1. Background
High-intensity of exercise or unaccustomed eccentric exercise can cause the phenomenon of Exercise-Induced Muscle Damage (EIMD) which usually results in cramps, muscle strain, impaired muscle function and delayed-onset muscle soreness (1). Delayed-Onset Muscle Soreness (DOMS) is a common neuromuscular condition which is characterized by endogenous muscle hyperalgesia that affects individuals on the day after they perform vigorous or unaccustomed eccentric exercise (2). Some activities, such as walking down stairs, downhill running, stepping and weight training, are composed of eccentric muscle contraction which has the potential to initiate greater injury to muscles (3).
Clinicians and coaches are concerned with EIMD as it may interrupt the sports performance, phase of training and the rehabilitation process of the individual. Therefore, preventative intervention for EIMD is important as it may minimize the treatment cost, duration of rehabilitation along with prevention of further muscle injury. In addition, it allows the individual to continue with exercise training and competition (2-4). Several methods are in practice to prevent or reduce EIMD such as physical modalities, massage and warm-up. Evidence suggests that increasing tissue temperature can reduce muscle tension and promote flexibility (4, 5). Therefore, pre-conditioning of muscle hyperthermia may lessen the muscle damage during eccentric exercise.
Previous studies by Nosaka et al. (6) and Brock Symons et al. (7) showed that an application of local heat (i.e. microwave or ultrasound) to the specific body’s region provided a minimal effectiveness in prevention of muscle damage. Application of a localized heat to muscle in a small area may not affect the whole body systems. It has also been mentioned that a systemic warm-up such as sauna could contribute to the body’s systems (i.e. circulation, immune, hormone, neuromuscular and body regulation) more than the local hyperthermia method (8-10). It is renowned that sauna bathing is a systemic hyperthermia method which is normally applied to the whole body, with the aim of warming up muscles, increasing tissue temperature, and promoting body adaptation (8). Many reports have suggested that sauna is able to promote blood flow to muscle and raise plasma endorphin level which may enhance immunosuppressive anti-inflammatory functions (10, 11). However no previous studies have investigated the prophylactic effect of sauna on muscle damage.
2. Objectives
Much of the research into preventive measures and effective treatment of muscle damage has concentrated on the big muscles only, for example, biceps and quadriceps (6, 7, 12, 13). However EIMD may present with varied responses across different muscle groups (1, 14-16). In spite of wrist extensors playing an important functional role in activities of daily living such as grasping, carrying, very limited research has addressed on the EIMD of forearm muscle group. Therefore, the main aim of this study is to investigate the prophylactic effects of sauna towards the symptoms associated with EIMD from eccentric exercises of wrist extensor muscle group.
3. Patients and Methods
This was a quasi-experimental study design that considered the factor of time (changes within subjects from baseline throughout the follow-up period) and factor of conditions (differences between control group and sauna group). Based on a pilot study, the primarily clinical outcomes of DOMS (i.e. PPT, GS, WES) was determined for obtaining the power of 0.80 at alpha level of 0.05 with effect size of 0.84, total sample size estimation would be approximately 14 subjects per group (17). A total of twenty-eight male volunteers from the campus and community settings (mean age 20.9 years, SD = 1.6; mean height 171.6 cm, SD = 5.1; mean body weight 61.6 kg, SD = 9.1) participated in this study. Only male participants were recruited in this study due to the fact that gender affects pain outcome measures (18-21). The subjects were divided into 2 groups (14 subjects in control and 14 subjects in sauna group). The control and experimental conditions were a block randomization equally with drawing lots (concealed envelop), which was run by an assistant researcher. All of the subjects were recruited by pre-defined selection criteria. The inclusion criteria were that the subjects must have no history of musculoskeletal and neurological disorders in upper limb and they must not be practicing any form of arm resistance training at least 3 months prior to the participation of this trial. Any subjects with contraindications to sauna therapy such as severe myocardial infarction, kidney disease, fever, and anemia were not recruited. All of the subjects were briefed about the study procedure and written informed consents were obtained from all the subjects prior to the commencement of the trial. An institutional ethical committee gave ethical approval for this study.
3.1. Study Procedures
The subjects were familiarized to the measurement and study procedures (Figure 1). All the subjects were requested not to take any form of exercises or any medications during the study period. Sauna was administered for the subjects in the sauna group prior to EIMD. Each subject was asked to shower before entering a sauna room, size of 3 m2. The subject was seated and rested on the bench with a towel. The sauna temperature was kept between 170 - 180°F (76.67 - 82.22°C) at a comfortable level of relative humidity (15 - 30%) for 15 minutes (11, 22). In the control group, EIMD was induced with a Contrex dynamometer (CON-TREX Multijoint System, CMV AG manufacture, Switzerland). Subjects were seated on the chair of the dynamometer, and the trunk was fully supported and fixed by the double-cross chest belts. The forearm to be tested was placed on the handle bar in 90° pronated position, and the subjects were asked to maximally resist against the dynamometer’s movement from wrist extension to wrist flexion. The subjects were verbally encouraged to generate maximal force for the whole range of wrist extending motion, and the force output was displayed on a screen to motivate maximal effort. It required the subjects to passively extend the wrist at a speed of 100°/second and to flex the wrist at a speed of 25°/second. The protocol included five sets of exercise. In each set, the subjects were required to complete 60 repetitions in 5 minutes. There was a one-minute rest period between each set (23). Peak torque and total work during eccentric exercise of the subjects were collected by the average of 5 sets of 60 repetitions using the software program of Contrex dynamometer. All the variables were measured at baseline, immediately after EIMD induction and for the next 8 consecutive days. Additionally, each subject’s blood flow and skin temperature was measured using a Laser Doppler Blood Flow Monitor (Moor instruments DRT4, UK) and a skin temperature biofeedback (Myomed 342 Enraf Nonius, USA) respectively over the muscle belly of ECRB for a duration of 5 minutes before starting the experiment to serve as baseline data and re-measured again immediately after completion of the experimental condition. The room temperature was controlled at 25.0 ± 1.0 °C during the whole process of the measurement procedures.
3.2. Outcome Measures
PI and PPT were quantified as outcome variables of pain threshold. The PI was measured using a Visual Analog Scale (VAS) to rate the intensity of average pain. Subjects were asked to rate their perceived level of pain during movement of the wrist. For Pressure Pain Threshold (PPT), it was quantified through an incremental raise of the pressure stimuli applied at a rate of 30 kPa/second using a pressure Algometer (Somedic Production, Algometer type II, Sweden) with a probe of 1.0 cm2. The subjects were instructed to press the button of the control switch to report the pain when the applied pressure was painful. The PPT were measured at the same spot with forearm in the position of 0° elbow extension and 90° pronation pronated while the subject is in supine lying. The PPT was assessed 3 times for each site, and the mean of the 3 trials was used for further analysis (22). Passive range of motion of wrist flexion (PF-ROM) and extension (PE-ROM), Grip Strength (GS) and Wrist Extension Strength (WES) were measured as outcome variables of wrist extensor muscle function. An electrogoniometer (Biometrics, DLK 900, U.K.) was used to measure the pain-free passive range of motions at wrist. The position for measuring range of motion was standardized between subjects and from day to day. Subjects sat on an arm supporting chair, and were asked to rest their arms on the support. The shoulder adducted by side and neutrally rotated, with forearm in 90° pronation and the elbow in 45° flexion. The placement of goniometer was followed as per an established protocol (24). The subjects performed the active movements at wrist while the investigator performed the passive movements at wrist into flexion and extension. The subject was instructed to report the point of onset of pain during movements and the joint range at wrist was measured instantly.
Grip strength was evaluated with an electronic digital hand dynamometer (Model MLT003/D, Power lab, Australia) as per the recommendations of American Hand Society of Hand Therapists (25). The grip width of the hand dynamometer was standardized for each subject throughout the study. The GS was measured with maximal isometric contraction for 6 seconds with the subjects feeling no pain (i.e. pain free GS) during the grip test (26). The protocol for measuring WES is explained below. While the subject was sitting in a chair facing against the table, the upper extremity was positioned with forearm in 90° pronation and 45° elbow extension. A force transducer (Model MLT003/D, Power lab, Australia) was attached on a platform under the table to measure the WES. From the initial position of 20° extension at wrist, the subjects were asked to press the centre of force transducer with the 3rd knuckle up to the point of onset of pain (i.e. pain-free WES) (26). All of the measurements were performed 3 times with 1 minute between trials, and the mean of 3 trials for peak values was used for further analysis.
The order of measurements was pain intensity, PPT, ROM-AF, ROM-PF, and ROM-PE. Muscle strengths were measured after these measurements with randomization between GS and WES. The same investigator conducted all measurements in a single-blinded manner. All outcome measures including PPT, ROM, grip and wrist extension strength, were considered to be reliable with a 24-hour interval of test-retest (ICC > 0.85) (27).
3.3. Statistical Analyses
The absolute values of VAS and LS were used for analysis of PI. The normalized percentages of pre-exercise values (i.e. normalized to pre-exercise) were used for analysis of PPT, ROM, GS and WES. The results were analyzed using a mixed model of two-way repeated measures ANOVA (group × time). Turkey post-hoc test analysis was followed up to detect differences in the main effect (groups or time). Statistical significance was set at P < 0.05 for all analyses.
4. Results
4.1. Subject Characteristics
All the 28 subjects in the study were right hand dominant and attended regular exercise of no more than 2 times per week. Both groups were relatively similar in the mean age, height, weight, and baseline values for most of the outcome measures (Table 1). All 28 subjects completed the follow-up sessions as allocated. There was no difference in the average of maximal torque and total work during induction exercises between the control and the sauna groups. Averages of maximal torques in the control and sauna group were 3.25 ± 0.87 N and 4.01 ± 1.30 N, respectively. Averages of total work in control and sauna group were 180.03 ± 56.61 J and 175.78 ± 73.09 J, respectively. There was a significant increase in blood flow measurements taken before sauna (14.91 ± 9.18 flux/minute) and after sauna applications (70.15 ± 30.74 flux/minute) (P < 0.001). The skin temperature was also significantly different between before (31.76 ± 1.30°C) and after application of sauna (33.85 ± 1.40°C) (P < 0.001).
| Characteristics | Control Group b | Sauna Group b |
|---|---|---|
| Age, y | 21.07 ± 1.59 | 20.64 ± 1.69 |
| Height, cm | 173.14 ± 5.14 | 170.07 ± 4.76 |
| Weight, kg | 61.29 ± 9.55 | 61.93 ± 9.03 |
| Pressure pain threshold, kPa | 300.17 ± 112.00 | 476.52 ± 111.06 |
| Range of motion [passive flexion] (degrees) | 98.86 ± 6.74 | 92.57 ± 12.65 |
| Range of motion [passive extension] (degrees) | 98.17 ± 8.69 | 111.26 ± 12.36 |
| Grip strength-Pain free (N) | 300.96 ± 50.06 | 303.39 ± 78.25 |
| Wrist extension strength-Pain free (N) | 96.29 ± 26.18 | 75.82 ± 17.36 |
a There were homogenous of all subjects’ characteristics for both groups (P > 0.05), excepted in pressure pain threshold, passive extension, and wrist extension strength. Therefore, normalized data was used for further analysis.
b N = 14.
4.2. Main Outcomes
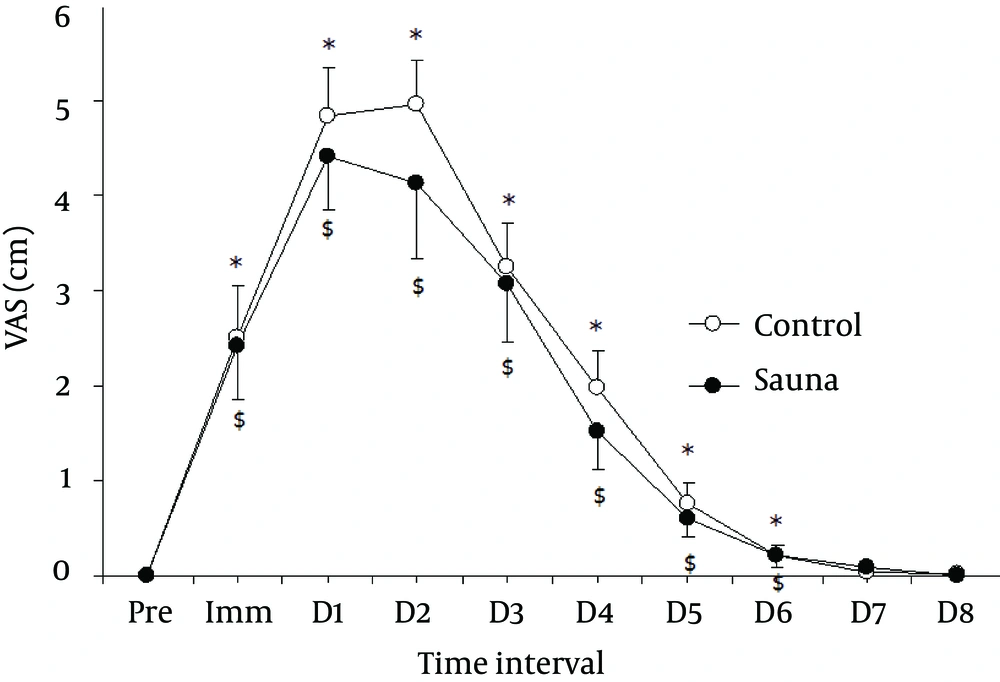
The changes in the variables (Mean ± SD) and mean difference between groups (95% CI) for the outcome measures before (pre), immediately (Imm) and 1 - 8 days following eccentric exercise between sauna (n = 14) and control (CON; n = 14) groups were shown in (Table 2). The sauna group showed a trend of lesser pain intensity when compared to control group (P = 0.47; power = 0.11) (Figure 2).
| Pre | Imm | Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | Day 7 | Day 8 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Visual Analog Scale (VAS) [10 cm] | ||||||||||
| CON | 0 | 2.5 (2.1) a | 4.8 (1.9) a | 5.0 (1.7) a | 3.2 (1.7) a | 2.0 (1.5) a | 0.8 (0.8) a | 0.2 (0.4) a | 0 (0.09) | 0.02 (0.07) |
| Sauna | 0 | 2.4 (2.1) a | 4.4 (2.1) a | 4.1 (3.0) a | 3.1 (2.3) a | 1.5 (1.5) a | 0.6 (0.7) a | 0.2 (0.4) a | 0.1 (0.3) | 0 (0) |
| Mean dif (95% CI) | 0.0 (0.0 – 0.0) | 0.0 (-1.5 – 1.7) | 0.4 (-1.1 – 2.0) | 0.8 (-1.1 – 2.7) | 0.2 (-1.4 – 1.8) | 0.5 (-0.7 – 1.6) | 0.2 (-0.4 – 0.7) | 0.0 (-0.3 – 0.3) | 0.0 (-0.2 – 0.1) | 0.0 (-0.0 – 0.0) |
| Pressure Pain Threshold (PPT), kPa | ||||||||||
| CON | 300.2 (112.0) | 269.3 (103.9) a | 172.2 (80.4) a | 174.3 (82.8) a | 202.8 (79.0) a | 238.2 (83.0) a | 253.7 (79.1) a | 284.5 (116.9) | 292.0 (99.4) | 302.3 (120.8) |
| Sauna | 476.5 (111.0) | 395 (115.4) a | 315.1 (140)a | 360.8 (170.4) a | 391.0 (143.4) a | 410.9 (142.4) a | 431.1 (138.8) a | 448.9 (123.9) | 462.1 (117.4) | 468.9 (119.2) |
| Mean dif (95% CI) | -176.4 (-263 – -89) | -125.9 (-211 – -40) | -142.9 (-231 – -54) | -186.5 (-290 – -82) | -188.3 (-278 – -98) | -172.7 (-263 – -82) | -177.3 (-265 – -89) | -164.3 (-257 – -70) | -170.2 (-254 – -85) | -166.5 (-259 – -73) |
| Range of Motion-PassiveFlexion (ROM-PF) [degree] | ||||||||||
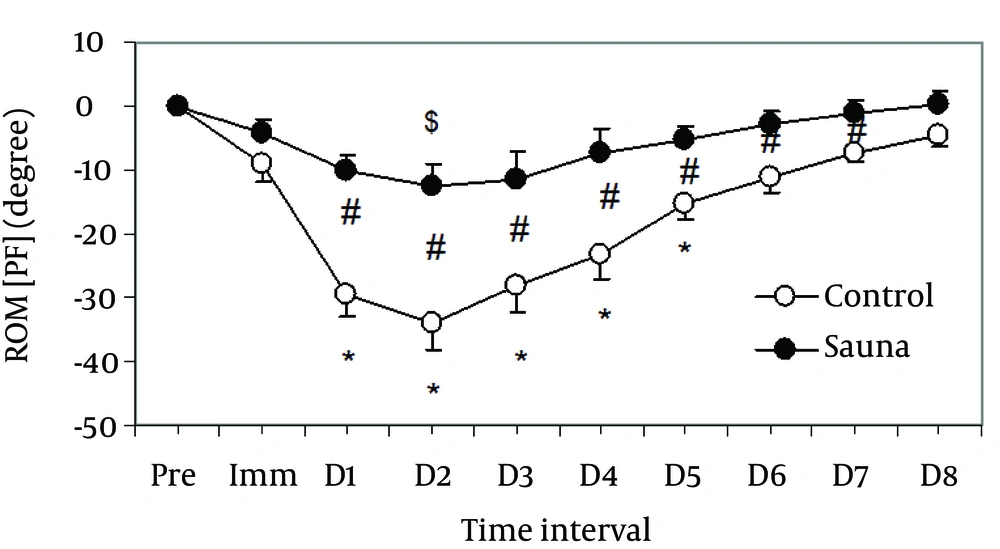
| CON | 98.9 (6.7) | 89.8 (13.3) | 69.2 (10.8) a | 64.6 (13.1) a | 70.6 (11.7) a | 75.7 (12.1) a | 83.7 (9.6) a | 87.6 (11.6) | 91.7 (8.9) | 94.2 (8.8) |
| Sauna | 92.6 (12.6) | 88.2 (13.4) | 82.5 (17.0) a, b | 80.7 (23.0) a, b | 81.1 (23.7) a, b | 85.2 (21.0) a, b | 87.3 (17.1) a, b | 89.9 (15.3)b | 91.5 (14.1)b | 92.9 (13.9) |
| Mean dif (95% CI) | 6.3 (-1.6 – 14.2) | 1.6 (-8.8 – 11.9) | 13.3 (-24.3 – -2.2) | 15.4 (-29.9 – -0.8) | 10.5 (-25.0 – 4.0) | 9.5 (-22.8 – 3.8) | 3.6 (-14.4 – 7.2) | 2.3 (-12.8 – 8.2) | 0.1 (-8.9 – 9.3) | 1.3 (-7.7 – 10.3) |
| Range of Motion-Passive Extension (ROM-PE) [degree] | ||||||||||
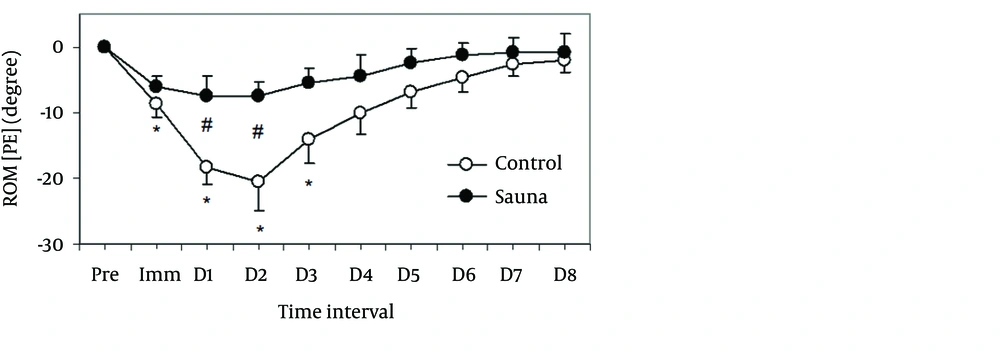
| CON | 98.2 (8.7) | 89.6 (9.9) a | 79.9 (13.4) a | 77.6 (16.9)a | 84.0 (15.4) a | 88.0 (12.4) | 91.4 (12.4) | 93.5 (11.4) | 95.5 (11.3) | 96.2 (12.3) |
| Sauna | 111.3 (12.4) | 105.1 (14.5) | 103.9 (12.4) b | 103.9 (11.7) b | 105.8 (12.6) | 106.9 (15.6) | 108.9 (13.1) | 110.0 (11.8) | 110.5 (10.6) | 110.5 (12.4) |
| Mean dif (95% CI) | -13.1 (-21.4 – -4.8) | -15.5 (-25.2 – -5.9) | -24.0 (-34.0 – -13.9) | -26.3 (-37.6 – -14.9) | -21.7 (-32.8 – -10.7) | -18. (-29.8 – -7.9) | -17.5 (-27.4 – -7.6) | -16.5 (-25.5 – -7.5) | -14.9 (-23.5 – -6.4) | -14.3 (-23.9 – -4.7) |
| Grip Strength-Pain free (GS) [N] | ||||||||||
| CON | 301.0 (50.1) | 156.1 (68.8) a | 155.7 (79.0) a | 152.3 (77.8) a | 208.8 (99.2) a | 236.3 (73.3) | 261.2 (65.8) | 268.7 (75.6) | 284.3 (81.5) | 286.2 (76.5) |
| Sauna | 303.4 (78.3) | 212.8 (65.8) a, b | 221.7 (93.7) a, b | 235.3 (90.2) b | 255.4 (89.9) | 275.9 (82.9 ) | 290.5 (78.6) | 293.1 (68.6) | 293.6 (82.3) | 308.9 (72.8) |
| Mean dif (95% CI) | -2.4 (-53.5 – 48.6) | -56.7 (-109.0 – -4.4) | -65.9 (-133.3 – 1.4) | -82.9 (-148.4 – -17.6) | -46.5 (-120.1 – 27.0) | -39.5 (-100.3 – 21.3) | -29.4 (-85.7 – 26.9) | -24.4 (-80.5 – 31.7) | -9.4 (-72.9 – 54.3) | -22.7 (-80.7 – 35.3) |
| Wrist Extensor Strength-Pain free (WES) [N] | ||||||||||
| CON | 96.3 (26.2) | 37.2 (18.0) a | 32.3 (13.7) a | 36.8 (16.4) a | 52.3 (24.9) a | 67.6 (26.8) a | 71.4 (25.1) a | 75.9 (27.4) a | 82.5 (25.1) a | 88.3 (25.5) |
| Sauna | 75.8 (17.4) | 41.2 (15.3) a | 49.5 (25.7) b | 56.2 (24.5) b | 60.9 (23.9) b | 66.8 (26.9) | 68.0 (26.8) | 73.6 (26.0) | 75.4 (24.9) | 80.5 (32.4) |
| Mean dif (95% CI) | 20.5 (3.2 – 37.7) | -4.0 (-16.9 – 8.9) | -17.1 (-33.1 – -1.1) | -19.5 (-35.7 – -3.3) | -8.6 (-27.5 – 10.4) | 0.8 (-20.0 – 21.7) | 3.5 (-16.7 – 23.7) | 2.3 (-18.4 – 23.0) | 7.0 (-12.3 – 26.5) | 7.8 (-14.8 – 30.4) |
a Significantly different from the baseline (pre).
b Significantly different from CON.
The sauna group demonstrated a significantly lower deficit in PF-ROM than the control group on days 1 - 7 post-exercise (P = 0.029) (Table 2, Figure 3). In addition, a significant lesser deficit in PE-ROM was present in sauna group than the control group on days 1 - 2 post-exercise (P = 0.012) (Table 2, Figure 4).
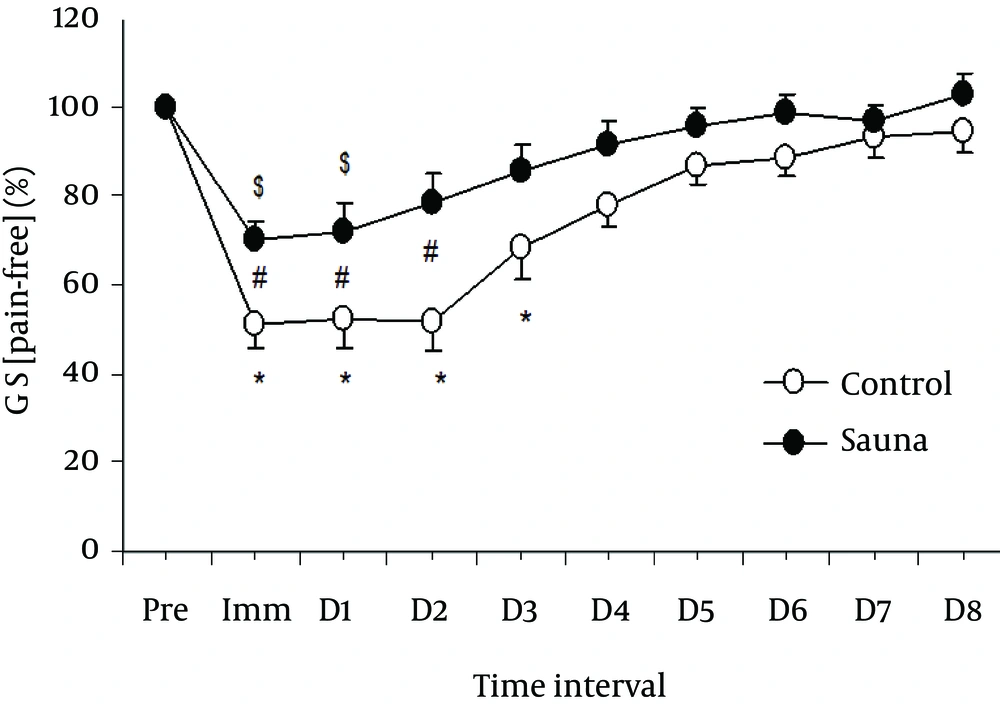
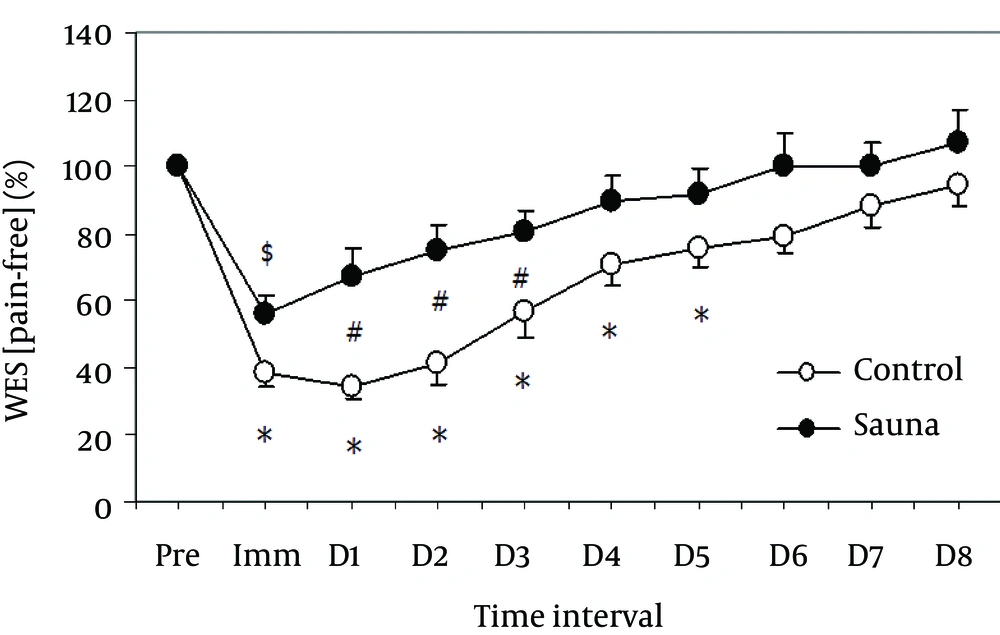
In muscle strength, lesser deficit in GS [pain-free] was observed in sauna group than the control group in the immediate post-exercise period and on days 1 - 2 post-exercise (P = 0.038) (Table 2, Figure 5). Similarly, a significant lesser deficit in WES [pain-free] was noticed in sauna group than that of the control group in the immediate post-exercise period and on days 1 - 3 post-exercise (P = 0.018) (Table 2, Figure 6).
5. Discussion
The main finding of this study demonstrated positive effects of sauna intervention on EIMD symptoms in pain intensity and muscle function. In EIMD, muscle soreness may occur due to either an eccentric action or a concentric function of the muscle in any functional activity (15). In such instances, muscle soreness in EIMD may lead to muscle guarding during clinical examination and in various functional activities of the involved muscle group (14, 28). The subjects in the sauna group when subjected to EIMD demonstrated a significantly lesser deficit in PF-ROM and PE-ROM at wrist when compared to the control group. Thus application of sauna may appear to have a therapeutic effect in reducing EIMD in wrist extensor muscle group. Mild to moderate warming of the whole body promotes capillary dilatation, and relieves muscular spasms related to tonic muscle contraction and pain (29). Perhaps, sauna bathing of the whole body induced sedative, pleasurable and relaxing effects via the sensory nerve endings that might have resulted in lesser deficit in the PF-ROM and PE-ROM among the subjects in sauna group.
Muscle strength is one of the best muscle damage indicators, which is normally reduced after exercise with slow recovery (14). In the current study, GS and WES also demonstrated fewer deficit in sauna group when compared to the control group. It is possible that sauna hyperthermia appears to improve left ventricular function, cardiac output, and vascular activity by increasing blood flow to muscle. This mechanism helps to bring fresh blood, oxygen, nutrition and hormone to the muscle which is crucial for its efficient function (10, 11).
The therapeutic effects of sauna in EIMD claimed by this study is comparable to the previous research in this area. For example, Nosaka et al. (6) found that an increase of pre-exercise muscle temperature using a microwave heat source had no effect on muscle soreness, range of motion and muscle strength. Symons et al. (7) also found no prophylactic effects of raising pre-exercise muscle temperature with continuous ultrasound on the muscle damage symptoms. The localized nature of using microwave and ultrasound as the source of heat on the muscle, in comparison to the results achieved by using a sauna to heat the whole body, suggests the latter has more potential for muscle recovery. However another study by Nosaka (30) has shown that passive hyperthermia treatment 1 day prior to eccentric exercise-induced muscle damage has a prophylactic effect. Previous studies (31, 32) reported that sauna could raise the skin temperature up to 40°C, enhance blood flow to muscle by 0.2 L/minute while the core temperature is relatively unchanged (38°C). The effects of a sauna were increased body temperature, skin blood flow, and muscle relaxation (10). Thus, the application of sauna before exercise warms up the muscles in general and thus prepares the target muscles for the prevention of EIMD symptoms.
Several pieces of evidence explain the benefits of warming up the muscles prior to any form of physical exercises. The therapeutic benefits include decreased resistance of muscle and joints, increased nerve-conduction, increased blood flow, improved carbon dioxide–oxygen exchange rate and enhanced supply of nutrients to the active structures (33). The increased circulation also helps: (a) to clear inflammatory mediators from the muscle tissue; (b) to improve the fatigue characteristics of skeletal muscle during exercise; and (c) to inhibit pain signals as the analgesic properties of increased blood circulation work to relieve muscle damage (34). Increase in tissue temperature and muscle blood flow will diminish muscle viscosity and spasm thereby resulting in smoother muscle contractions (11). The increased amount of elongation can occur without rupture, and potentially offers some protection against strain injury in warmed muscles (34). Evidence also suggests that warm up exercises that increase the tissue temperature make the tissue less sensitive to EIMD (35, 36). Thus the hyperthermia effects of warming up the muscles may explain the therapeutic effects of sauna among the subjects in the study.
Heat has been mentioned for its prophylactic effect on muscle damage by increasing heat shock protein (HSP). This has the advantage of inducing muscle relaxation, decreasing muscle viscosity, and increasing connective tissue extensibility. In this respect HSP would be the explanation of the reduction of muscle damage after eccentric exercise. It has been proposed that HSP helps to stabilize actin and intermediate filaments (the cytoskeleton in skeletal muscle) against stress during or after eccentric contractions (37). Some possible limitations of this study should also be noted. These include the young male population used, which cannot be extrapolated to older age group or women. Secondly, the present study did not measure muscle temperature directly and hence it is not possible to prescribe an optimal heating temperature for the acclaimed therapeutic benefit for the patients. Therefore, assumptions regarding the effect of sauna on muscle temperature as a possible mechanism of action should be considered with caution.
Sauna application has therapeutic effects on the GS, WES, PF-ROM and PE-ROM in the wrist extensor group of muscles. Sauna intervention prior to the exercise provides some benefits to minimize the symptoms and facilitate recovery of EIMD in wrist extensor group of muscles.