1. Background
Diabetes mellitus is a metabolic disease characterized by hyperglycemia due to lack of access to insulin or reduced sensitivity to it, or a combination of both (1). Type 1 diabetes is the result of the destruction of pancreatic beta cells (pancreas), which usually leads to an absolute deficiency of insulin. The classic symptoms of type 1 diabetes are weight loss, polyuria, and polydipsia, which usually quickly become apparent (2).
Recent research findings indicate that adipose-derived mediators are effective factors in insulin sensitivity and beta-cell dysfunction; therefore, adipose tissue is recognized as one of the tissues associated with diabetes (3). Adipose tissue is divided into two categories, including white adipose tissue and brown adipose tissue. With the activation of brown adipose tissue, cyclic adenosine monophosphate (cAMP) levels increase rapidly, leading to lipolysis and upregulation of uncoupling protein-1 (UCP1) and increased mitochondrial density, which play a thermogenic role (4). There are several mechanisms that affect the increase of metabolism in brown adipose tissue of which we can refer to exercise and herbal medicines.
Many researchers around the world are working to prevent, treat, or reduce the complications of diabetes using a variety of methods. Today, the use of medicinal plants and extracts to treat diseases has increased (5). One of the suggested methods for reducing the complications of diabetes, including its cardiovascular complications, is physical activity combined with diet and medication
Cinnamon, with the scientific name of Cinnamomum zeylanicum, is one of the plants that seem to have blood-glucose-lowering effects. A study showed that cinnamon extract increases glucose consumption and glycogen production and increases IR phosphorylation, all of which increase sensitivity to insulin (7). An effective ingredient in cinnamon that improves insulin levels, glucose, and insulin resistance is a polymer called methyl hydroxy chalcone, which works like insulin (8). On the other hand, regular exercise has beneficial effects on overall health, and the role of exercise in the treatment and prevention of metabolic diseases is well defined. Exercise improves glucose homeostasis of the whole body and increases insulin sensitivity, which is considered to be related to the adaptations of skeletal muscle, adipose tissue, etc. (9). Blood glucose uptake and blood sugar regulation are complex processes, and defects in these processes lead to consequences such as increased insulin resistance, diabetes, defects in the process of metabolism and energy production, and decreased athletic performance (10).
Among these, glucose transporters play an essential role in glucose uptake into the cell. GLUTs are one of the glucose transporters, and so far, five isoforms of them have been identified. They are insulin-dependent transporters that are mainly expressed in skeletal adipose tissue and muscle. Insulin and exercise stimulate rapid and intense movement to cell membranes to induce glucose uptake into muscle cells and brown adipose tissue (11). Lehnen et al. (2010), in their research on rats, showed that exercise could increase GLUT4 in adipose and muscle tissue (12). However, some studies have shown that this transporter does not change as a result of exercise. For example, in Gurley et al.’s (2016) study, although four weeks of running in obese diet-fed rats improved plasma fasting insulin, it had no effect on muscle GLUT4 mRNA (13).
The other human IR gene is located on chromosome 19 and is more than 150 kb. The abundance of mRNA and receptor protein is upregulated by differentiation of adipocyte and muscle precursor cells as an insulin sensitivity phenotype. In some cells, insulin exposure reduces the receptor mRNA abundance, which may play a role in regulating the number of receptors in the body (14). Exercise can increase the body's response to insulin by increasing the intracellular glucose transporter and IR substrate and increases insulin sensitivity (15). So far, few studies have been performed on the simultaneous effect of aerobic training and cinnamon consumption on the gene expression of GLUT4 and IR.
2. Objectives
The present research aimed to investigate the effect of swimming training and cinnamon consumption on the gene expression of GLUT4 and IR in the brown adipose tissue of diabetic rats.
3. Methods
3.1. Subjects
In this experimental and fundamental study, initially, 28 rats with an age range of 8 to 10 weeks were purchased from the Laboratory Animal Breeding and Reproduction Center of Islamic Azad University, Marvdasht Branch, and were maintained for one week in the Animal Sports Physiology Laboratory of this branch to pass an environmental adaptation course. It should be noted that during the research, these animals were kept in the standard conditions of 12-hour light-dark cycle, humidity 55%, temperature 22 to 24°C in autoclave cages made of transparent polycarbonate, and special food for rats were ad lib.
One week later, 28 rats underwent peritoneal injection of streptozotocin (manufactured by Sigma Co., USA) at a dose of 55 mg/kg body weight. After 4 days, the blood glucose of rats was measured by tail punching method and based on fasting blood glucose and to homogenize, the diabetic rats were assigned in 1- control (C), 2- cinnamon (Ci), 3- swimming (S) and 4- swimming plus cinnamon (S + Ci) groups. Then, rats in the training groups were trained for six weeks, five sessions per week, five days a week, for two minutes in the first week, and in each training session, 30 seconds were added to the training until it reached 4 minutes (16). Also, rats in the cinnamon groups received the aqueous cinnamon extract orally at a dose of 200 mg per kg of animal body weight (17). At the end of the sixth week and 48 hours after the last training session, rats in the study groups were anesthetized with ketamine and xylazine in a ratio of 3:1 by intraperitoneal injection following 16 hours of fasting. After diagnosis of complete anesthesia with pain reflex tests by squeezing the tail and ensuring complete analgesia, brown adipose tissue was extracted by laboratory specialists and immediately kept at -80°C.
3.2. Measurement of the Research Variables
To do molecular studies at the level of gene expression, first RNA extraction of brown adipose tissue was performed according to the manufacturer’s protocol (Sinagen, Iran), then using light absorption at 260 nm wavelength, the concentration and purity of RNA samples were quantitatively obtained using the following formula:
After extracting RNA with very high purity and concentration from all the studied samples, the instructions for cDNA synthesis in the Fermentas kit (K1621) were followed, and then the synthesized cDNA was used for reverse transcription reaction. First, the designed primers related to genes were examined, and then the expression of genes was evaluated by the quantitative q-RT-PCR method, and their relative expression was calculated using the 2-ΔΔCt formula (Table 1).
| Genes | Primer Sequences | Sizes (bp) |
|---|---|---|
| Beta-actin | 122 | |
| Forward | 5'-TCTATCCTGGCCTCACTGTC-3' | |
| Reverse | 5'-AACGCAGCTCAGTAACAGTCC-3' | |
| GLUT 4 | 231 | |
| Forward | 5'-GCCGGGACACTATACCCT-3' | |
| Reverse | 5'-TGTAGCTCTGTTCAATCACTTTCT-3' | |
| IR | 131 | |
| Forward | 5'-CTTCTTCCGACATGTGGTCCT-3' | |
| Reverse | 5'-AGGGGGATCCAGATAGCCTC-3' |
Sequences of Forward-Reverse Primers of Genes in Real-time Polymerase Chain Reaction
3.3. Statistical Analysis
To obtain the results, the data were reported using descriptive statistics as mean and standard deviation (SD). Then, the Shapiro-Wilk test was used to investigate the normality of data distribution. One-way analysis of variance (ANOVA) and Tukey's post hoc test were used to compare the study groups. All statistical analyses were performed using SPSS software (version 24), and a significance level of P < 0.05 was considered.
4. Results
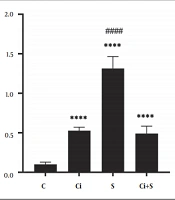
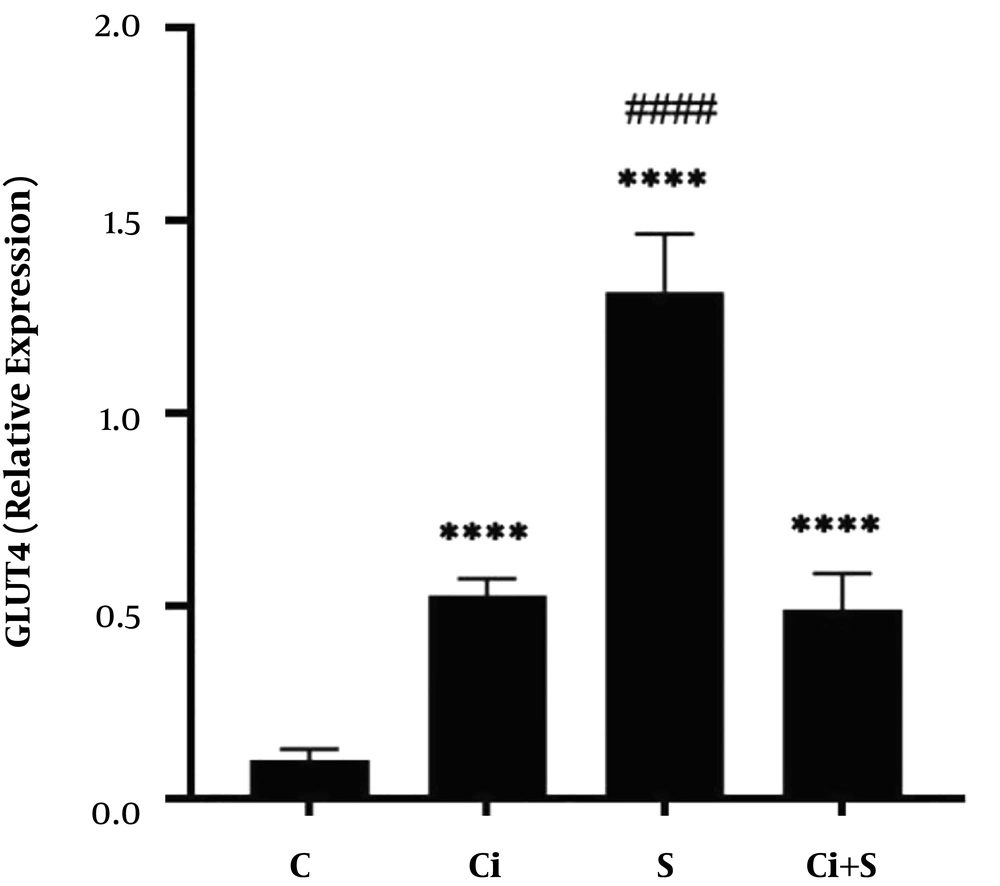
The results of one-way analysis of variance (ANOVA) showed that six weeks of swimming training with cinnamon consumption (P = 0.001, F = 216.00) had a significant effect on increasing GLUT4 gene expression in the brown adipose tissue of diabetic rats. The results of Tukey’s post hoc test showed that GLUT4 gene expression in the brown adipose tissue in the Ci, S, and Ci + S (P = 0.0001) groups was significantly higher than the C group. Also, GLUT4 gene expression in the brown fat tissue in the S group (P = 0.0001) was significantly higher than the Ci + S and Ci group. There was no significant difference in GLUT4 gene expression in the brown fat tissue of diabetic rats between the Ci + S group (P = 0.89) compared to the Ci group (Figure 1).
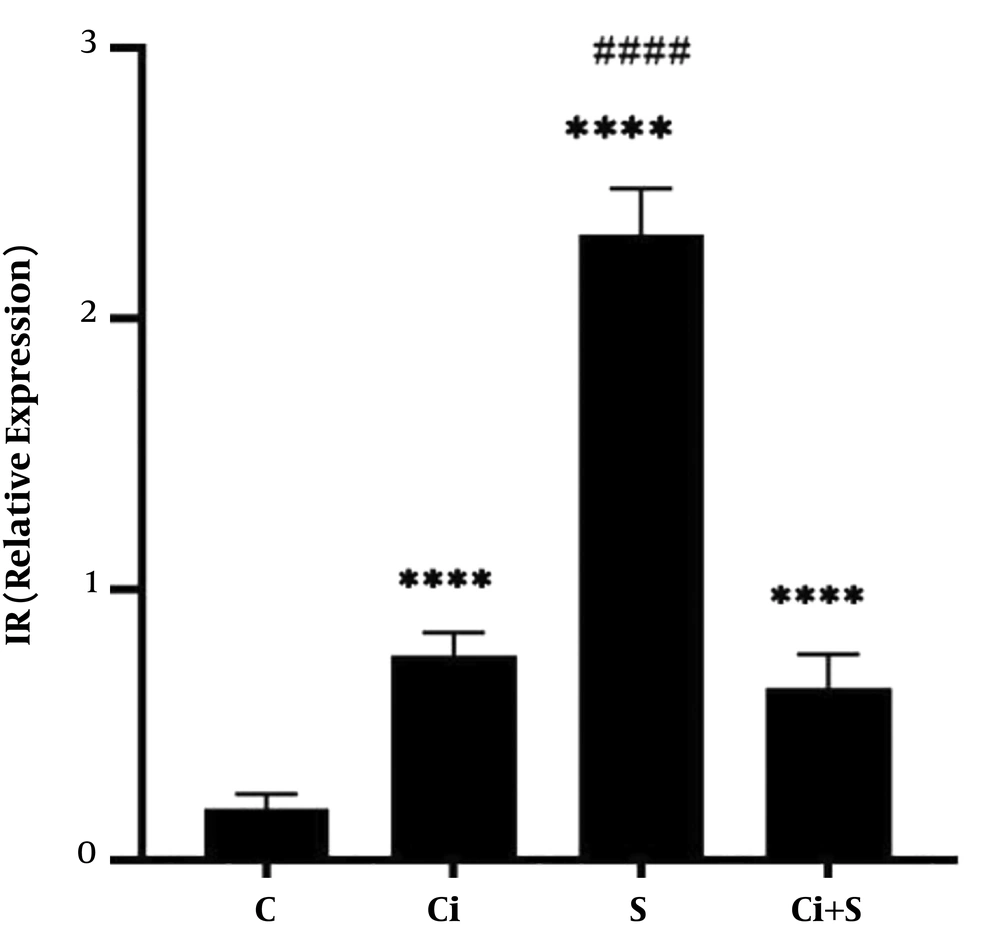
The results of ANOVA showed that six weeks of swimming training with cinnamon consumption (P = 0.001, F = 424.30) had a significant effect on increasing IR gene expression in the brown fat tissue of diabetic rats. The results of Tukey’s post hoc test showed that IR gene expression in the brown fat tissue in the Ci, S, and S + Ci (P = 0.0001) was significantly higher than the C group. Also, IR gene expression in the brown fat tissue in the S group (P = 0.0001) was significantly higher than the Ci + S and Ci groups. There was no significant difference in IR gene expression in the brown fat tissue of diabetic rats between the Ci + S group (P = 0.27) compared to the Ci group (Figure 2).
5. Discussion
The results of the present study showed that six weeks of swimming training had a significant effect on the gene expression of GLUT4 and IR in the brown adipose tissue of diabetic rats. One method of inducing diabetes in rodents is the intraperitoneal injection of streptozotocin (STZ). Streptozotocin-induced diabetes mellitus leads to a deficiency of pancreatic beta cells, decreased insulin secretion, and decreased GLUT4 gene expression. Research findings by Park et al. (2011) showed that induction of diabetes significantly reduces GLUT4 protein levels; this finding is also obtained in the present study. Interestingly, STZ-dependent changes in GLUT4 gene expression in adipose tissue occur earlier than skeletal muscle (18). The results of the present study are in line with the results of Park (2011) and Machrina (2018) studies (15, 18), yet they are inconsistent with the results of Zare et al.'s research (19). Some researchers have suggested that exercise can increase insulin response by increasing GLUT4 and IR substrates (20). Physical activity affects glucose homeostasis and increases insulin sensitivity by increasing the function and signaling of insulin, increasing glucose transporters from the inside to the cell membrane, increasing the rate of glucose uptake, increasing capillary density, increasing gene expression or activities of various proteins involved in insulin messaging, increasing glycogen synthetase activity, and finally, increasing glycogen storage (21). The results of Park et al.’s (2011) study showed that exercise increased the expression of GLUT4 protein in diabetic rats (18). One of the effective factors in the expression of GLUT4 is the increase in kinase activity due to exercise. AMP-dependent protein kinase, which is activated by increasing the ATP/AMP ratio, is a key intracellular signaling pathway in glucose uptake (22). Cortisol released during exercise increases the rate of gene transcription. The results of some studies have shown that the amount of cortisol in the blood plasma during exercise increases significantly with moderate to severe intensity. High cortisol levels, enzyme metabolism, and PGC-1 alpha activation stimulate IR gene transcription (23).
Other results of the present study showed that six weeks of cinnamon consumption had a significant effect on GLUT4 gene expression and IR in the brown adipose tissue of diabetic rats. The results of the present study are consistent with the results of Solomon et al.’s (2009) study (24), whereas inconsistent with the results of Wang’s study (25). Cinnamon extract has been shown to reduce blood glucose levels and improve insulin resistance in rats by increasing insulin activity by up to 20-fold and increasing glucose metabolism by several times in fat cells (26). The methyl hydroxy chalcone polymer in cinnamon activates insulin receptor-kinase enzyme and inhibits the action of insulin-receptor phosphatase enzyme in adipocytes, which increases the sensitivity of these cells to insulin and decreases insulin resistance (26). The results of some studies showed that the compounds in cinnamon increase insulin action three times and decrease insulin resistance in epidermal fat cells of rats (27). Cinnamon stimulates glycogen synthesis by activating IR and increases glucose uptake. The bioactive complements extracted from cinnamon are like a hydroxychalcone polymer that mimics the action of insulin and maximize insulin receptor phosphorylation by activating the insulin receptor kinase and inhibiting insulin receptor dephosphorylation. All of these effects increase insulin sensitivity and decrease insulin resistance (28).
Regarding the interactive effects of swimming training and cinnamon consumption, the results of the present study showed that six weeks of cinnamon consumption had interactive effects on the gene expression of GLUT4 and IR in the brown adipose tissue of diabetic rats. Aerobic training facilitates GLUT4 transfer to the cell membrane; as a result, glucose uptake in active adipose tissue and skeletal muscle is increased by protein carriers (29). Another problem in the diabetic group is the lack of inhibition of two key enzymes, gluconeogenesis PEPCK, and glucose 6-phosphate, and there is evidence that these two enzymes are involved in insulin resistance. Exercise seems to reduce fasting glucose by affecting the expression of these two proteins and inhibiting the key enzyme gluconeogenesis PEPCK and catalytic units of glucose 6-phosphate. Research also shows that exercise increases the substrate of IR in adipose tissue and muscle (30). Aerobic exercise increases the expression of GLUT4 in the sarcolemma membrane and facilitates the transfer of GLUT4 to the cell membrane. As a result, uptake is increased in adipose tissue and active skeletal muscle by protein carriers (31). In laboratory studies, it has been shown that cinnamon extract increases the phosphorylation activity of beta-IR; meanwhile, it reduces the activity of tyrosine phosphatase, thereby showing insulin-like properties. A number of studies have introduced stimulating insulin secretion and preventing the increase of cellular resistance to insulin as the mechanism of cinnamon action; also, like insulin hormone, cinnamon polyphenols have been shown to stimulate glucose uptake and stimulate glycogen biosynthesis by activating glycogen synthetase kinase (28). Regarding the modulatory effect of cinnamon on GLUT4 and IR levels, exercise seems to have insulin-independent pathways. This mechanism appears to be partly related to oxidative phosphorylation dependent on protein kinases B, A, and cell redox pathways. As a result, increasing oxidative stress in each exercise session activates the forkhead box O (FOXO) transcription factor pathway, which, in turn, activates nuclear respiratory factor (NRF1/2). However, some researchers believe that the use of dose-dependent antioxidants has a beneficial effect on the cell status, modulating the effect of exercise on biological adaptation by inhibiting redox-dependent pathways (32, 33). Considering the effect of exercise activities on non-insulin-dependent pathways, it seems that the lack of study of oxidative phosphorylation pathways such as protein kinases and cAMP is one of the limitations of the present study. Therefore, it is suggested that these factors be considered in future studies. Also, considering that in this study, cinnamon consumption moderated the effects of training, it seems that different doses of this medicinal plant along with exercise on glucose metabolism in brown adipose tissue need further studies. Therefore, it is suggested that different doses of cinnamon should be evaluated in future studies.
5.1. Conclusion
The results of the present study generally show that swimming training with cinnamon consumption has an effect on GLUT4 and IR in the brown adipose tissue of diabetic rats, but its effect is less than swimming training alone.