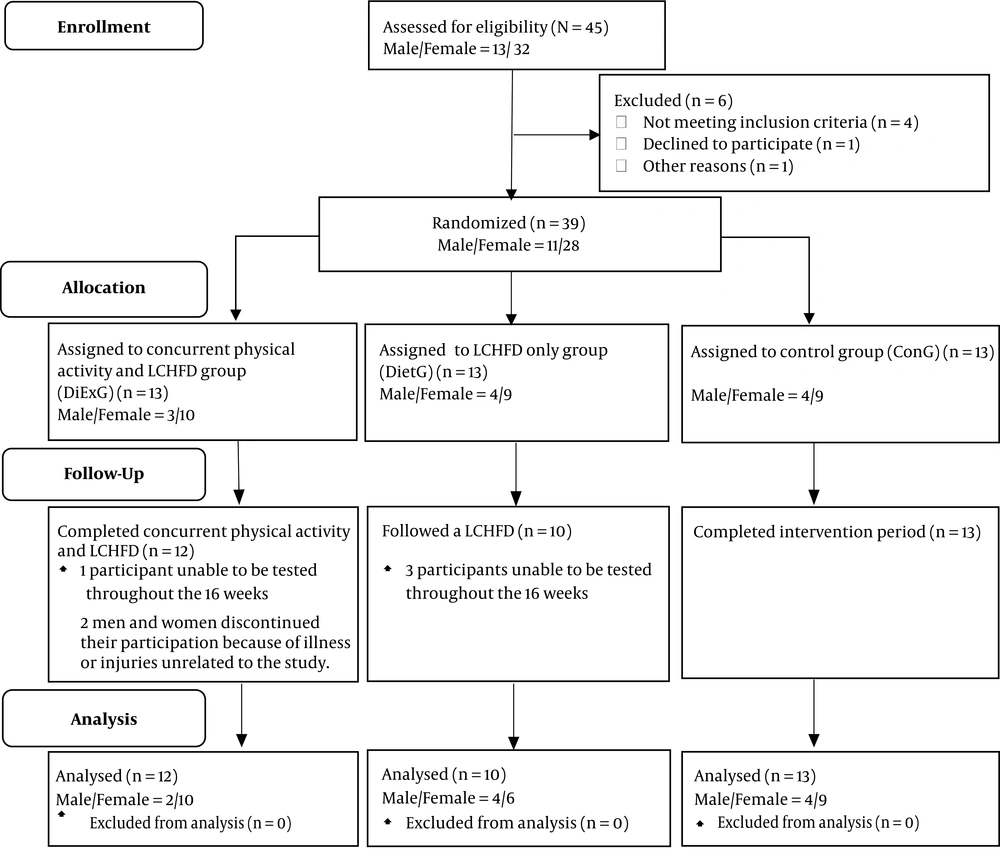
The primary intention of this study was to determine if a LCHFD provides any benefits on lipoprotein-lipids, either alone or in conjunction with physical activity in type 2 diabetics. The major result of present study is that a 16-week LCHFD with or without physical activity does not have any benefit on lipoprotein-lipids in type 2 diabetics. Specifically, no significant changes were observed in TC, TG, LDLC and HDLC in either the DiExG or DietG.
Following the 16-week experimental period, TC was found to be relatively stable in all three groups. This study’s findings are in agreement with Thompson et al. (
13) who previously demonstrated that a high-fat diet failed to elicit any improvements in TC. This finding and that of Thomson et al. (
13) are in contradiction to the findings of Crouse et al. (14) who purport the substitution of SFA with polyunsaturated fatty acids might have a lowering effect on TC. In terms of low-carbohydrate diets, Volek et al. (
30) demonstrated that very low carbohydrate diets (VLCD) (< 50 g carbohydrates), as utilized in the present study, actually resulted in a harmful increase in TC. In terms of exercise, previous studies have demonstrated that exercise has little/no effect on TC levels (
21). This may not be due to the inability of exercise to alter any specific biological mechanism, but rather that TC is a generalized measure of lipoprotein-lipids and includes the “good” (i.e. HDLC) and “bad” (i.e. LDLC) components of cholesterol and any increase and/or decrease in its constituents creates an unchanged TC.
While it has been claimed that TG is the most consistent and predictable of the lipid changes when a LCHFD is used (
31), in this study, TG increased, albeit non-significantly, by 17.9% in the diet-only group and supports the general assumption that TG is only reduced using a low-fat diet (
32). More disconcerting is the finding that physical activity failed to improve TG in this study. This may indicate that the addition of the high-fat diet actually inhibited the TG-lowering benefits of exercise. This is because exercise, irrespective of modality, is associated with decreased serum levels of TG (
33,
34). Further, evidence for the interference effect of a high-fat diet on physical activity is supported by Lian et al. (
35) who have found that even walking, such as utilized in this study, can lower TG.
This study’s findings are in agreement with Thompson et al. (
13) who previously demonstrated that a high-fat diet failed to elicit any improvements in LDLC. In terms of low-carbohydrate diets, Volek et al. (
30) demonstrated that very low carbohydrate diets (VLCD) (< 50 g carbohydrates), as utilized in this study, actually resulted in a harmful increase in LDLC. Again, this study uniquely demonstrates that the use of a LCHFD may deleteriously counterpoise the positive effects of physical activity in that exercise, even walking, may decrease LDLC levels (
33,
34).
High-density lipoprotein is considered cardioprotective and every 0.03 mmol.l
-1 increase in total HDLC translates into as much as a 2% to 3% reduction in CVD risk (
36). While previous research has indicated that diets that substitute fats with carbohydrates are associated with a lowered HDLC (7), the present study indicated no change in HDLC following the 16-week experimental period. In addition, Volek et al. (
37) found that the consumption of a low-carbohydrate diet increases HDLC, especially when compared to a low-fat diet. Once again, even the concurrent group that also engaged in exercise failed to elicit any improvements in this parameter. This is problematic in that exercise, even walking as utilized in this study, is associated with an improved HDLC (
33-
35). One possible reason for a lack of improvement in the DiExG may be due this study’s 16 weeks being of insufficient intensity or duration not meeting the time of latency for this mode of exercise to improve HDLC (
21).
The lack of change in the measured lipoprotein-lipid profiles in this study are difficult to attribute to any specific mechanism. In this regard, the specific biological mechanism(s) responsible for adaptations in lipoprotein-lipids following dietary and/or exercise interventions are as yet unknown (
24). It is for this reason and due to the limited amount of scientific research on the influence of LCHFDs on lipoprotein-lipid profiles, especially in individuals with diabetes, that the health benefits of LCHFDs remain controversial.
5.1. Limitations
The present study had some limitations. Due to the small sample, results should not be universally directed to the entire diabetic populace. In addition, the study did not utilize a single gender and differences in gender hormones and their effect on lipoprotein-lipids could have affected the results. While there are challenges with using dietary self-report measures and pedometry as a tool for physical activity measurement, both will continue to be popular approaches due to the lack of inexpensive and more sensitive objective means of assessment. Further, it is unclear whether a longer intervention period would result in positive improvements since the effect of exercise may require a certain time of latency (such as required on HDLC) before the changes can be proved.
5.2. Conclusions
In conclusion, we found that 16-weeks of a low carbohydrate, high fat diet, alone or in conjunction with physical activity, did not improve lipoprotein-lipids in type 2 diabetics and may actually result in unfavorable, albeit insignificant, lipoprotein-lipid adaptations. In fact, it appears that health professions should not unquestionably include LCHFDs in a diabetic treatment regime to manage or improve lipoprotein-lipid in type 2 diabetics. Rather health professionals should apply proven well-established dietary guidelines on an individual needs basis.