1. Background
Escherichia coli is a widely distributed Gram-negative intestinal bacillus found throughout the world among warm-blooded animals and is known as the normal intestinal flora.
This non-invasive bacterium found in the intestines of animals and humans prevents the growth and spread of other pathogenic bacteria in the intestines. It is also responsible for a very important disease in poultry called colibacillosis”, which is characterized by symptoms, such as inflammation of the air sacs, cellulite, pericarditis, peripheral hepatitis, and respiratory distress (1).
Escherichia coli is a common germ in the digestive tract (the small intestine) of poultry and other mammals, and it may be pathogenic in both hosts (2). Escherichia coli is an opportunistic pathogenic bacterium that causes secondary disease (3, 4). Infections caused by E. coli are responsible for economic losses to the poultry industry (5, 6).
At the beginning of the twentieth century, advances in chemistry and the discovery of sophisticated systems of organic synthesis led to the development of the pharmaceutical industry and the replacement of synthetic drugs with herbal medicines, but as advances in the production of new chemical drugs and antibiotics, the harmful effects of these drugs gradually appeared, and since the 1950s, numerous pathogenic bacteria have shown resistance to antibiotics, still expanding. Therefore, the use of herbal medicines to obtain new compounds to overcome bacterial resistance was examined.
Peppermint is a plant that has long been considered due to its healing and aromatic properties (7).
Peppermint is the most important and famous type of mint (8). It is used as a stomach tonic, analgesic, anticonvulsant, and nerve relaxant (9, 10). This plant is also used in the treatment of gastrointestinal infections (11). Medicinal properties of mint include antispasmodic, anticonvulsant, antiemetic, antiflatulence, antibacterial, and antifungal properties (9).
Peppermint also has antimicrobial and anticancer properties. Mint oil is used in preparing home medicines to relieve migraines, headaches, abdominal pains, as well as reducing anger and insomnia (12). In general, the antibacterial effect of extracts depends on the compounds in them, including phenolic compounds. These compounds disrupt the vital activity of bacteria by permeating cell membranes with chelated membrane cations (13).
Rosemary is an herbaceous plant with green, fragrant, and sharp leaves. Rosemary essential oil is an antimicrobial substance and its chemical composition, antioxidant properties, and antimicrobial effects have been proven in various studies.
Withania somnifera belongs to the Solanaceae family and is widely used in Ayurveda medicine (a traditional medical system in India and Nepal) (14).
This plant has several benefits in medical science, including anti-inflammatory, anticancer, antistress, and antioxidant properties (15).
Withania somnifera is a medicinal plant that grows botanically as a perennial shrubby plant, with 30 to 100 cm tall shrubs in Pakistan, southwest India, and Afghanistan.
The therapeutic efficacy and pharmacological properties of this plant include sedative, tonic, and regulating effects on uterine hormones (14).
Conventional extraction methods are based on the plant being placed in a suitable solvent, which is used to speed up the stirring or heating process. Common methods include soxhlet, distillation, soaking, and percolation. It is worth mentioning that ultrasound is one of the new extraction methods in which waves with a frequency higher than 200 kHz penetrate into the material, causing successive expansion and contraction in the molecules in the environment, and consequently, result in cavities inside the plant material. These cavities are asymmetrically interconnected, causing the material to quickly escape from inside the cell. These waves can also destroy the walls of biological cells and facilitate the release of material (16). Extraction by sound waves is cheap, simple, and efficient compared to conventional methods. Increased performance and fast extraction process are among the main advantages of using sound waves in solid-liquid extraction. Sound waves can lower the process temperature and allow the extraction of unstable compounds against heat (17).
2. Objectives
The present study aimed to investigate the antimicrobial activity of plant extracts prepared by ultrasound against E. coli isolated from poultry stool.
3. Methods
3.1. Plant Materials
Mentha piperita, Rosmarinus officinalis, and Withania somnifera samples were collected from Sistan and Baluchistan province, southeast of Iran. The plant samples were dried at 25°C, powdered and transferred into glass containers, and then preserved until the next experiment.
3.2. Preparation of Extracts by Ultrasound
The samples were cut and 10 g of the plant dried powder was placed into half-liter flasks containing 100 mL of methanol. The contents of flasks were shaken 24 hours at room temperature using a shaker device with a speed of 130 rpm, were kept in the ultrasonic carrier for 10 minutes, and were filtered through Whatman paper. Separation of solution from the extract was done using a rotary device and vacuum pump. The obtained extract was weighed. Then, the samples were placed in the fridge at 4 degrees of temperature to be used in antimicrobial tests.
3.3. Preparation of Methanol Extracts
The samples of each plant were separately dried, powdered, and dissolved in 200 mL of methanol using a shaker water bath for 24 h at room temperature. After filtration with Whatman no.: 1 filter paper, the extract was concentrated using a rotary evaporator (Heidolph-Germany) at 40°C for 40 min. The semisolid extract produced was kept at -80°C overnight and then subjected to freeze-drying for 24 h, at -70°C in 200 mL of vacuum. For further use, the extract was stored in an airtight container at 4°C in the refrigerator.
3.4. Isolation of Escherichia coli
This cross-sectional study was performed using the stool. All 10 strains of E. coli were isolated from the stool samples.
The samples were examined microscopically by a Gram staining technique. Samples with Gram-negative results were inoculated on plates of nutrient agar, cystine-lactose-electrolyte-deficient (CLED) agar, MacConkey agar, and blood agar, and then were incubated at 37°C for 24 h. Those colonies that showed fermentation of lactose on MacConkey agar and CLED agar were purified and identified according to their morphology as circular, rose-pink to red colonies on MacConkey agar and yellow colonies on CLED agar. The isolates were identified by biochemical tests such as catalase, potassium hydroxide, indole, methyl red, Voges-Proskauer, urease, citrate, H2S, and oxidase.
3.5. Antimicrobial Test of the Extract
To prepare the microbial suspension, several colonies of the fresh and 24-hour culture of bacteria were transferred to Müller-Hinton Broth culture medium, and turbidity equivalent to half McFarland (turbidity equivalent to 1.5 × 108 bacteria per mL) was prepared.
3.6. Antibiotic Activity
The susceptibility of all antibiotics was carried out using the disc diffusion method on Muller-Hinton agar, as recommended by the Clinical and Laboratory Standards Institute (CLSI) (18). The procedure is briefly described here: E. coli isolated plates were grown overnight on blood agar and nutrient agar, and the colony suspension was prepared using sterile saline water, equivalent to 0.5 McFarland standard. The suspension (10 µL) was spread over the media plate and the antibiotic disc was transferred aseptically to the surface of inoculated media. Isolated plates were tested with different antibiotics and their concentration was shown in parenthesis: Gentamicin, amoxiclav, azithromycin, and amikacin (developed by Padtan Teb Co., Iran). After 24 hours of incubation at 37°C, each zone of growth inhibition was measured and the sensitivity and resistance of the strains were determined; then, their results were compared with the standard CLSI table.
3.7. Determination of Minimum Inhibitory Concentration and Minimum Bactericidal Concentration of Medicinal Plant Extracts
The broth microdilution method was used to determine determination of minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC). All tests were performed in Mueller-Hinton broth supplemented with Tween 80 at a final concentration of 0.5% (v/v). Briefly, serial doubling dilutions of the extract were prepared in a 96-well microtiter plate ranged from 0.3 mg/mL to 20.00 mg/mL. To each well, 10 µL of indicator solution (prepared by dissolving 10 mg of the extract in 2 mL of dimethyl sulfoxide (DMSO)) and 10 µL of Mueller-Hinton broth were added. Finally, 10 µL of bacterial suspension (106 CFU/mL) was added to each well to achieve the concentration of 104 CFU/mL. The plates were wrapped loosely in cling film to ensure that the bacteria would not get dehydrated. The plates were prepared in triplicate, and then they were placed in an incubator at 37°C for 18 - 24 h. The color change was then assessed visually. The lowest concentration at which the color change occurred was taken as the MIC value. The average of 3 values was calculated providing the MIC and MBC values for the tested extracts. The MIC was defined as the lowest concentration of the extract at which the microorganism did not demonstrate visible growth. The microorganism growth was indicated by turbidity. The MBC was defined as the lowest concentration of the extract at which the incubated microorganism was completely killed.
4. Results
The results of this study showed that resistance to antibiotics such as ceftazidime, gentamicin, azithromycin, and amoxiclav was 80%, 20%, 20%, and 10%, respectively, while sensitivity to antibiotics such as gentamicin, amoxiclav, azithromycin, and amikacin was 70%, 60%, 60%, and 30%, respectively.
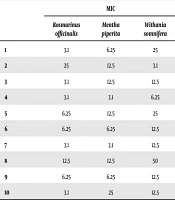
The results of this study showed that the lowest inhibitory concentration of rosemary extracts prepared by ultrasound was equal to 3.1 mg/mL; 5 strains were inhibited at this concentration, while the highest inhibitory concentration was equal to 25 mg/mL (Table 1).
| MIC | |||
|---|---|---|---|
| Rosmarinus officinalis | Mentha piperita | Withania somnifera | |
| 1 | 3.1 | 6.25 | 25 |
| 2 | 25 | 12.5 | 3.1 |
| 3 | 3.1 | 12.5 | 12.5 |
| 4 | 3.1 | 3.1 | 6.25 |
| 5 | 6.25 | 12.5 | 25 |
| 6 | 6.25 | 6.25 | 12.5 |
| 7 | 3.1 | 3.1 | 12.5 |
| 8 | 12.5 | 12.5 | 50 |
| 9 | 6.25 | 6.25 | 12.5 |
| 10 | 3.1 | 25 | 12.5 |
The Minimum Inhibitory Concentration of Ultrasonic Plant Extracts Against Escherichia coli Bacteria mg/mL)
The lowest inhibitory concentration of peppermint extracts prepared by ultrasound was equal to 3.1 mg/mL that inhibited growth of 2 strains. Also, the highest inhibitory concentration was 25 mg/mL, with one strain being inhibited at this concentration, while 4 strains were inhibited at a concentration of 12.5 mg/mL (Table 1).
The lowest inhibitory concentration of W. sominefra extracts prepared by ultrasound was equal to 3.1 mg/mL, unilaterally inhibited in this concentration, and the highest inhibitory concentration was equal to 50 mg/mL. Also, 5 strains were inhibited at a concentration of 12.5 mg/mL (Table 1).
The highest bactericidal concentration of rosemary extracts prepared by ultrasound was equal to 50 mg/mL; one strain was eliminated at this concentration, while the lowest bactericidal concentration of ultrasonic rosemary waves was equal to 6.25 mg/mL, and 5 strains were lost at this concentration (Table 2).
| MBC | |||
|---|---|---|---|
| Rosmarinus officinalis | Mentha piperita | Withania somnifera | |
| 1 | 6.25 | 12.5 | 50 |
| 2 | 50 | 25 | 6.25 |
| 3 | 6.25 | 25 | 25 |
| 4 | 6.25 | 6.25 | 12.5 |
| 5 | 12.5 | 25 | 50 |
| 6 | 12.5 | 12.5 | 25 |
| 7 | 6.25 | 6.25 | 25 |
| 8 | 25 | 25 | 100 |
| 9 | 12.5 | 12.5 | 25 |
| 10 | 6.25 | 50 | 25 |
The Minimum Bactericidal Concentration of Ultrasonic Plant Extracts Against Escherichia coli Bacteria (mg/mL)
The highest bactericidal concentration of peppermint extracts prepared by ultrasound was equal to 50 mg/mL, and one strain was lost at this concentration, while the lowest lethal concentration of peppermint ultrasonic waves was equal to 6.25 mg/mL, in which 2 strains were lost at this concentration (Table 2).
The highest bactericidal concentration of W. sominefra extracts prepared by ultrasound was equal to 100 mg/mL; one strain was lost at this concentration, while the lowest lethal concentration of wind cheese ultrasonic waves was equal to 6.25 mg/mL. One strain was lost at this concentration (Table 2).
5. Discussion
Escherichia coli is one of the bacteria involved in various diseases in poultry as well as in humans.
The results of this study showed that resistance to antibiotics such as ceftazidime, gentamicin, azithromycin, and amoxiclav was 80%, 20%, 20%, and 10%, respectively, while sensitivity to antibiotics such as gentamicin, amoxiclav, azithromycin, and amikacin was 70%, 60%, 60%, and 30%, respectively.
In a study performed by Kechaei and Rawa (19), the antibiotic resistance of E. coli strains isolated from urinary tract infections in outpatients referred to Nabi Akram Hospital in Zahedan during the past years was evaluated. The results of this study showed that out of 87 samples, 2.70% belonged to women and 8.29% belonged to men. Patients were in the age range of 12 - 45 years and their resistance to antibiotics was as follows: Cotrimoxazole (6.66%), nalidixic acid (63%), ceftazidime (8.44%), nitrofurantoin (1.26%), amikacin (5.19%), gentamicin (7.13%), and imipenem (5.4%).In the study by Babaei Kasmaei et al. (20), the antibiotic sensitivity in E. coli with multiple drug resistance was investigated in samples isolated from outpatients with urinary tract infection in Tehran. The results showed that out of 123 patients, 59 cases (45.52%) had multiple drug resistance and the highest drug sensitivity was observed in imipenem, nitrofurantoin, and amikacin (20).
From ancient times, the natural substances in plants have played an important role in the discovery of therapeutic agents. Plant extracts with antimicrobial properties are very important in the treatment of some diseases. So far, many studies have been performed in this regard.
The results of the present study showed that the lowest inhibitory concentration of M. piperita extracts prepared by ultrasound was equal to 3.1 mg/mL, of which 2 strains were inhibited in this concentration, and their highest inhibitory concentration was equal to 25 mg/mL, of which one strain was inhibited in this concentration and 4 strains were inhibited in a concentration of 12.5 mg/mL.
In a study by Heydari and Bagheri (21), the antimicrobial effects of nanoemulsions of aqueous extracts of M. piperita on Gram-negative bacteria E. coli were determined. The results showed nanoemulsions in concentrations of 1% to 5% of M. piperita essential oil with an average particle size of 12 nm and antimicrobial properties with an average of 75% and 3.12 mm zone of no growth against Gram-negative bacteria of E. coli (21).
In their study, Yazdani et al. (22) examined the antimicrobial and antioxidant activity of M. piperita essential oil. The results showed that menthol, neomenthyl acetate, and menthofuran were the main constituents of the samples of peppermint essential oil collected from Marivan, Iran, respectively. Menthol was the main constituent of essential oil. The essential oil of this plant was effective against Gram-positive bacteria Staphylococcus epidermidis, Bacillus subtilis, and Staphylococcus aureus and Gram-negative bacteria Shigella dysentery and Klebsiella pneumoniae (22).
In a study by Zandi et al. (23), the antibacterial activity of M. piperita extracts was investigated to control pathogenic bacteria. The results showed that the lowest inhibitory concentration of M. piperita extracts was observed on the tested microorganisms equal to 3.25 mg/mL and in the case of Staphylococcus aureus and Enterococcus faecalis. Also, the largest diameter of the growth inhibition zone was related to Staphylococcus aureus (32 mm) (23).
In their study, Muntean et al. (24) examined the antimicrobial activity of M. piperita essential oil against several antibiotic-resistant species. The highest inhibitory concentration was 40 mg/mL, which was inhibited by Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter baumannii (24).
According to Satmi and Hossain’s study (25), the hexane extracts of Mentha piperitahad a moderate potential for antibacterial activity against E. coli, S. aureus, and Xanthomonas campestris at all concentrations of 2.5 mg/mL, 1.25 mg/mL, 0.675 mg, and 0.355 mg/mL in the range of 7 - 14 mm. The crude extracts of ethyl acetate and butanol showed partial activity against the three mentioned bacterial strains (E. coli, Xanthomonas campestris, and S. aureus) at all active concentrations. The hexane extracts of M. piperita had a moderate potential for antibacterial activity against E. coli, S. aureus, and Xanthomonas campestris at all concentrations of 2.5 mg/mL, 1.25 mg/mL, and 0.675 mg/mL, and showed 0.335 mg/mL in the range of 7 - 14 mm (25).
The results of this study showed that the lowest inhibitory concentration of rosemary extracts prepared by ultrasound was equal to 3.1 mg/mL and the highest inhibitory concentration was 25 mg/mL.
In the study performed by Gomez-Estaca et al. (26), rosemary oil was reported to inhibit the growth of common food bacteria involved in food spoilage. Burt (27) also showed antibacterial activity of rosemary essential oil against E. coli, Bacillus cereus, and S. aureus. In addition, Sirocchi et al. (28) showed that rosemary essential oil inhibited the growth of Brochothrix thermosphacta and Enterobacteriaceae. Govaris et al. (29) also reported the inhibitory effect of dietary supplementation of turkeys with rosemary (5 and 10 g/kg) on the growth of bacteria responsible for spoilage (psychrotrophs, mesophilics, enterobacteria, and lactic acid bacteria).
In a study by Jarrar et al. (30), that examined the antimicrobial activity of rosemary on methicillin-resistant Staphylococcus aureus, the results showed that the MIC of rosemary ethanol extracts was in the range of 0.39 - 3.13 mg/mL. The MBCs were usually equal to or twice that of MICs. The antimicrobial activity of rosemary and cefuroxime ethanolic extract compounds showed their synergistic effects against methicillin-resistant Staphylococcus aureus (MRSA) (30).
The results of this study showed that the lowest inhibitory concentration of W. somnifera extracts prepared by ultrasound was equal to 3.1 mg/mL, which was unilaterally inhibited in this concentration, and the highest inhibitory concentration was equal to 50 mg/mL and 5 strains were inhibited in a concentration of 12.5 mg/mL.
Based on a study performed by Bokaeian and Saeidi. (31) to examine the antimicrobial activity of W. somnifera leaf extracts against S. aureus, the highest inhibitory concentration was 250 ppm in 12 strains and the lowest inhibitory concentration was 63 ppm, in which two strains were inhibited at this concentration.
The study of Bisht and Rawat (32) investigated the antimicrobial potential of W. somnifera leaf extracts against Gram-positive stems. The results showed that the methanolic leaf extracts of W. somnifera were very effective in inhibiting experimental pathogens, including Staphylococcus aureus and methicillin-resistant Enterococcus, with an average inhibition zone of 20.6 mm and 19.4 mm in a 2 mg/l mL (100 µL) concentration (32).
Similar to the previous study (33), methanolic root extracts of W. somnifera showed a 4 mm inhibitory zone against Escherichia coli and a 10 mm inhibitory zone at 10 µg/mL against enterococci, much lower than that observed in our study. This discrepancy may be due to the use of another part of the plant extracts. Owais et al. (34) showed a 22 mm region of inhibition against S. aureus in a concentration of 20 mg/mL W. somnifera leaf extracts. In another study (35), a 15 mm zone of inhibition in a concentration of 100 µg/mL against S. aureus and E. coli by W. somnifera leaf extracts was reported.
5.1. Conclusions
In this study, a medium power ultrasonic device was used to obtain plant extracts. According to the results of this study, no significant difference was observed between the extracts of different plants. Currently, there are devices with higher temperature power, e.g., 350 watts. By depleting the device power, it can lead to better extraction, i.e. the release of substances with greater concentration and variety from the plant. In this case, it seems that the differences in the extracts obtained are greater. This study shows that the ultrasonic extraction method is a fast, economical, and effective method for extracting antibacterial substances in plants. Based on the results obtained from the antimicrobial activity of each extract, the extract obtained from the extraction of peppermint methanolic solvent showed the greatest antimicrobial effect.