INTRODUCTION
The skeleton is a very active organ metabolically as it is in a continuous dynamic remodeling process; maintaining a tightly coupled balance between resorption of old bone and formation of new bone [1]. The balance between bone resorption and formation is influenced by age and the level of strain on the bone generated by muscle contraction during movements. Specific biochemical markers of bone turnover that allow us to estimate bone metabolic processes and have been established, which are useful parameters in assessing changes in bone turnover [1–4].
It has been well recognized that exercise of appropriate intensity and frequency stimulates mineralization and therefore bone gains [5, 6]. It is also equally well recognized that cessation of exercise causes a loss in exercise-induced bone gains in humans [7, 8] and animals [9, 10]. Thus, continuous exercise is needed to maintain exercise-induced bone gains. Currently, there is little information on the minimum quantum of continuous exercise that is required to maintain exercise induced gains in bone physical properties, and equally significantly, it is also unknown if this could be ascertained through the markers of bone metabolism. The present study, therefore, investigated the impact of a series of exercise loads that were lower than the exercise load used to induce bone gains on serum biochemical markers of bone turnover in female rats over a period of 24 weeks. The exercise load used to induce initial bone gains consisted of an 8-week jumping exercise program consisting of 40 jumps per day at 5 days per week. It is hoped that results obtained from this study could help design sub-maximal exercise programs, which could be used for developing age-specific exercise regimens, by formulating guidelines for the enhancement and maintenance of bone health in human beings.
METHODS AND SUBJECTS
Animals Grouping and Exercise Regimen
One hundred and twenty, 12-week old female Wistar Kyoto rats with mean initial body weight of 179.9 ± 10.8 g, were randomized into twelve weight matched groups, with ten rats per group. The groups were as follows: 8 weeks of exercise with a standard training program of 40 jumps per day for 5 days per week (8STP); 8 weeks sedentary control (8S) (S, free cage activity only); 32 weeks sedentary control (32S); and nine groups of rats given 8 weeks of standard training program followed by 24 weeks of continuous exercise (8STP24E), with either maintaining or decreasing the frequency of training days from 5 days per week (5d/w) to 3d/w or 1d/w, and by either maintaining or decreasing the intensity of training from 40 jumps per day (40J/d) to 20J/d or 10J/d (Table 1).
| Groups | n | Standard training program | Training at different frequencies and intensities |
|---|---|---|---|
| 8S | 10 | S | - |
| 8STP | 10 | 5d/w,40J/d | - |
| 32S | 10 | S | S |
| 8STP24E (5d/w,40J/d)(200J/w) | 10 | 5d/w,40J/d | 5d/w,40J/d (200J/w) |
| 8STP24E (5d/w,20J/d)(100J/w) | 10 | 5d/w,40J/d | 5d/w,20J/d (100J/w) |
| 8STP24E (5d/w,10J/d)(50J/w) | 10 | 5d/w,40J/d | 5d/w,10J/d (50J/w) |
| 8STP24E (3d/w,40J/d)(120J/w) | 10 | 5d/w,40J/d | 3d/w,40J/d (120J/w) |
| 8STP24E (3d/w,20J/d)(60J/w) | 10 | 5d/w,40J/d | 3d/w,20J/d (60J/w) |
| 8STP24E (3d/w,10J/d)(30J/w) | 10 | 5d/w,40J/d | 3d/w,10J/d (30J/w) |
| 8STP24E (1d/w,40J/d)(40J/w) | 10 | 5d/w,40J/d | 1d/w,40J/d (40J/w) |
| 8STP24E (1d/w,20J/d)(20J/w) | 10 | 5d/w,40J/d | 1d/w,20J/d (20J/w) |
| 8STP24E (1d/w,10J/d)(10J/w) | 10 | 5d/w,40J/d | 1d/w,10J/d (10J/w) |
These nine exercise groups with different exercise regimens were identified as 8STP24E (5d/w,40J/d) (200J/w); 8STP24E (3d/w,40J/d)(120J/w); 8STP24E (5d/w,20J/d)(100J/w); 8STP24E (3d/w,20J/d)(60J/w); 8STP24E (5d/w,10J/d)(50J/w); 8STP24E (1d/w,40J/d) (40J/w); 8STP24E (3d/w,10J/d)(30J/w); 8STP24E (1d/w,20J/d)(20J/w); 8STP24E (1d/w,10J/d)(10J/w) according to number of jumps per week. The jumping height used in this study was 40 cm. All animals were housed in cages measuring 55cm x 33 cm x 19cm with five animals per cage. They were exposed to a constant 12:12 light/dark cycle and had ad libitum access to water and chow (Gold Coin, Port Klang, Malaysia) throughout the study. Rats in 8S and 8STP groups were euthanized at the end of the eighth week. Rats in 32S, 8STP24S and in the nine exercised groups with different exercise regimens (8STP24E) were euthanized at the end of thirty-two weeks (Table 1). After recording their final body weight (Ohaus Navigator™ Balance, U.S.A.), the rats were anaesthetized with diethyl ether and then decapitated using an animal guillotine (Scientific Research Instrument, U.K.) for collection of blood for subsequent blood biochemical analysis. Tibial bones of the rats’ hind limbs were collected for measurement of bone mass. The experimental protocol was approved by the Research Ethics Committee of Universiti Sains Malaysia.
Jump Training
Jump training was carried out using a previously described protocol [4]. Briefly, each rat in the jumping exercise groups was placed on a metal grid at the bottom of a special wooden box measuring 30.5 x 30.5 x 40 cm in length, width and height respectively. The jumping exercise was initiated by applying an electrical current to the wired floor (electrical grid) of the box through a stimulator. When stimulated, each rat jumped from the floor of the box to catch the top edge of the box with its forepaws. The rat was then immediately returned by hand to the floor of the box to repeat the procedure. The time required per jump was about 4 seconds. After a few days of training, the rats jumped without electrical stimulation. Rats in the sedentary groups were not given any electrical stimulus but to mimic the stress induced by handling before and after jumping exercise, the sedentary rats were also similarly handled for 5 days per week.
Blood Collection and Bone Mass Measurement
At the end of the experimental period, the rats were anaesthetized, one at a time, by placing them for 2-3 minutes in a desiccated jar containing 5 ml of diethyl ether before being decapitated using a small animal guillotine (Scientific Research Instrument, U.K.). Blood was collected from the decapitation site into a 10 ml test-tube through a funnel and left aside for one hour to ensure complete clotting. Serum was then obtained by centrifugation (15 minutes, 3000 RPM, 4oC; Hettich-Rotina 46RS, Germany), after which the serum was divided into three equal portions (approximately 0.5 ml per portion) and stored at -80o C (Heto Ultra Freezer 3410, Denmark) until analysis at a later date.
The tibiae were dissected from the right hind limb. Jumping in these animals depends on forces generated by the hind limb, thus the hind limb tibiae were selected in this study [11]. The tibiae were then wrapped in saline-soaked gauze pads, to prevent dehydration, and put into labelled plastic bags and stored at -80o C for the measurement of bone mass at a later date. For bone mass measurement, the harvested tibiae were immersed in a mixture of chloroform and methanol solvent (2:1 by volume respectively) for one week to remove the fat from the bones [12–14]. Following this the bones were oven dried at 80° C for 24 hours (Isuzu Model 2-2020, Isuzu Seisakusho Co., Ltd., Japan). After drying, the bone weight (to the nearest 0.01 mg) was determined on an electronic balance (ER-180A, A&D Company, Japan).
Blood biochemical analysis
Serum concentrations of osteocalcin and alkaline phosphatase (Alk-Ph) (markers of bone formation) and C-terminal telopeptide of type 1 collagen/1CTP (marker of bone resorption) were estimated in the terminal blood. Serum Alk-Ph was analyzed colorimetrically (Hitachi Automatic Analyzer 912, Bohringer Mannheim, Germany) using commercially available reagent kits (Roche Diagnostics GmbH, Germany). The intra-assay coefficient of variation (CV) values of the assay kits were 0.67%, 0.47%, 0.45% and 0.61% at 63.3, 84.1, 214.7 and 304.7 U.l−1 of serum Alk-Ph concentration respectively, and the inter-assay CV values were 0.67%, 0.27%, 0.47% and 0.67% at 62.0, 85.6, 210.6 and 298.3 U.l−1 of serum Alk-Ph concentration respectively.
Serum osteocalcin was analyzed using a commercially available enzyme-linked immunosorbent assay kit (Nordic Bioscience Diagnostics Rat-MID™ Osteocalcin ELISA, Denmark) and the concentration was determined using a photometric microplate reader (Molecular Devices; Versamax tunable microplate reader, U.S.A.). The intra-assay coefficient of variation (CV) values of the assay kits were 3.6%, 5.0% and 3.4% at 152, 215 and 948 ng.ml−1 of serum osteocalcin concentration respectively, and the inter-assay CV values were 6.6%, 5.5% and 7.7% at 152, 215 and 948 ng.ml−1 of serum osteocalcin concentration respectively.
Serum C-terminal telopeptide of type 1 collagen (1CTP) was analysed using a commercially available enzymeimmunoassay kit (Orion Diagnostica UniQ 1CTP EIA, Finland), and the concentration was determined by a photometric microplate reader (Molecular Devices; Versamax tunable microplate reader, U.S.A.). The intra-assay coefficient of variation (CV) values of the assay kits were 8.1%, 8.9% and 7.6% at 5.6, 13.7 and 26.4 µg.l−1 of serum 1CTP concentration respectively, and the inter-assay CV values were 7.5%, 9.8% and 6.4% at 7.1, 13.1 and 28.2 µg.l−1 of serum 1CTP concentration respectively.
Statistical Analysis
Statistical tests contained in the Statistical Package for Social Sciences (SPSS) Version 10.0 were used for the statistical analysis. One-way analysis of variance (ANOVA) was performed to determine the significance of the differences between groups. When the one-way ANOVA revealed a significant difference, post hoc (least significant difference test) was used to determine the differences between specific means. All data are reported as mean ± standard deviation (SD).
RESULTS
During the experimental period, one of the rats in group 8STP24E (3 d/w, 10 J/w; 30J/w) died of an unidentified cause, and 119 rats were therefore left for data analysis.
Body weight
Initial mean body weight of all the experimental rats was 179.9 ± 10.8 g. Post hoc tests revealed no significant differences in initial body weight between all the experimental groups as all the rats were weight and age-matched. The final mean body weights of the rats in 8S and 8STP were 225.2 ± 21.3 and 227.3 ± 16.8 g respectively, and no significant difference was observed in final mean body weight between these two groups. Furthermore, no statistically significant differences in final mean body weight were observed between 32S group and all the other nine exercised groups (8STP24E). Final mean body weight of rats in 32S and 8STP24E groups was 269.1 ± 18.7 g, and the final body weights of the 32S and 8STP24E groups ranged from 257.8 ± 15.1 to 277.1 ± 17.3 g.
Bone mass
Tibial mass was significantly higher in 8STP than 8S (Table 2). In rats that were further trained for 24 weeks with different exercise regimens after 8STP, mean tibial mass were significantly higher (P<0.01) in all the rats given 24 weeks of jumping exercise that ranged from 30J/w to 200J/w generally.
Blood bone turnover markers
Mean concentrations of all the serum parameters of the various groups are presented in Table 2. No significant differences were evident in serum osteocalcin concentrations between the exercised rats and their respective age-matched sedentary controls. Similarly, no significant differences were evident in serum Alk-Ph concentrations between the exercised rats and their respective sedentary controls except for rats receiving exercise loads of 40, 50 and 200J/w. Nevertheless, non-significant higher values of serum Alk-Ph were evident in groups given 100J/w and 120 J/w when compared to 32S.
| Groups | Tibial mass(mg) | Osteocalcin (ng.mL−1) | Serum Concentration Alk-Ph (U.L−1) | 1CTP (µg.L−1) |
|---|---|---|---|---|
| 8S | 393.25 (42.49) | 124.45 (57.13) | 177.25 (31.29) | 1.01 (0.53) |
| 8STP | 424.47 (27.57) * | 151.14 (48.03) | 189.00 (36.46) | 1.20 (0.52) |
| 32S | 430.04 ( 30.00) | 66.01 (30.58) | 119.70 (29.12) | 1.08 (0.48) |
| 8STP24E (1d/w,10J/d)(10J/w) | 444.67 (28.25) | 62.68 (18.65) | 113.89 (18.71) | 0.96 (0.32) |
| 8STP24E(1d/w,20J/d)(20J/w) | 435.04 (33.94) | 76.79 (40.95) | 118.14 (15.20) | 0.60 (0.28) # |
| 8STP24E(3d/w,10J/d)(30J/w) | 493.53 (25.46) ‡ | 83.89 (43.60) | 110.00 (37.70) | 0.97 (0.08) |
| 8STP24E(1d/w,40J/d)(40J/w) | 455.66 (24.56) | 88.27 (56.31) | 147.40 (34.59) # | 0.33 (0.10) ‡ |
| 8STP24E(5d/w,10J/d)(50J/w) | 476.24 (28.90) † | 4.68 (29.34) | 146.50 (26.56) # | 0.31 (0.14) ‡ |
| 8STP24E(3d/w,20J/d)(60J/w) | 484.92 (20.71) † | 70.74 (38.01) | 110.00 (23.80) | 0.18 (0.14) ‡ |
| 8STP24E(5d/w,20J/d)(100J/w) | 482.62 (37.61) † | 61.16 (20.06) | 130.60 (33.64) | 0.18 (0.15) ‡ |
| 8STP24E(3d/w,40J/d)(120J/w) | 479.06 (31.93) † | 72.21 (26.92) | 125.50 (24.94) | 0.13 (0.07) ‡ |
| 8STP24E (5d/w,40J/d)(200J/w) | 497.51 (50.09) ‡ | 86.48 (14.57) | 152.40 (32.77) + | 0.17 (0.10) ‡ |
P<0.05 compared to age-matched sedentary controls (8S).
P<0.05
P<0.001 compared to age-matched sedentary controls (32S)
P<0.01
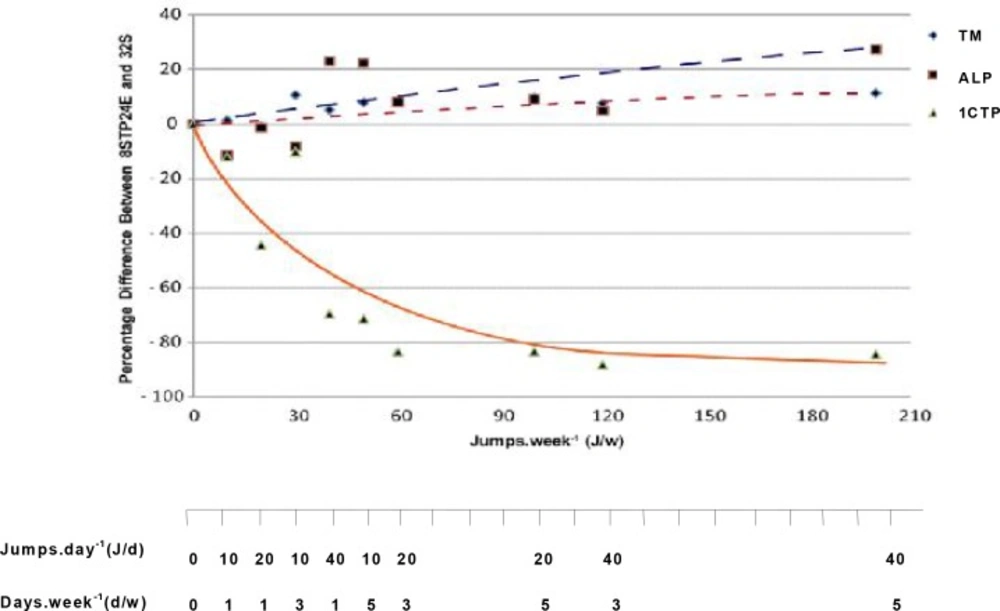
There was no significant difference in serum 1CTP between 8STP and their age-matched sedentary control (8S). Serum 1CTP concentrations, however, were significantly lower (P<0.001) in all rats given 24 weeks of jumping exercise that ranged from 40J/w to 200J/w when compared to that in the sedentary control (32S). Fig. 1 shows the relationship between tibial mass, serum Alk-Ph and 1CTP concentrations among the various exercised groups. Bone mass and serum formation marker increased, while serum bone resorption marker decreased during exercise with increasing exercise load.
DISCUSSION
Bone is always in a continuous dynamic remodelling process involving resorption of old bone and formation of new bone [1], and specific biochemical markers of bone have been commonly used in evaluating changes in bone turnover [1–4]. To date, most measurements of markers of bone turnover have been restricted to short-term interventions, such as acute or short-term effects of a single bout of exercise [15–18] or to cross-sectional studies [19–21]. Thus the longitudinal approach with 32 weeks of continuous jumping exercise used in the present study to examine the effect of long-term exercise in female rats as they age provides an additional picture of the effect of long-term exercise on bone turnover. In addition it also examines the minimum level of physical activity required to maintain bone gains through the estimation of bone formation and resorption markers.
The major findings of this study include (i) a gain in tibial mass that is significantly greater in rats given eight weeks of jumping exercise (8STP) compared to gains in age-matched sedentary controls (8S), (ii) significantly greater increase in mean tibial mass in rats that continued to receive exercise training for a further 24 weeks (Table 2), (iii) the increase was greater if the exercise was given 3 times per week instead of just once per week, and (iv) serum 1CTP levels were significantly lower in rats receiving exercise loads of 40 J/w and above (Table 2). Serum 1CTP levels also correlated negatively with increasing exercise loads at up to about 120 J/w after which it seems to plateau (Fig. 1). The present results indicate that the minimum level of exercise load required to elicit beneficial effects on bone mass after elevation of bone mass induced by 8STP was estimated at 30J/w, that is 10J/d for 3d/w, i.e. given in three divided doses in a week. This study confirms observations of previous studies in humans [22–25] and animals [26, 27], showing that bone mineral content and mass could be maintained with follow-up exercise loads that are lower than those initially used to induce the gains. In the present study, investigation of cellular mechanism responsible for the bone gains was unfortunately not carried out. However, it is speculated that bone cells might behave as a neuronal network [28] and incorporation of memories of mechanical loading events during growth and development might strongly influence bone biology. The acquired long-term memory of a mechanical loading environment might influence the responsiveness of bone tissue to external stimuli. That is, the history of weight bearing activities imparts long-term
cellular memory to the bone cell network. The above explanation may be applicable to the present observation that after 8 weeks of jumping exercise of 40 J/d at a frequency of 5 d/w, during the growing period, the reduced level of continuous exercise at 10 J/d at a frequency of 3 had the ability to maintain the exercise-induced gain in bone mass in female rats. It is speculated that bones of the rats might respond to low and reduced level of mechanical loading after acquiring long-term cellular memory on pre-existing mechanical loading. In the present study, unfortunately we are unable to provide any data on the primary measurement of bone mineral density. Additionally, bone responses at cellular level and genetic factors were also not determined. It is suggested that future studies with measurement of bone mineral density and investigation at cellular level and genetic factors could be carried out to clarify the precise underlying mechanism of the present findings.
Another finding in the 20 week-old young rats that performed 8STP was that no significant changes in bone turnover markers were observed even though there was a significant increase in bone mass when compared to the sedentary control (8S)(Table 1). One possible explanation for the absence of significant changes in blood osteocalcin, Alk-Ph and 1CTP concentrations after exercise in young rats in our study and in other exercise studies in young mice [29–32] might be due to the age of the rats or the localised effect of jumping exercise, which is limited to the hind limbs only, although the latter reason is somewhat contradicted by the finding of lower serum 1CTP concentrations following further continuous exercise (Table 2 and Fig. 1). It is possible the changes in blood parameters might have been too small to be detected, even though serum turnover markers are supposed to reflect the overall level of bone turnover of the entire skeleton [33–35]. In agreement with the present finding, absence of significant changes in blood bone turnover markers was also reported following an 18-month high-impact training in young male gymnasts [36], and another recent study following 6 weeks of aerobic dance exercise in young females [37]. The use of blood bone turnover markers in these types of exercise protocols in young animals and humans therefore remains uncertain and debatable.
Interestingly, when the rats were further trained for additional 24 weeks after 8 weeks of exercise, two positive relationships could be observed in bone mass and blood formation markers in rats with exercise loads of 40J/w (40J/d, 1d/w) and above. First, the tibial mass and serum Alk-Ph concentration increased with increasing exercise load (Table 2, Fig. 1). This positive correlation was evident when the differences in both tibial mass and serum Alk-Ph between exercised rats and their age-matched sedentary controls were plotted on a logarithmic curve fit against workload (J/w). The increasing levels of bone mass and serum alkaline Alk-Ph with exercise load may indicate the presence of net bone formation in these rats. Secondly, a negative relationship was observed in serum 1CTP, which reflects decreased bone resorption with increasing workload. Here, serum 1CTP concentration decreased with increasing exercise loads (Table 2, Fig. 1). The present findings show that when bones are mechanically loaded during exercise, bone resorption decreases and bone formation may increase. This finding is also supported by studies using other markers of bone turnover. For example, increase in serum Alk-Ph was seen following 12 weeks of treadmill running exercise in aging ovariectomised rats [38], whereas decreases in urinary deoxypyridinoline concentrations following 90 days of treadmill running in rats have also been reported [39]. Similarly, in humans, lower serum 1CTP concentrations were found in runners who had been active in their sports for about 12 years when compared to their more sedentary controls [20].
In our study, there were no significant changes in serum turnover markers with 8STP in young rats, however when the rats were further trained for additional 24 weeks after 8 weeks of exercise, significant changes in serum turnover markers were observed in some of the exercised groups with age. These results may imply that, maybe, a longer jumping duration is needed for eliciting discernable changes in bone turnover markers in the rats.
CONCLUSION
It appears that for maintaining the acquired gains in bone mass following 8STP, an exercise load of 30J/w, i.e. 10J/d for 3d/w, was the minimum level of continuous exercise load that was required after 8STP to maintain the 8STP-induced bone gains. It also appears that significant increases in bone mass in young rats following 8STP might not always be reflected by changes in serum levels of osteocalcin, Alk-Ph and 1CTP. However, measurement of these parameters, particularly 1CTP, might be more useful when evaluating the maintenance of bone gains after bone loading. While these findings suggest that bone mass and serum markers of bone turnover responded differently to jumping exercise in young rats, more measurements of bone responses at cellular level are needed to confirm our observations.