1. Background
Alzheimer’s disease (AD) is one of the most common neurological disorders that are prevalent worldwide. In the pathogenesis of this disease, complex factors such as environmental factors, biological factors, and mutations in some genes (amyloid β (Aβ), amyloid precursor protein (APP), persinilin 1 and 2 (PS1/2)) are involved (1, 2). Increased amyloid plaques and hyperphosphorylation of tao protein increase neuroinflammation. This neuroinflammation leads to increased oxidative stress, activated nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, and interactively with increasing inflammation leads to disruption of ATP production in neurons (1). It is also believed that neuronal loss following AD plays a role in transcriptional dysfunction of metabolic genes and impaired DNA methylation (3). Thus, disruption of enzymes responsible for DNA splicing, such as O-6-methylguanine DNA methyltransferase (MGMT), inhibits DNA-dependent ATPase-encoding genes (4, 5).
However, due to the rapid progression of this disease, researchers consider lifestyle changes as effective ways to treat chronic and non-communicable diseases so that researchers have suggested that aerobic and endurance training have beneficial effects on brain function, neuroplasticity, cerebral blood flow, and memory and learning in these patients (6). Exercise seems to activate some of the DNA splicing enzymes, such as 8-oxoguanine DNA glycosylase-1 (OGG1), and reduces DNA damage in muscle and brain by activating the respiratory nucleus proteins 1 and 2 (NRF1/2) and sirtoin 1 and sometimes modulating cellular redox (7). Researchers have reported beneficial effects of aerobic exercise on the mental, physical and cognitive health of patients with AD, the intensity, duration of activity, and the most effective duration of exercise, as well as the type of exercise based on differences in contraction and related adaptations, are not yet fully understood (8). In this regard, the results showed that endurance training reduced C-reactive protein in the muscle tissue of AD rats (9). In addition, endurance training on different slopes has been one of the challenges in assessing the type of adaptation in the cognitive function of AD animal models. Accordingly, the researchers showed that the positive slope running (ETPS) reduced the pain tolerance threshold, but the negative slope running had no significant effect on it (10); eight weeks of voluntary and forced training improved avoidance memory and spatial memory in AD rats (11). Also, the endurance training, resistance training, and combined training have beneficial effects on reducing DNA damage in the brain tissue. Brain-derived neurotrophic factor (BDNF), cAMP-activated protein kinases, and activation of cyclic AMP response element-binding (CREB) protein reduce 8-hydroxy-2-deoxyguanosine and decrease DNA damage (12); eight weeks of aerobic exercise reduced oxidative stress and increased MGMT expression (13, 14).
On the other hand, the use of natural antioxidant supplements has been considered by researchers. Among the herbal antioxidant supplements, royal jelly (RJ), due to its anti-inflammatory, antioxidant, anti-diabetic effects, and beneficial impacts on neurotrophins has favorable effects on patients with AD. In this regard, researchers have shown that the use of royal jelly by decreasing oxidative stress, increased neuroplasticity and BDNF in the brain tissue of AD rats (15). Consumption of honey and its products also improves nerve tone function, reduces oxidative stress, reduces apoptosis, and reduces DNA damage in the brain tissue (16). In another study, researchers showed that the use of RJ by cAMP/PKA/CREB/BDNF mechanism inhibits nerve cell apoptosis and reduces DNA damage (17). Although in previous studies, the favorable interactive effects of training and RJ consumption on neurotrophins (15), cognitive function (11), and inflammatory factors (9) have been reported, the exact mechanism of these two interventions on the pathways for DNA repair and transcription of metabolism-related genes (ATPase) are not yet fully understood.
2. Objectives
On the other hand, due to the increasing prevalence of AD and the need for effective treatment methods to reduce the complications of this disease and reduce brain damage in the patients, it seems that conducting basic studies in this field provides more information to researchers in the field of nutrition and sport; therefore, the present study aimed to investigate the effects of eight weeks of ETPS and ETNS with RJ consumption on MGMT and ATPase gene expression levels in the hippocampus tissue of trimethyltin (TMT)-induced AD rats.
3. Methods
In this experimental trial, 42 male Sprague-Dawley rats were prepared and kept for one week in the Animal Breeding Center of the Laboratory of the Islamic Azad University in order to adapt to the environment.
In the present study, all the ethical principles of working with laboratory animals were observed in accordance with the Helsinki Agreement and the Ethics Committee of the Islamic Azad University, Marvdasht Branch. Also, during the research period, standard conditions for keeping rats were provided, including keeping in washable Plexiglas cages, temperature 22°C to 24°C, humidity 55 to 60%, 12-hour light-dark cycle, and access to water and food ad libitum (18).
Then, on day 8, to induce AD, 36 fasting rats were exposed to intraperitoneal injection of 8 mg/kg neurotoxin trimethyltin (Sigma Aldrich). Two weeks after TMT injection, the Shuttle box and Maze Y tests were performed to diagnose AD and its effect on the hippocampus. To evaluate AD induction, the results of Y maze and shuttle box test in the TMT injection group were compared with the healthy control group. In addition to clinical signs such as aggression, bruising, and bleeding around the eyes, rats’ tail tremors were other symptoms that the researcher used to identify rats with AD.
Rats with AD were then randomly assigned to six groups, including: (1) control + RJ dissolvent (Sham/Sh); (2) Endurance training on positive slope (ETPS); (3) Endurance training on negative slope (ETNS); (4) ETPS + royal jelly (RJ) (ETPS + RJ); (5) ETNS + RJ; and (6) RJ groups. In addition, 6 healthy rats were placed in the healthy control (HC) group to evaluate the effect of disease induction on the research variables.
The ETPS (+ 15) and ETNS (-15) groups trained for eight weeks, five 60-minute sessions per week at 16 m/min (10). The daily dose of royal jelly prepared by Marvdasht Agricultural Jihad and approved by the experts of this center was 100 mg/kg RJ as peritoneal injection (10). Subsequently, 48 hours after the last training session, the rats were anesthetized using a combination of ketamine and xylazine. After ensuring complete anesthesia and performing analgesia test, the hippocampus was carefully isolated by laboratory specialists.
3.1. Running on Positive and Negative Slopes
Initially, to familiarize the rats, they ran three sessions per week for one week at 5 to 10 m/min for 5-10 min on positive and negative slopes. Rats were trained on a 5-channel treadmill made by the Iranian Danesh Salar Company. Then rats in the endurance training on positive slope (ETPS) (15%) trained for eight weeks, five sessions per week and each session 60 minutes at a speed of 16 m/min and rats in the endurance training on negative slope (ETNS) trained for eight weeks, five sessions per week and each session 60 minutes at a speed of 16 m/min (15%). Endurance training on positive and negative slopes was considered on a 9-degree slope for the first week, and one degree was added to the slope every week; also, 5 minutes of running at a speed of 8 m/min was considered for warming up and cooling at the beginning and end of the training (10).
3.2. Measurement of MGMT and ATPase Gene Expression Levels
To measure MGMT and ATPase gene expression levels, total cell RNA content was first extracted using the Hong Kong-made RNA extraction column kit (FavorPrepTM Tissue Total RNA Mini Kit) according to the manufacturer’s instructions.
The first step in extracting RNA from animal cells was to remove the cell wall using a lysing buffer called RB Buffer. In the following, 350 µL of RB Buffer was added to the sample (centrifuged cell pellet) (10 µL of mercaptoethanol-β per 1 mL had already been added to the buffer) and placed at room temperature for 5 min. Then the filter column was placed in a collection tube, and the sample mixture was transferred to the filter column and centrifuged at 14,000 rpm for 2 minutes. After centrifugation, the clear solution was transferred from the collection tube to a new microfluidic tube. Afterward, the same volume of 350 µL ethanol 70% was added to it, and then it was well vortexed.
The RB mini-column was placed in the collection tube, and the sample to which the ethanol was added was transferred to the RB mini-column and centrifuged at 14,000 rpm for 1 minute, and the solution in the collection tube was discarded. In the next step, 500 µL of wash buffer1 was added to the RB mini-column and centrifuged at 14,000 rpm for 1 minute, and the solution in the collection tube was discarded. The RB mini-column was then centrifuged with 750 µL of wash buffer2 at 14,000 rpm for 1 minute, and the solution in the collection tube was discarded. This step was repeated twice. The centrifuge was then performed for 3 minutes at 14,000 rpm. The RB mini-column was then placed in an elution tube, and 50 µL of RNase-free ddH2O was added to the RB minic-olumn; it was given 1 minute and then centrifuged at 14,000 rpm for 2 minutes. The solution in the elution tube was the extracted RNAs, which were stored at -70°C.
To ensure the quality of the extracted RNA, 5 µL of it was read on agarose gel electrophoresis as well as its absorption at 260 nm with Sigma (made in USA) pico drop device. The resulting RNA was then stored at -80°C until use. The following formula was also used to evaluate the quality of RNA.
C (µg/µL) = A260 × ε × d/1000
Afterward, cDNA synthesis was provided according to the instructions in the Fermentas kit (K1621). The reverse transcription reaction was performed using RevertAidTM M-MuLV Reverse transcriptase enzyme.
When preparing the cDNA from the purified sample following reading the adsorption, a volume containing 1,000 ng of RNA was taken, then 0.5 µL of random hexamers and 0.5 µL of oligodT primer (oligodeoxy ribonucleotide, used as a primer to start cDNA synthesis) were added to it and 12 µL of DEPC water was added and transferred to 65°C for 5 min. It was then placed on ice for 2 minutes to disintegrate secondary RNA structures. In the next step, 4 µL 5X reaction buffer, 2 µL dNTP, 1 µL RiboLock RNase inhibitor, and 1 µL RevertAid RT were added to the previous mixture, which was placed at 65°C for 5 minutes.
In the next step, to evaluate the expression of genes using Real-time PCR, all primers were designed by Allele IDv7.8 software, and β2m gene (beta 2 microglobulin) was used as internal control (Table 1); all primers were designed as exon-exon connections. After completing the activity of the device and observing the graphs based on increasing the number of desired fragments and the amount of fluorescence propagation, by calculating ΔΔCt, the amount of change in the expression of the desired gene compared to B2m and the control group was calculated using the following formula.
ΔCt = Cinterest - CtB2m
ΔΔCt = ΔCt Treated - ΔCt Un-treated
| Genes | Primer Sequences | Product Size, bp |
|---|---|---|
| B2m | Forward: 5’- CGTGCTTGCCATTCAGAAA -3’ | 244 |
| Reverse: 5’-ATATACATCGGTCTCGGTGG -3’ | ||
| MGMT | Forward: 5’- CGATGAGGAGCAATCCAGTCC-3’ | 228 |
| Reverse: 5’- CTCAATTTCAGCCAGACGGC-3’ | ||
| ATPase | Forward: 5’- CTTGATAAACTGGAGCCCAGTAAGA-3’ | 103 |
| Reverse: 5’- CATCAGACCTGATATTGCCCAAGAG-3’ |
3.3. Statistical Analysis
The one-way analysis of variance (ANOVA) and Tukey’s post hoc test in Graphpad prism 8.3.0 software (P ≤ 0.05) were used to examine the differences between the groups.
4. Results
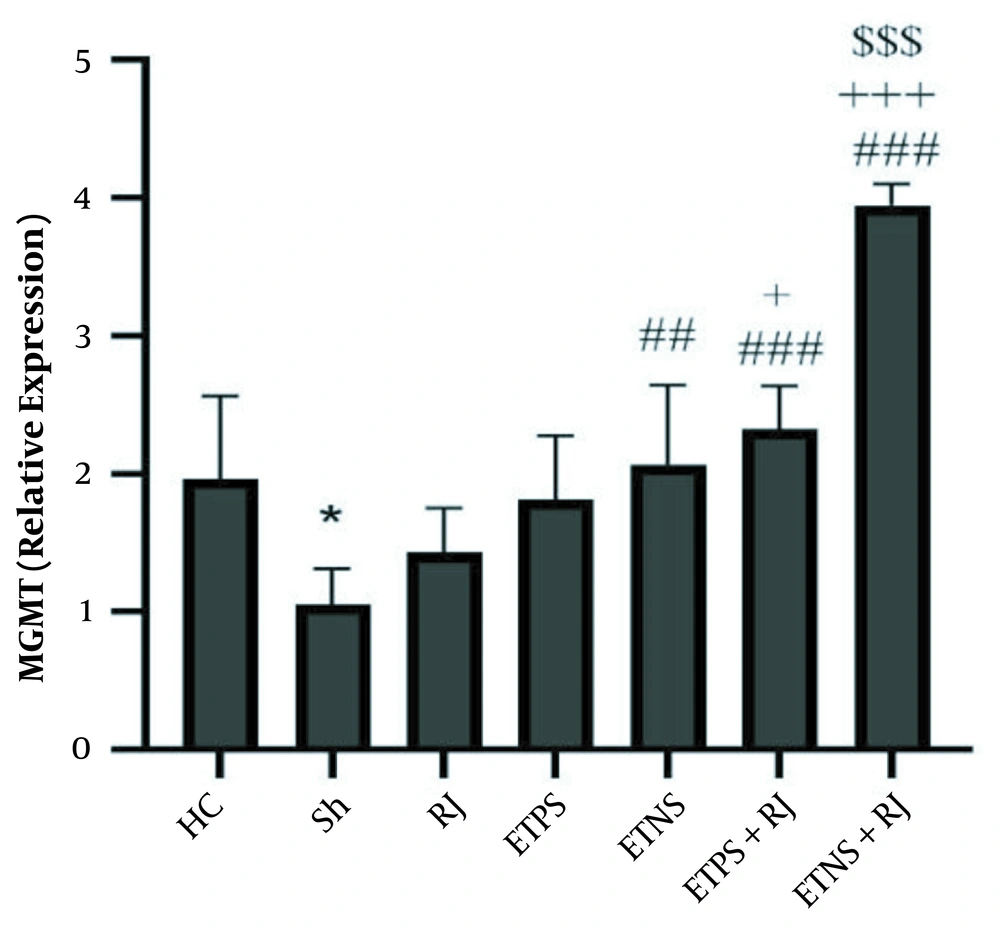
The results of one-way ANOVA showed that there was a significant difference in MGMT (P = 0.001) and ATPase (P = 0.001) gene expression levels in the research groups. The results of Tukey’s post hoc test showed that MGMT levels in the Sh group were significantly lower than the HC group (P = 0.02), MGMT levels in the RJ (P = 0.77) and ETPS (P = 0.08) groups were not significantly different from the Sh group, but in the ETNS (P = 0.008), ETPS + RJ (P = 0.001) and ETNS + RJ (P = 0.001) groups, the levels were significantly higher than the Sh group.
But in the ETPS + RJ (P = 0.02) and ETNS + RJ groups (P = 0.001), the levels were significantly higher than the RJ group. In the ETNS (P = 0.95), ETPS + RJ (P = 0.47) groups, the levels were not significantly different from the ETPS group. In the ETNS + RJ group, the levels were significantly higher than the ETPS (P = 0.001) group; however, no significant difference was observed in the ETPS + RJ and ETNS groups (P = 0.95). In addition, levels in the ETNS + RJ group were higher than the ETNS group (P = 0.001) and in the ETNS + RJ group were significantly higher than the ETPS + RJ group (P = 0.001) (Figure 1).
MGMT gene expression levels in the research group. * (P = 0.02) significant decrease compared to the HC group; ## (P = 0.008) and ### (P = 0.001) significant increase compared to the Sh group; + (P = 0.02) and + + + (P = 0.001) significant increase compared to the RJ group; $$$ (P = 0.001) significant increase compared to the ETPS, ETNS, and ETPS + RJ groups.
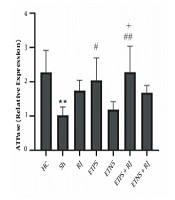
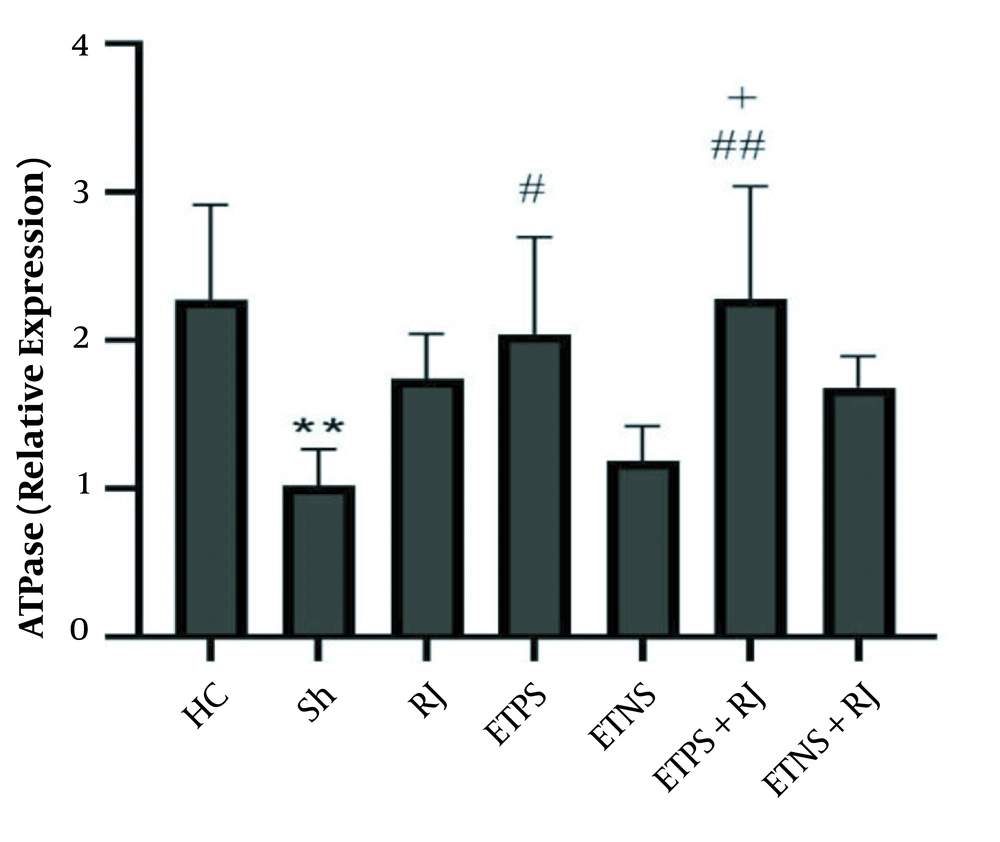
ATPase levels in the Sh group were significantly lower than the HC group (P = 0.005). There was no significant difference in the RJ (P = 0.25), ETNS (P = 0.99), and ETNS + RJ (P = 0.34) groups compared to the Sh group. However, in the ETPS (P = 0.03) and ETPS + RJ (P = 0.005) groups, the levels were significantly higher than the Sh group. There were no significant differences in the ETPS (P = 0.95), ETNS (P = 0.54), ETPS + RJ (P = 0.59), and ETNS + RJ (P = 0.99) groups compared to the RJ group. Also, there were no significant differences in the ETNS (P = 0.11), ETPS + RJ, (P = 0.98), and ETNS + RJ (P = 0.90) groups compared to the ETPS group. ATPase levels in the ETPS + RJ group were significantly higher than the ETNS group (P = 0.02); however, there was no significant difference in the ETNS (P = 0.66) and ETPS + RJ (P = 0.47) groups compared to the ETNS + RJ group (Figure 2).
5. Discussion
The results of this study showed that ETNS and ETPS increased MGMT gene expression in the hippocampus tissue of rats with AD. Moreover, ETPS increased ATPase expression. In addition, mutations in genes such as PS1/2 and Aβ disrupt the function of DNA transcription-dependent protein kinases, DNA polymerases, and gene transcription proteins and reduce MGMT, and eventually metabolic proteins involved in ATP synthesis in AD (19, 20). Researchers have shown that exercise training has beneficial effects on neurons. Even though the role of training on different slopes is still unknown since DNA damage depends on cell regeneration capacity, exercise training begins with the neurotrophic pathway of brain-derived neurotrophic factor (BDNF), which binds to the tropomyosin kinase B receptor and cleaves its intracellular pathway by phosphorylating cAMP response element-binding protein; this continues to modulate the expression of apolipoprotein E1 (APE1), increase 8-exoguanine DNA glycosylase-1, which inhibits 8-hydroxy-2-deoxyguanosine, and decreases DNA methylating agents (12). DNA repair in the nucleus, and mitochondria, appears to improve transcription of enzymes involved in aerobic metabolism (21). Also, phosphorylation of AMPK, increased expression of GLUT-4, NRF-1, and increased expression of protein kinase A, increase the transcription factors of mitochondrial DNA (22).
In this regard, the researchers showed that eight weeks of exercise decreased 8-OHdG and increased MGMT (14). Also, aerobic exercise for eight weeks increased ATP concentration, increased MGMT, and decreased oxidative stress in rats (23). However, there have been different results on the effect of exercise on MGMT levels, and it seems that the type of DNA damage, the baseline levels of oxidative stress, the type of tissue involved in exercise, and its impactability to exercise cause differences in results. For example, a study reported that aerobic exercise reduced MGMT protein levels in the liver tissue of rats exposed to arsenic (13). Although the beneficial role of exercise in improving neurotrophin and DNA repair has been reported, the study of exercise on different slopes was one of the novelties of this study that was not examined in other studies.
The results of the present study showed that RJ had no significant effect on increasing MGMT and ATPase gene expression levels in the hippocampus tissue of rats with AD. Furthermore, RJ enhances angioplasty by increasing the enzyme nitric oxide synthase and inhibits NF-κB and interleukin-1 (IL-1), thereby improving cell and DNA damage to the nervous system and microglia (24). In this regard, researchers have shown that the combination of honey, royal jelly, and propolis increased antioxidants, decreased acetylcholine activity, and also decreased DNA damage markers in the brain (25) and skeletal muscle of elderly rats by reducing oxidative stress (26). In this study, although the results showed that this increase in MGMT and ATPase was not significant, it prevented further damage. Nevertheless, its optimal effects appear to be dose-dependent, in a study by Hosseini et al. (10), 100 mg/kg of RJ had no significant effect on reducing the pain threshold in AD rats.
The results of the present study showed that ETPS + RJ and ETNS + RJ increased MGMT gene expression. Also, ETPS + RJ increased ATPase gene expression in the hippocampus tissue of rats with AD. Although there are limited studies in this regard, it seems exercise with the BDNF/TRKB/CREB mechanism modulates the APE1, increase in NRF-1 and PKA expression reduces DNA damage markers in cells (12, 21) and mitochondrial DNA (22). Nonetheless, depending on the different signaling pathways, the type of exercise depends on the activation of AMPK and mTOR (27), and the type of exercise has different effects on the activation of BDNF/TrkB by activating the cPKCα and PKCβI subunits in the synaptic space (28). Also, dose-dependent RJ reduces nerve cells DNA damage by increasing enzymatic and non-enzymatic antioxidants, amino acids, modulating acetylcholine, decreasing inflammatory factors and apoptosis, increasing nitric oxide synthetase, inhibiting NF-κB and IL-1 (24).
In line with the interactive effect of training and RJ, the researchers showed that training and RJ had an interactive effect on reducing skeletal muscle CRP (9) and increasing hippocampal dopamine expression (29), while forced and optional training with RJ had no interactive effect on increasing NGF and BDNF levels in the hippocampus tissue. Therefore, it seems that the neurotrophin effects of these two interventions in the present study have been influenced by exercise-induced cell-molecular adaptation. Regarding the neuroprotective effects and BDNF-dependent DNA damage, it is suggested that the BDNF/TRKB/CREB pathway be evaluated in training on different slopes in future studies.
In addition, changes in oxidative stress following independent and contextual variables of this study appear to be important factors in the results. Also, the lack of measurement of oxidative stress in this study is another limitation of this study, so it is suggested oxidative stress should be measured both quantitatively and qualitatively in future studies.
5.1. Conclusions
It seems that the training on positive and negative slopes along with royal jelly consumption has an interactive effect on DNA repair markers, while training on a positive slope and royal jelly consumption have an interactive effect on ATPase expression.