1. Background
Pregnancy brings about adaptive changes in the metabolism (1) and physiology of the female body (2). These gestational changes may be associated with disorders like pregnancy-associated liver disease observed in 3% of pregnant women worldwide (3, 4). The higher metabolic demand of the growing fetus and the increased metabolic activity in placental mitochondria leads to increased production of reactive oxygen species (ROS) that puts the mother in a state of oxidative stress (5).
Previous studies have shown that ethanol (Eth) consumption during pregnancy can increase ROS levels and cause liver damage (6, 7). Eth induces both oxidative and ER stress in the liver (8). The brain is another organ that is most susceptible to Eth toxicity following its oxidation (9). Chronic administration of Eth is reported to accompany oxidative stress in the adult mouse brain (10).
Unfolded protein response (UPR), as a protective strategy protecting cells against ER stress (11), works toward the reduction of unfolded protein accumulation (12). UPR activity involves the three signaling proteins, activating transcription factor 6 (ATF6), protein kinase R (PKR)-like endoplasmic reticulum kinase, and inositol-requiring enzyme 1 (IRE1) (13). The activation of these proteins occurs upon detachment from an endoplasmic reticulum chaperon, GRP78/BiP (binding immunoglobulin protein), when the accumulated unfolded proteins compete with the receptors attachment to GRP78 (14, 15). Neuronal death after prolonged activation of UPR following ER stress has been introduced as the mechanism of ethanol neurotoxicity (16).
Autophagy plays a protective role against oxidative stress (17). Beclin1/ATG6 (autophagy-related gene 6) is a key protein in the initiation of autophagy that directly interacts with GRP78 leading to the formation of the BECLIN1-Vps34-Vps15 complex and eventually triggering the autophagy cascade (18). Autophagy is shown to be activated by Eth exposure (19).
As a polyphenolic compound, resveratrol (RSV) is found in various foods and drinks such as grapes, plums, berries, peanuts, grape juice, and red wine (20). Numerous beneficial health effects of RSV, such as preventing cardiovascular diseases, diabetes, aging, neurodegenerative complications, and cancer, have been discovered, where RSV acts as a potent antioxidant (21). RSV has shown alleviating effects on fatty liver disease by introducing autophagy in cellular and animal models of hepatic steatosis (22, 23). Besides, long-term consumption of RSV is associated with protective effects on optic nerves via activation of UPR pathway effector proteins, CHOP (CCAATenhancer-binding protein homologous protein), and XBP-1 (X-box binding protein 1) (24).
We have previously studied the brain effects of alcohol consumption during pregnancy and have shown that Eth decreased the expression of brain-derived neurotrophic factor (BDNF) in the hippocampus of female rats, which was reversed by RSV (25).
2. Objectives
Here, we have hypothesized that pregnancy affects the basal mRNA expressions of genes for autophagy and UPR and affects their response to Eth and RSV.
3. Methods
3.1. Rats
Twenty-four female Sprague-Dawley rats were purchased from Shiraz University of Medical Sciences’ Animal Laboratory with an average weight of 220 ± 20 g. Subjects were kept in a controlled condition using a 12-hour light and dark cycle. Six nulligravida female rats were randomly selected as the non-pregnant control group. Other animals mated and were examined for pregnancy. The detection of the first vaginal plaque was an indication of pregnancy. Pregnant rats were divided into three groups and treated with normal saline (pregnant control group) and either Eth or combined Eth and RSV as test groups (Table 1). The doses are selected based on our results from previous studies (25, 26). Rats were gavaged by 200 µL of the daily treatments for 20 days. At the end of the treatment period, rats were euthanized in a CO2 chamber, and their brains and livers were surgically removed, snap-frozen in liquid nitrogen, and stored at -80°C till the time of RNA extraction. Two brain samples of each group were pooled. All experiments were done in agreement with the animal ethics guideline of Shiraz University of Medical Sciences and were approved by the Vice-Chancellor for Research Affairs with grant number 91-6140.
3.2. Quantitative-reverse Transcription Polymerase Chain Reaction (qRT-PCR)
RNA was extracted from 50 - 100 mg of tissues using Trizol reagent (Invitrogen, CA, USA), following the manufacturer’s protocol. First-strand cDNA was synthesized from RNA with reverse transcriptase Kit (Fermentas, Maryland, USA). Quantitative analysis was carried out by Real-Time RT PCR using the Power SYBR Green PCR Master Mix (Takara) and Real-Time PCR System (Applied Biosystems 7500, Foster City, CA, USA). qRT-PCR was performed with 40 cycles for amplification. We evaluated single-peak melting curves as specific qRT-PCR reactions. Electrophoresis of qRT-PCR products confirmed the accuracy of the molecular weight of amplicons.
The primers of BECLIN1, GRP78, and ACTIN were designed by AlleleID software (Table 2). The 2-ΔΔCt method was used to evaluate the expression of genes.
| Gene | Accession Number | Sense Sequence (5’ to 3’) | Anti-sense Sequence (5’ to 3’) | Product Size |
|---|---|---|---|---|
| ACTIN | NM-031144.3 | CCACACCCGCCACCAGTTCG | CTAGGGCGGCCCACGATGGA | 138 |
| BECLIN1 | NM-001034117.1 | CCAATGTCTTCAATGCGACCTTC | GGCAGCATTGATTTCATTCCAT | 119 |
| GRP78 | NM-013083.2 | GTTCTTGCCATTCAAGGTGG | TGGTACAGTCACAACTGCATG | 181 |
Primers Used for Real-time Analysis
3.3. Statistical Analysis
Nonparametric analysis of variance (Kruskal-Wallis test) followed by Dunn’s post hoc analysis for multiple comparisons was used to determine any significant changes between groups. The significance level was set at P < 0.05 for all tests. All analyses were conducted with Graph Pad Prism statistical software (version 7; San Diego, CA).
4. Results
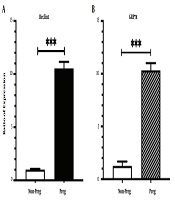
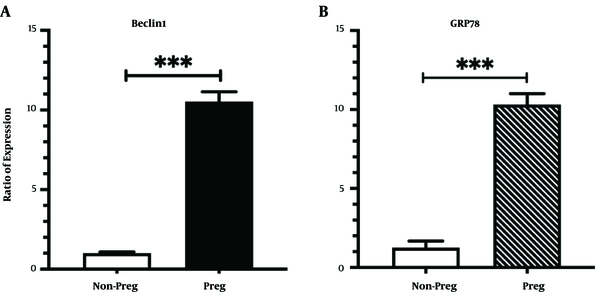
4.1. Pregnant Rats Express Higher Levels of Autophagy and UPR Markers in Their Liver
As shown in Figure 1, BECLIN1 encoding mRNA levels in the liver of pregnant rats treated with normal saline were significantly higher compared to the non-pregnant control group (Figure 1A, P-value < 0.001). Similar results were observed for the expression level of GRP78 (Figure 1B, P-value < 0.001).
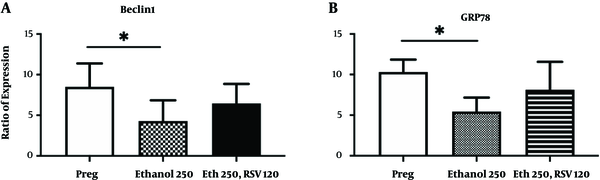
4.2. RSV Restored the Expression of Autophagy and UPR Markers in the Liver of Pregnant Rats Exposed to Ethanol
Results of real-time RT-PCR showed that Eth 250 mg/kg.BW significantly decreased the expression levels of BECLIN1 mRNA in the liver of pregnant rats compared to the normal saline group (Figure 2A, P-value < 0.05). However, RSV 120 mg/kg.BW reversed the ethanol-induced decrease in the mRNA levels of BECLIN1. Likewise, the expression levels of GRP78 mRNA significantly decreased in response to Eth 250 mg/kg.BW in the liver of pregnant rats compared to pregnant control rats (Figure 2B, P-value < 0.05). Addition of RSV 120 mg/kg.BW to Eth could effectively increase the expression of GRP78 to the level of normal saline receiving pregnant rats.
Levels of BECLIN1 and GRP78 transcripts in the liver of pregnant rats treated with Eth alone or combined with RSV. Control pregnant rats were treated with normal saline. Data are presented as the fold of increase in the mRNA level in the treatment groups to that in the pregnant control group. Data are mean and SEM (n = 3). *, significance at P < 0.05 (Dunn’s test).
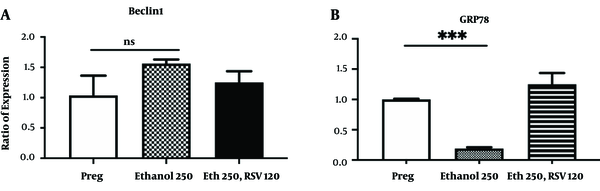
4.3. RSV Increased the Expression Level of the UPR Marker in the Brain of Pregnant Rats with no Significant Effect on the Expression of the Autophagy Marker
Results of Real-time RT-PCR showed that Eth 250 mg/kg.BW alone or combined with RSV 120 mg/kg had no significant effects on the mRNA expression of the BECLIN1 in the brain of pregnant rats compared to the saline-treated pregnant rats. (Figure 3A). However, in response to oral Eth 250 mg/kg.BW, the expression levels of the GRP78 mRNA, significantly decreased in the brain of pregnant rats compared to saline-treated pregnant rats (Figure 3B, P-value < 0.0001). Addition of RSV 120 mg/kg.BW to Eth leads to an increase in the mRNA expression of GRP78 compare to saline receiving pregnant rats (Figure 3B, P-value < 0.05).
5. Discussion
The present study revealed increased levels of mRNAs encoding GRP78 and/or Beclin-1 in the liver of pregnant rats.
GRP78 is a negative regulator of UPR and a strong inhibitor of apoptosis (27). GRP78 has an ER stress response element in the promoter of its gene. Thus, GRP78 is shown to be a marker and regulator of ER stress (28). It is established that pregnancy is associated with ER stress in the placenta and reproductive systems due to induced oxidative stress (29). However, this pregnancy-related oxidative situation on other organs like the liver and brain is not apparent yet. Our results revealed an up-regulation of GRP78 expression in pregnant rats’ liver, possibly as a protective function of the liver in stress conditions.
In this study, we have also shown that the expression levels of BECLIN1 were increased in the liver during pregnancy. Pregnancy is associated with increased levels of ROS, and autophagy is triggered by oxidative stress (30). Therefore, the overexpression of the BECLIN1 gene as an autophagy marker in the liver of pregnant rats can be interpreted as a defense mechanism against ROS’s gestational overload, which prevents liver injury and reduces oxidative stress through autophagic activity. Autophagy has also been shown in humans’ first trimester placental villi, a finding that suggests a pro-survival role for autophagy during pregnancy (31).
Previous studies have suggested that acute Eth exposure induces autophagy in the liver through its reactive metabolites (32). We have demonstrated a decrease in the expression levels of BECLIN1 upon chronic exposure of pregnant rats to Eth. In line with our results, Chao et al. reported that impaired transcription factor EB (TFEB) -mediated autophagy promoted the liver injury caused by chronic ethanol intake in mice. Autophagy is also shown to protect hepatocytes from adverse effects of acute alcohol exposure by removing lipid droplets and damaged mitochondria (32). Therefore, lower levels of BECLIN1 we observed in the liver of pregnant rats exposed to chronic Eth are conceivable to decrease autophagic activity and predispose the tissue to injury.
Despite the effect on the liver, Eth failed to decrease the expression of BECLIN1 in the brain samples from our pregnant rats. This finding may be explained by the high basal level of autophagic activity reported in cells with neuronal origin (33).
Recent studies have introduced RSV as an inducer of autophagy. Other investigators have shown that beneficial effects of RSV on the hepatic lipid accumulation induced by ethanol or by high-fat high-sucrose diet associated with the elevated levels of BECLIN1 and the activation of autophagy (22, 23). In line with these studies, our results have shown that RSV 120 mg/kg.BW tended to reverse the decreased expression of BECLIN1 in the Eth treated pregnant rats.
In 2018, Li et al. (34) reported that Eth exposure during pregnancy increased the expression of GRP78 and other ER stress markers such as IRE1α and ATF4 (Activating Transcription Factor 4) in the brain of four-day-old offsprings. However, our results demonstrated decreased GRP78 transcripts in the brain of pregnant rats chronically exposed to ethanol. RSV has shown ameliorating effects on the ER stress induced by free fatty acid in HepG2 cells (35). Further, Gaballah et al. (36), in a rat model of rotenone-induced Parkinson’s disease, showed that RSV alleviated ER stress by decreasing the expression of CHOP and GRP78. Despite these findings, our results revealed that RSV recovered the reduced expression of GRP78 partially in the liver and fully in the brain of Eth-treated pregnant rats. The discrepancy between our results and those of other researchers may be attributed to the physiological and metabolic situations in pregnancy, as we have observed a higher basal level of GRP78 expression in the liver of pregnant rats compared to the non-pregnant group. Alternatively, the discrepancy may be due to the effect of RSV on decreasing the oxidative injury via the activation of TFEB and subsequent induction of autophagy as has been shown in endothelial cells (37).
This study is limited by evaluating the effect of pregnancy on autophagy and UPR marker to the transcriptome level. These observations need to be confirmed by evaluating the protein level of these markers.
5.1. Conclusions
Here, for the first time, we have disclosed that the basal mRNA levels of autophagy and UPR marker are higher in the liver of pregnant rats compared to non-pregnant ones. Further, we have shown that chronic consumption of Eth disturbs the pregnant levels of these markers and that RSV could recover them.