Mature teratoma cyst or dermoid cyst is the most common teratoma, and ovarian neoplasm (10 - 25%) constitute 60% of all benign ovarian neoplasms (
22). They are mostly unilateral (
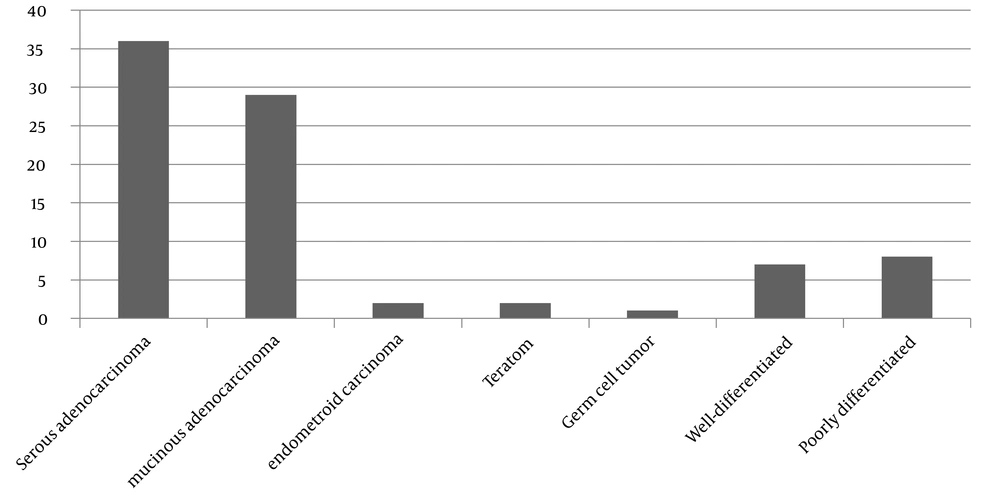
23). In our study, 80.6% of dermoid cysts were unilateral. This cyst grows slowly and is bilateral in 10% of cases (
24). Malignant changes in this type of ovarian cyst are in about 1 - 2% of patients, and torsion is their most complication (
25). Some radiologic features, like ultrasonography, can suggest MCT, but tumor markers alongside the imaging accommodate the diagnosis (
8). The majority of signs and symptoms of ovarian cancer are nonspecific. Abdominal pain was the most frequent symptom (59.6%), followed by menstrual disorders in dermoid cyst cases in our study.
Lack of specific and sensitive biomarkers can lead to late diagnosis and even patient death (
26). It showed that the mean level of CA19-9 was higher in the dermoid group than in the cancer group, and the mean level of CA125 was much lower in the dermoid group than in the cancer group. It is similar to Wang et al.’s study at Wenzhou Medical University. They found that CA19-9 was likely the most reliable tumor marker for diagnosing MCT (
27). It also resembles Mughir and Al-Hilli’s study in 2019 in Iraq on 50 cases of ovarian dermoid cysts and 50 control patients. Their results show that the CA19-9 level in the case group was about four times higher than in the control group. In our study, there was a direct relationship between the size of the cyst and the level of CA19-9; likewise, in their study, there was a direct relationship between the size of the tumor and the level of CA19-9. In their study, most dermoid cysts are unilateral, and about 10 - 15% of them were bilateral, as well as our research (
28).
In Prodromidou et al.’s study and colleagues, the meta-analysis showed that in patients with elevated CA19-9, there was an increased size in mature cystic teratoma. At the same time, there was no relationship between the patient’s age, bilaterality, site of the lesion, and the simultaneous elevation of CA-125 in patients with elevated CA19-9 (
29). It is consistent with our findings that there was no correlation between CA19-9 and age or being unilateral/bilateral, but there is a significant correlation with size. In Lertkhachonsuk et al.’s study, CA125 was the best diagnostic tumor marker for borderline and malignant mucinous ovarian tumors, followed by CA19-9 and CEA (
30). This is similar to our study that in the cancer group, 88% of patients have elevated CA125. Ali et al.’s study demonstrated that the combination of microRNA-204, CA125, and CA19.9 was the best test for the early detection of ovarian tumors and cancers, but in our study, elevated CA19-9 was more related to the dermoid cyst group and elevated CA125 more related to cancer group (
26). Also, in Bagde et al.’s study LDH, BHCG, and CA19-9 did not significantly differ in malignant and benign cases (
31).
In Sagi-Dain et al.’s study, they demonstrated that combining CA19-9 with CA125 compared with CA125 alone did not significantly affect sensitivity and specificity in differentiating malignant from benign adnexal masses. The mean CA19-9 levels were higher in metastatic carcinoma and could help differentiate metastatic tumors from primary ovarian malignancy (
32). The strength point of this study is that all imaging assessments and tumor marker measurements are done in an academic referral center. Increased sample size and assessment of other tumor markers can be suggested for future studies to cover the weak points of our study. Most of the dermoid cysts in the present study were unilateral. According to the relationship of CA19-9 in the dermoid cyst group with cyst size, it can be said that the larger the cyst size, the higher the tumor marker level will be. The increase in the level of CA125 suggests the possibility of malignancy in the ovarian cyst alongside an increase in age and cyst size. Finally, CA19-9 can be used in diagnosis along with other diagnostic tools, especially ultrasound. This tumor marker has no diagnostic value in small cysts, but if the cyst size increases, it will help in a definitive diagnosis.