1. Background
Sedentary behavior refers to any waking activity characterized by an energy expenditure ≤ 1.5 metabolic equivalents and a sitting or reclining posture (1). Sedentary behavior, which includes sitting and TV viewing, has been identified as an independent risk factor for several diseases such as type 2 diabetes and cardiovascular disease (2). In addition, 1 day of inactivity has been shown to deteriorate insulin function in healthy adults (3). Some experimental studies have reported that interrupting prolonged sitting improves postprandial glycaemia and insulinemia in both healthy and overweight/obese adults (4, 5). Notably, a recent cohort study has shown that daily standing declined mortality rates in Canadian adults (6). However, the precise mechanisms underlying these physiological responses are unclear.
Nonetheless, one of the possible mechanisms underlying the improvement in metabolism by interrupting prolonged sitting may be the reduction of oxidative stress (7). In fact, metabolic disorders, including hyperglycemia and hyperinsulinemia, are caused by the endothelial dysfunction through elevation of oxidative stress (8). Several researchers have reported that acute exercise and exercise training status improve postprandial oxidative stress (9, 10). However, little is known regarding the effects of interruption of sedentary behavior by standing or acute exercise on postprandial oxidative stress in humans.
2. Objectives
The purpose of the present study was to examine the effects of breaking sitting by standing and acute exercise on postprandial oxidative stress in humans.
3. Patient and Methods
3.1. Participants
Fifteen healthy young men (range age, 23 - 30 years) of normal weight participated in this study after giving written informed consent. The physical characteristics of participants were as follows (expressed as means ± standard error (SE)): height 1.73 ± 0.05 m, body mass 67.7 ± 5.3 kg, and BMI 22.5 ± 1.5 kg/m2. This study was conducted according to the guidelines laid down in the Declaration of Helsinki and was approved by the ethics committees of Waseda University. The inclusion criteria were: non-smoker, no known history of cardiovascular disease, body mass index (BMI) < 30 kg/m2, and no intake of medication or antioxidant supplements.
3.2. Main Trials
The protocols used in the present study have been published previously (11). A randomised crossover design was used. Each participant performed 3 trials (sitting, standing, and exercise), each lasting 2 days, in a randomised order. The trials were separated by more than 1 week. In addition, the participants were asked to remain inactive on the day before day one of each trial and throughout the main trial (other than the exercise performed as part of the experiment). In addition, they were asked to refrain from any structured exercise (i.e. no physical activity beyond activities of daily living) and alcohol intake on the day before each trial and to maintain their normal diet during the experimental period.
On day one of each of the main trials, the participants arrived at the laboratory at 08:15 hours after 10-hours overnight fast (no food or drink except water). After a 15-minute rest period, a fasting venous blood sample was collected by venipuncture with the participant in a seated position. For the sitting trial, participants sat in a chair (reading, writing, working at a computer, or watching television) in the laboratory until 16:00 hour. For the standing trial, the participants stood 6 times, for a 45-minute period each time. For the exercise trial, the participants walked or ran at approximately 60% of age-predicted maximum heart rate on a treadmill for 30 minutes in the afternoon (15:15 – 15:45 hour). After each trial, a second venous blood sample was collected by venipuncture with the participant in a seated position. During every trial, a standardised breakfast and lunch were given at approximately 9:00 hour and 12:00 hour, respectively.
On day two of each main trial, the participants arrived at the laboratory at 08:15 hours after an overnight fast of at least 10-hours (no food or drink except water). After a 15-minute rest period, a fasting venous blood sample was collected by venipuncture with the participant in a seated position. Participants then consumed a standardised breakfast. The time when the participants began eating was recorded, and they were required to rest (i.e. reading, writing, working at a computer, or watching television) in the laboratory for 6 hours after the initiation of breakfast. A second test meal (identical to the first meal) was consumed 3 hours after the initiation of breakfast. Further venous blood samples were collected at 2, 4, and 6 hours after the initiation of breakfast.
3.3. Test Meals
On the day before each trial (i.e. day one), all participants replicated their dietary intake from the first trial on their subsequent trials. Thus, meals were standardised across trials, on day one. Average of energy intake for day one of the trials was 8.4 ± 1.9 MJ (29 ± 9% from fat, 59 ± 9% from carbohydrate, 12 ± 3% from protein).
The test meal of day two was prescribed according to the participants’ body mass; it provided 0.35 g fat, 1.17 g carbohydrates, 0.29 g protein, and 38 kJ energy per kilogram body mass. Of the total energy derived, 35% was from fat, 52% from carbohydrates, and 13% from protein. This macronutrient composition was set to mimic a real life meal, so that the macronutrient composition of the test meal reflected the background diet consumed by about two-thirds of Japanese adults (12).
3.4. Blood Collection and Analysis
For measurement of serum blood markers, the samples were allowed to clot for 30 minutes at room temperature and then centrifuged at 3000 rpm for 10 minutes at 4°C. The obtained serum was dispensed into plain microtubes and stored at -80°C until the assay. For measuring plasma blood markers, the blood samples collected into tubes containing ethylenediaminetetraacetic acid (EDTA) were immediately centrifuged and stored at -80°C until the assay.
Serum concentrations of derivatives of reactive oxygen metabolites (d-ROMs) and the biological antioxidant potential (BAP) were measured using assay kits from Diacron (Italy). Plasma concentrations of thiobarbituric acid reactive substances (TBARS), along with superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPX) activity were measured using assay kits from Cayman Chemicals (USA). Concentrations of plasma thioredoxin (TRX; Immuno-Biological Laboratories Co., Ltd., Japan) were measured by enzyme-linked immunosorbent assay.
3.5. Statistical Analysis
Data analysis was performed using PASW Statistics 18 software (SPSS Japan Inc., Japan). The Kolmogorov-Smirnov test was used to check for normality of distribution of all blood parameters. The distribution of these parameters did not differ significantly from normal. One-factor analysis of variance (ANOVA) was used to examine differences in the fasting state on days one and two. Moreover, one-factor ANOVA was used to examine differences among the 3 trials for all parameters on day one. A two-factor within-participants repeated measures ANOVA was used to test the main effects of trial, time, and the interaction between time and trials (sitting, standing, and exercise) on day two. When significant interaction effects were detected, we used the Bonferroni method for post-hoc comparisons. All data were reported as means ± SE. Statistical significance was set at P < 0.05.
4. Results
4.1. Fasting Oxidative Stress Markers on Days 1 and 2
In the fasting state on days one and two, there were no statistically significant differences in the concentrations of serum d-ROMs, BAP, plasma TBARS, TRX, and the activities of SOD, CAT, and GPX among trials (Tables 1 and 2). In addition, there were no statistically significant differences in all parameters in the fasting state on days one and two.
| Parameters | M | A | P Value (Trial) |
|---|---|---|---|
| BAP, μM | 0.931 | ||
| Sitting | 2669 ± 69 | 2574 ± 77 | |
| Standing | 2608 ± 62 | 2559 ± 101 | |
| Exercise | 2657 ± 55 | 2572 ± 61 | |
| SOD, U/mL | 0.015 | ||
| Sitting | 12.8 ± 1.5 | 14.5 ± 1.5 | |
| Standing | 14.0 ± 1.3 | 14.3 ± 1.3 | |
| Exercise | 13.5 ± 1.3 | 12.2 ± 1.3 | |
| CAT, U/mL | 0.556 | ||
| Sitting | 51.3 ± 4.2 | 58.2 ± 5.6 | |
| Standing | 49.9 ± 3.5 | 57.6 ± 5.4 | |
| Exercise | 54.3 ± 4.5 | 66.2 ± 5.7 |
a Data are presented as mean ± SE, and N = 15.
b M, morning on days one; A, afternoon on days one.
c Data were analyzed using one-factor ANOVA.
| Parameter | 0 Hour | 2 Hours | 4 Hours | 6 Hours | P Value | ||
|---|---|---|---|---|---|---|---|
| Trial | Time | Trial × Time | |||||
| BAP, μM | 0.882 | 0.001 | 0.594 | ||||
| Sitting | 2600 ± 51 | 2387 ± 64 | 2436 ± 72 | 2459 ± 72 | |||
| Standing | 2621 ± 63 | 2332 ± 67 | 2440 ± 84 | 2527 ± 68 | |||
| Exercise | 2643 ± 70 | 2402 ± 61 | 2408 ± 75 | 2507 ± 54 | |||
| SOD, U/mL | 0.664 | 0.370 | 0.548 | ||||
| Sitting | 13.7 ± 1.2 | 14.1 ± 1.5 | 13.3 ± 1.3 | 13.7 ± 1.5 | |||
| Standing | 13.5 ± 1.4 | 14.2 ± 1.3 | 14.2 ± 1.4 | 13.8 ± 1.4 | |||
| Exercise | 13.1 ± 1.1 | 13.7 ± 1.2 | 13.4 ± 1.2 | 14.4 ± 1.2 | |||
| CAT, U/mL | 0.168 | 0.010 | 0.143 | ||||
| Sitting | 49.3 ± 5.7 | 53.6 ± 3.5 | 59.9 ± 4.7 | 64.2 ± 5.1 | |||
| Standing | 50.5 ± 3.0 | 64.7 ± 5.5 | 62.4 ± 4.5 | 56.6 ± 3.9 | |||
| exercise | 55.5 ± 5.2 | 65.4 ± 6.5 | 71.4 ± 6.9 | 60.0 ± 4.8 | |||
a Data are presented as mean ± SE, and N = 15.
b Data were analyzed using two-factor ANOVA.
4.2. The Change of Oxidative Stress Markers on Day 1
One-factor ANOVA revealed that there were no statistically significant differences between morning and afternoon in the concentrations of serum d-ROMs, BAP, plasma TBARS and TRX as well as the activities of CAT, and GPX irrespective of trials on day one. For plasma SOD activities, one-factor ANOVA revealed that there was a main effect of trial (P = 0.015) (Table 1). Post-hoc tests showed that the change of plasma SOD activities in the sitting trial was significantly higher than those in exercise trial (P = 0.013).
4.3. Postprandial Oxidative Stress Markers on Day 2
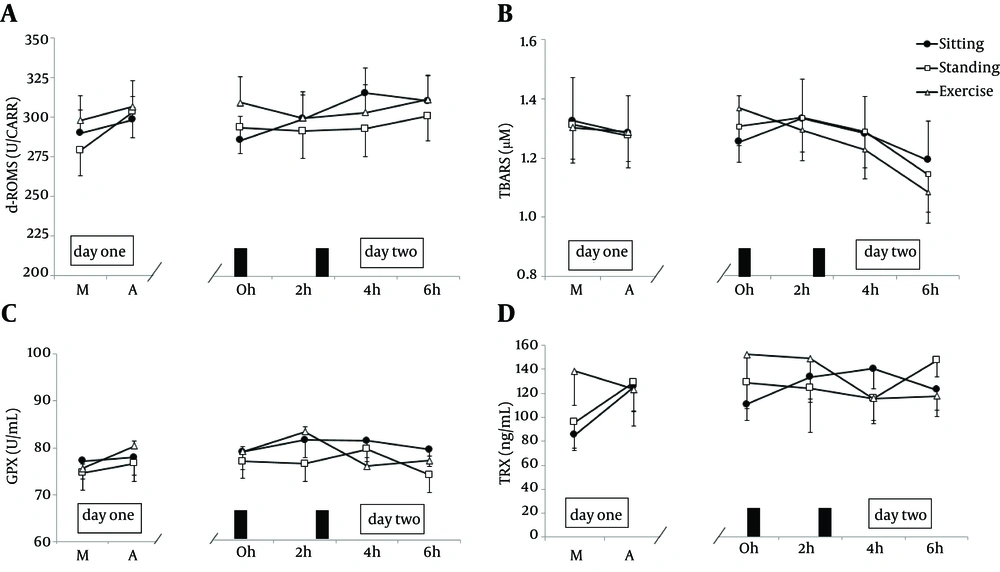
For serum d-ROMs concentrations, two-factor ANOVA revealed a significant trial × time interaction (P = 0.006) (Figure 1). Post-hoc tests showed that serum d-ROMs concentrations at 4 hours (P = 0.064) and 6 hours (P = 0.071) were higher than those in the fasting state (0 hour) in the sitting trial. For plasma TBARS concentrations, two-factor ANOVA revealed that there was a main effect of time (P = 0.001) (Figure 1).
(A), plasma thiobarbituric acid reactive substances (TBARS) (B), activity of glutathione peroxidase (GPX) (C), and concentrations of plasma thioredoxin (TRX) (D) measured on days one and two during the sitting, standing, and exercise trials (n = 15). Data were presented as means ± standard error (SE). M, morning on day one; A, afternoon on day one. The black rectangles indicate the times when the test meals were consumed. Data were analysed using two-factor ANOVA. The main effect of trials (d-ROMs, P = 0.352; TBARS, P = 0.850; GPX, P = 0.228; TRX, P = 0.904), time (d-ROMs, P = 0.116; TBARS, P = 0.001; GPX, P = 0.083; TRX, P = 0.738), and trials × time interaction (d-ROMs, P = 0.006; TBARS, P = 0.093; GPX, P = 0.017; TRX, P = 0.049).
For serum BAP concentrations, two-factor ANOVA revealed that there was a main effect of time (P = 0.001). For plasma GPX activity, two-factor ANOVA revealed a significant trial × time interaction (P = 0.017). Post-hoc tests revealed no significant change of GPX activity across trials or time. In addition, two-factor ANOVA revealed that there was a main effect of time (P = 0.001) for plasma CAT activity. For plasma TRX concentrations, two-factor ANOVA revealed a significant trial × time interaction (P = 0.049). Post-hoc tests revealed no significant change in TRX concentrations across trials or time. There were no statistically significant differences in plasma SOD activity, irrespective of trials or time.
5. Discussion
To the best of our knowledge, the present study is the first to examine the effects of reducing sitting on postprandial oxidative stress in humans. The main finding of the present study was that day one of sitting elevated the postprandial oxidative stress on the next day, but standing and acute exercise prevented an elevation of the postprandial oxidative stress markers.
It is well documented that elevation of postprandial oxidative stress markers decreases after acute exercise (13, 14). In the present study, acute exercise performed one day prior has prevented to elevate the postprandial oxidative stress markers on the next day. Notably, acute standing one day prior also hampered the elevation of postprandial oxidative stress markers. Recently, a cohort study showed that a longer standing period is associated with a lower risk of mortality (6). As for the underlying mechanisms, there is a report that the removal of intermittent standing and ambulation in rats by hind limb suspension resulted in decreased lipoprotein lipase (LPL) activity, triglyceride uptake into red skeletal muscle, and lead to a reduction in the HDL-cholesterol (15). However, another study has reported that postprandial lipaemia status, including inactive monomeric LPL protein level, was not statistically changed after experimentally-induced standing (11). These findings indicate that the effects of standing on postprandial oxidative stress responses may be different from postprandial metabolic responses. In addition, one review has suggested that sedentary behavior or sedentary activity results in low shear stress in the lower extremities, which may result in increased oxidative stress and impaired endothelial function (16). Thus, standing may be effective for improving postprandial oxidative stress by elevating shear stress.
Another possible factor underlying the attenuation in postprandial oxidative stress may be the endogenous antioxidant capacity, including that of enzymatic and non-enzymatic antioxidants. In the present study, acute sitting on day one showed the elevation of SOD activities. Moreover, various activities day one prior observed the different responses of the postprandial antioxidant capacity (i.e. GPX activity and TRX) on next day. Notably, TRX, which is an antioxidant protein, differed in each trial. TRX plays an essential role in protection by limiting oxidative stress directly via its antioxidant effects (17). This finding indicates that TRX adaptation was caused by elevated postprandial oxidative stress in the sitting trial. On the other hand, a statistically significant relationship between change in oxidative stress and antioxidant capacity markers has not been observed. There is some evidence demonstrating that oxidative stress is attenuated by the antioxidant capacity (18, 19). Therefore, the underlying mechanisms of reduction of postprandial oxidative stress by standing and acute exercise require further study including other antioxidant capacity markers.
In conclusion, our study suggests that various activities at one day prior may attenuate postprandial oxidative stress.