheimer’s Disease.
aValues are expressed as mean ± standard deviation
b* Significant (P ≤ 0.05) difference within-group pre-test compared to mid-test; **Significant (P ≤ 0.05) difference within-group pre-test compared to post-test; ***Significant (P ≤ 0.05) difference within-group pre-test compared to post-test.
5. Discussion
This study aimed to determine the effect of low-cost, group-based exercise on mental outcomes in patients with mild to moderate AD. In this regard, the positive influence of this mode of exercise is clearly demonstrated in terms of improved MMSE scores and a stabilization in ADCS-ADL scores, when compared to the CON, following the eight-week group exercise intervention.
The present study’s eight-week, low-cost, group-based exercise program resulted in a 10.89% improvement in MMSE scores, which agrees with the findings of Cancela et al. (24) who reported significant gains in cognitive function in home-care dementia patients following a 16-week aerobic training intervention. Other 16-week exercise interventions found 4.37% (16) and 26.98% (2) improvements in cognitive function using patients with AD. On the contrary, a 12-week exercise primary aerobic intervention failed to elicit any improvements in memory (3), as did a 24-week walking program fail to significantly impact cognitive function as assessed by the MMSE (25). While Tortosa-Martínez et al. (3) proposed that their lack of change was related to the “short” duration of their study, the results of the present study refute this supposition. However, further analysis of the available literature may indicate that the instructor supervising the exercise may be a factor influencing the successful outcome of an intervention. In this regard, those studies that utilized caregivers in the execution of the exercise were those that failed to elicit a significant change in cognitive function. Another factor that could influence the success of an exercise intervention is duration, since the studies that failed to demonstrate significant changes utilized sessions shorter than the present study’s 45 minutes or those successful studies.
As for ADCS-ADL scores, this study found no improvements following the eight-week, low-cost, group-based exercise program. This is similar to previous findings of ADL performance following a multidimensional community-based 16-week (16) and 24-week passive, motor-assisted, or active resistive training exercise intervention (26). In contrast, a 24-week walking program resulted in improvements in the ADLs (25), as did a multidimensional exercise intervention for 24 weeks (27). What is noteworthy regarding the present study’s findings is that while the eight-week intervention failed to improve ADCS-ADL scores, the control group was found to have significant deleterious deteriorations in their mean ADCS-ADL scores. This indicates that the exercise intervention utilized in the present study may have prevented further deterioration in the exercising participants. A possible explanation for the lack of changes in the present study’s ADCS-ADL scores could be due to the short duration of the intervention program. In addition to the longer durations of the successful studies cited previously, Toots et al. (28) proposed that ADL performance will increase only after longer duration exercise interventions, such as 12 months. Further possible reasons for the lack of positive change in ADL performance scores could be related to the lack of functional activities, lower intensity, and volumes prescribed in the present study’s exercise program.
This study’s eight-week intervention did not have any impact on QOL-AD scores. This was similarly found in a study undertaken by Steinberg, Sheppard Leoutsakos, Podewils, and Lyketsos (29), which used a 12-week multicomponent individual-based physical activity intervention. Also, an additional study reported that a 12-week lower-body resistance training program resulted in no significant changes in behavior (26). However, a 12-week multicomponent program resulted in improvements in depression levels (30). A likely explanation for the lack of changes in QOL-AD may be due to varying interpretations and perceptions of QOL (31). Furthermore, emotional and interactive variability is regularly observed in Alzheimer’s disease patients from day-to-day, which could affect their ability to answer a questionnaire with consistency (9). This assumption is supported by Haley (32), who has noted intra-individual variability in intervention studies in cognitively impaired individuals, particularly within functional assessments, as the stage of cognitive function deteriorates. Haley (32) also indicated that cognitive variance is one main factor that results in intra-individual variability in dementia patients, which impacts concentration, attention, and stimulation variability. Moreover, even if physical activity enhances physical function and ability, such improvements will not categorically stimulate improvements in behavior, mood, and interaction (29).
5.1. Limitations
The present study had some limitations that should be taken into account in future investigations. In this regard, it is a challenge to include and effectively engage frail cognitively-impaired individuals in exercise programs as a result of their fear of falling, medication effects, illness, fatigue, and/or deconditioning (9). However, these factors were not assessed, and their impact on the present study's results cannot be determined. In addition, it has been reported that cognitive variance is a major factor that could result in intra-individual variability in dementia patients, which results in concentration, attention, and stimulation variability (32). Again, concentration, attention, and stimulation variability were not assessed in the present study, and their impact on the findings cannot be determined. In addition, motivation from the home-care facility, family members, and caregivers differed, which could not only have influenced adherence rates but also the effort put in by the participants.
5.2. Conclusions
In conclusion, the present study found that a low-cost multidimensional group exercise intervention can positively influence cognitive function and stabilize patients' ability to perform ADL's in patients with mild to moderate AD, but not QOL. Thus, we advise that a multidimensional group exercise program be included as an adjunct lifestyle approach in the early stages of AD in combination with other treatments in patients with AD to prevent and/or delay the progression of AD.