1. Background
For decades, cancer has remained one of the major public health issues, even though researchers have made remarkable progress in fighting against this disease (1). Nearly 5% of global deaths are due to skin cancer. There are data indicating a soaring enhancement in cancer patients. Most deaths attributed to skin cancer are due to metastatic melanoma. Metastatic melanoma is the most deadly type of skin cancer caused by the rapid growth of transformed melanocytes from the basal region of the epidermis. Compared to non-melanoma skin cancers, melanoma causes three-quarter of all cancer deaths (2). As it causes high metastatic potential and mortality, human melanoma is introduced as an extremely aggressive skin tumor (3). So much so that the five years survival of cases with an advanced case ranges from 10 to 20%. This poor prognosis is mainly due to resistance to the conventional chemotherapy (4). A375 represents a well-established and characterized melanoma cell line belonging to a relatively undifferentiated and highly metastatic class of melanocytic cells characterized by epithelioid shape and lack of pigmentation (5, 6). HU2 cells like human dermal fibroblasts (HDFs) belong to a dermis cell type with a mesenchymal origin and are found in all connective tissues. Epithelial-mesenchymal interactions and wound healing are greatly influenced by these cells, playing a significant role in extracellular matrix proteins and collagen synthesis and secretion under cell culture conditions (7).
Microtubules, as one of the intra-cellular biofilaments, play a key role in several cell functions such as cellular morphology, cell division, and cellular transportation. These biofilaments, which contain alpha and beta-tubulin, indicate dynamic instability as an unstable function, which is expressed between the phases of growth and shortened during polymerization (8).
In cases with disruption in these necessary structures and defected corresponding biological activities, cells cannot perform a normal cell cycle, which causes apoptosis. Having various diverse natural compounds targeting the tubulin/microtubule system, microtubules have become a main objective for cancer chemotherapeutic substances (9). Tetraoxane paclitaxel (Taxol) can suppress the microtubule dynamics by affecting microtubules and blocking mitosis in the G2-M phase and apoptosis (10).
Taxanes are a class of diterpenes originated from the yew tree (11). In 1994, the Food and Drug Administration (FDA) officially approved the anticancer effect of Taxol (12). The drug has been reported to cause a significant reduction in the dynamic instability of microtubules even at low concentrations (9). Other plant compounds, such as vincristine and vinblastine, are successfully used to treat cancer either alone or in combinations (2). These compounds are potential mitotic inhibitors, which are clinically applied in treating a variety of neo-plasms. While being different in terms of molecular structure by only a carbonyl category, vinblastine and vincristine have revealed different functions concerning their antitumor function and dose-limiting toxicity (13).
Vincristine has an outstanding ability to block microtubule formation, which can be attributed to the dynamics of tubulin protein and the continuous growth of its long chain of building blocks. Dimers, which are merely two building blocks long, are located in tubulin molecule parts that are not much contagious. The high-affinity feature of vincristine can be assumed for tubulin dimers, and their reaction is fast reversible. Thus, the attachment of a vincristine molecule to a dimer at one site can take place, which is proceeded by breaking off, and then there is a reattachment to another site. For this reason, the ability of vincristine to destabilize tubulin is especially significant.
Vinca rosea Linn., a common flowering herb named the periwinkle, can result in the extraction of vinblastine sulfate, which is an alkaloid salt. It is a stathmo-kinetic on the colytic agent. The growing cells are arrested in the metaphase when treated in vitro with this preparation. It is worth mentioning that vinblastine should not be administered intramuscularly, subcutaneously, or intrathecally (14). The side effects of Vinca alkaloids can be decreased by benefiting from the promising strategy of combination therapy, in the sense that combination occurs with other treatments of chemotherapy for increasing the possible impacts. The simultaneous and sequential administration of drugs take place as a cocktail for their therapeutic effect to be maximized (15).
Carboplatin, diamminecyclobutane-1,1-dicarboxylate platinum(II) complex [Pt(cbdc)(NH3)2], is a member of the second generation of platinum-based anticancer drugs. Although both drugs use a similar carrier ligand (NH3), carboplatin has less toxicity in comparison with cisplatin, which means compensating lower consequences for carboplatin’s lower function, which is crucial for its use in cancer therapy (16). Similar to other chemo-drugs, carboplatin causes some negative consequences [e.g., nausea, hypersensitivity, anemia, and genitourinary (10)], and medical attention is needed upon their occurrence. Furthermore, carboplatin is more myelotoxic, which is the central reason for dose decrease (11). Therefore, proposing new therapeutic methods capable of improving carboplatin cytotoxicity effects and reducing its side effects is essential to treat different cancers (17).
2. Objectives
Novel therapeutic methods are required to improve the therapeutic effects of carboplatin and herbal anticancer compounds. To this aim, the present study investigated the possible inhibitions induced in the human melanoma A375 cell line using a combination of Taxol, carboplatin, vinblastine, and vincristine.
3. Methods
3.1. Chemicals
The culture medium, Dulbecco’s Modified Eagle Medium (DMEM), and other chemicals, such as phosphate-buffer saline (PBS) and penicillin-streptomycin, were provided by Inoclon (Iran), fetal bovine serum (FBS) from Gibco (USA), MTT (code 1001904784) from Sigma-Aldrich, Germany), and DMSO from Sigma-Aldrich, (Germany). In addition, Taxol, carboplatin, vinblastine, and vincristine were acquired from Sobhan oncology (Iran), and the plasticware were purchased from Sorfa (China) unless otherwise mentioned.
3.2. Human Cancer and Normal Cell Lines
The human cancer cell lines were purchased from the Iranian Biological Research Center (IBRC). The cell lines employed in the current study included the human malignant melanoma (A375; IBRC C11337) and immortalized human foreskin fibroblast (Hu2; IBRC C10721). All the cells were cultured following the IBRC guidelines. The culture medium was considered to be DMEM and supplemented with penicillin (100 IU/mL), streptomycin (50 mg/mL), and 10% fetal bovine serum. The cells were maintained in a humidified incubator at 37ºC together with 5% CO2.
3.3. Experimental Design
To examine the performance of Taxol, carboplatin, vinblastine, and vincristine on A375 and HU2 cell lines, various concentrations (i.e., 7. 5, 15, 30, 60, 120, and 240 µg/mL) of the drugs were investigated by means of the MTT cell proliferation assay. In these experiments, Taxol and carboplatin were used alone or in combination with each other and vinblastine or vincristine.
3.4. MTT Assay
A375 and HU2 cells were trypsinized at 80 - 90% confluence and plated into 96-well plates at a density of 1 × 104 cells/well overnight. After that, the cells were exposed to various concentrations of Taxol, carboplatin, vinblastine, and vincristine, as well as their combinations for 48 h. The MTT viability test [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide] was carried out according to the descriptions given by Shabani et al. (2017), with some modifications (18). In summary, following incubation, the changing of the cell medium with a fresh one was carried out (100 μL/well), and 50 μL of MTT solution (5 mg/mL in PBS) was added. Afterward, the plates were incubated for 4 h at 37°C, and mixing was used for dissolving the formed formazan crystals in 100-μL DMSO per well. Absorbance was recorded at 570 nm by a 96-well plate reader. The cytotoxicity associated with cell viability was described as the percentage of cell viability of the control. In all the cytotoxicity investigations, the viability of the untreated cells corresponded to 100%. Besides, the IC50 (half maximal inhibitory concentration) values were estimated using the nonlinear regression method via aatbio program (www.aatbio.com).
3.5. Synergy Testing
Interactions among Taxol, carboplatin, vinblastine, and vincristine on A375 cells were assessed by Chou-Talalay method, and CalcuSyn software was used for generating CI values (Biosoft, Cambridge, UK).
3.6. Statistical Analysis
At least three repetitions of each experiment were performed, and SPSS was for data analysis. Comparisons of the mean values were made using one-way analysis of variance (one-way ANOVA) followed by Duncan multiple-range test. The obtained results were expressed as mean ± SEM, A P-value of less than 0.05 was considered statistically significant.
4. Results
4.1. Effects of Taxol, Carboplatin, Vinblastine, and Vincristine on A375 and HU2 Cell Lines
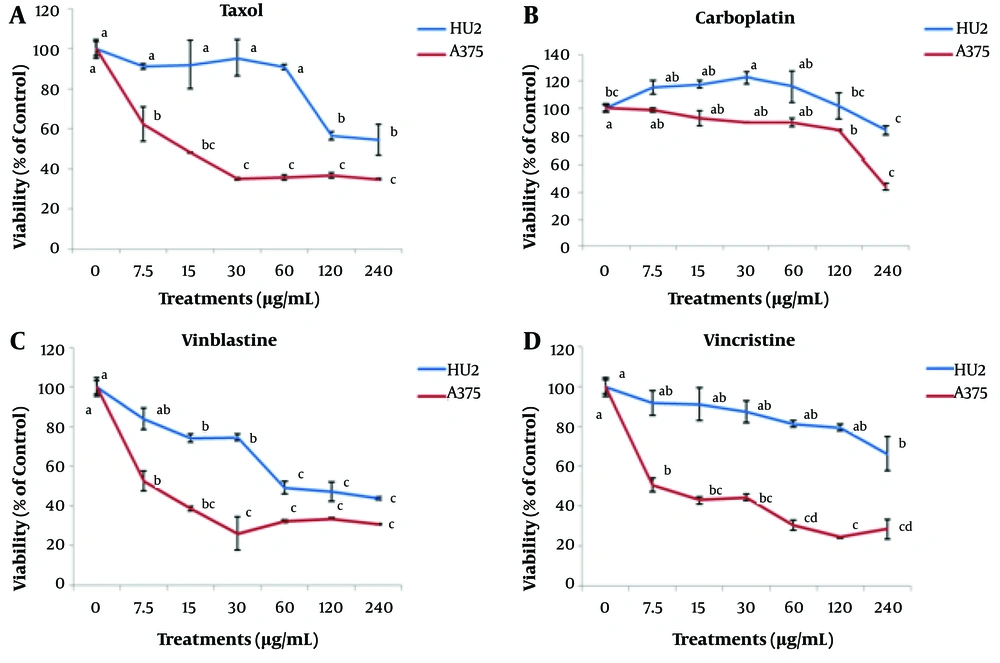
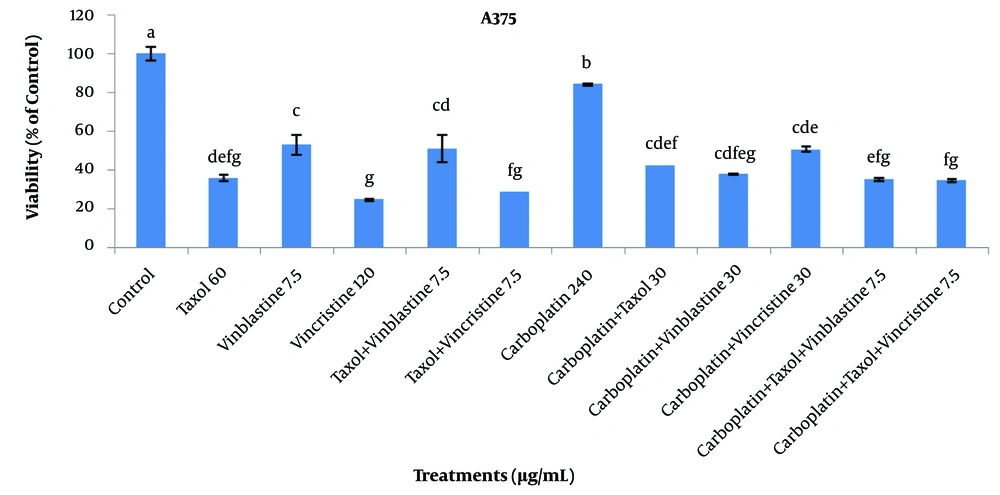
In this experiment, the effect of different concentrations of Taxol, carboplatin, vinblastine, and vincristine (0 - 240 μg/mL) was examined on the survival of A375 cancer cells. By increasing the concentration of Taxol to 30 μg/mL, the survival of A375 cells was significantly decreased. However, no significant difference was observed in the survivals associated with the concentrations equal to and greater than 30 - 240 μg/mL. Based on the results, by increasing the concentration of Taxol up to 60 μg/mL, no significant effect was observed on normal HU2 cells compared to the control. Hence, the concentration level of 30 μg/mL was introduced as the effective Taxol concentration on A375 cells. The survivals of normal and cancer cells at a Taxol concentration level of 30 μg/mL were estimated to be 95 ± 0.9 and 35 ± 0.6%, respectively. In addition, by increasing the concentration of vinblastine up to 30 μg/mL, the survival of cancer cells significantly decreased.
Comparing the effects associated with various concentrations of vinblastine on normal and cancer cells, it could be concluded that the effective concentration of vinblastine for A375 cells was 7.5 μg/mL (with the survival rates of 84 ± 5.6 and 53 ± 4.9% for normal and cancer cells, respectively). The survival of cancer cells was found to be significantly diminished with an increment in the concentration of vincristine, and the greatest effect was observed at a concentration of 120 μg/mL. However, in the case of normal cells, no significant difference was noted with respect to the control treatment up to a concentration level of 120 μg/mL. As a result, the treatment of 120 μg/mL vincristine was introduced as the effective concentration for treating A375 cells. In addition, the survivals of HU2 and A375 cells at a concentration of 120 μg/mL were reported as 79 ± 1.7 and 24 ± 0.6%, respectively. When the concentration of carboplatin was increased up to 60 μg/mL, there was no significant difference in the survival of A375 cells with respect to the control. However, concentration levels higher than 120 μg/mL significantly reduced the survival of cancer cells compared to the control. Due to the fact that no significant difference was observable in the survival of normal cells with respect to the control at a concentration of 120 μg/mL, this concentration was the effective carboplatin concentration in treating A375 cells. At this concentration level, the normal and cancer cells’ viabilities were obtained as 101 ± 9.3 and 84 ± 0.4%, respectively (Figure 1).
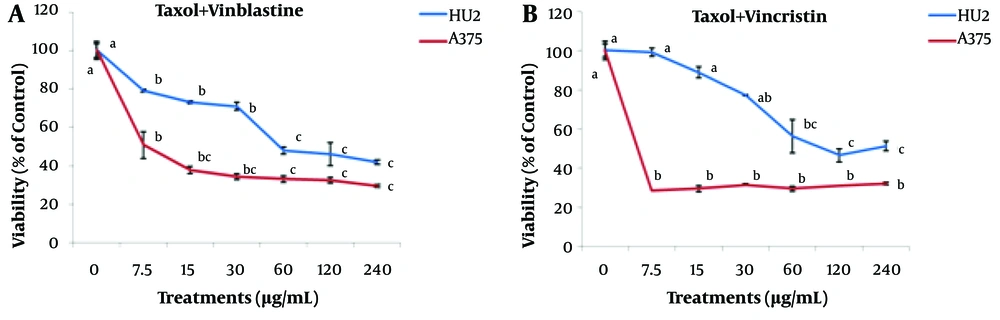
The effect of different concentrations of Taxol + vinblastine and Taxol + vincristine (0 - 240 μg/mL) is depicted in Figure 2. The results indicated a reduction in the survivals of A375 cancer and normal HU2 cells with increasing Taxol + vinblastine concentration. The effective concentration of this compound was found to be 7.5 μg/mL for cancer cells, and at this concentration level, the survivals of normal and cancer cells were obtained as 79 ± 0.7 and 51 ± 6.8%, respectively. The results associated with the Taxol + vincristine treatment showed that 7.5 μg/mL of this compound significantly reduced the survival of A375 cancer cells compared to the control, while no significant difference was observed when its concentration was increased up to 240 μg/mL. However, in the case of treating the normal HU2 cells with 7.5 μg/mL Taxol + vincristine compound, no significant difference was found with respect to the control. As a result, 7.5 μg/mL was the effective concentration of Taxol + vincristine treatment for A375 cancer cells. At this concentration, the normal and cancer cells’ viabilities were estimated as 99 ± 0.2 and 28 ± 0.2%, respectively.
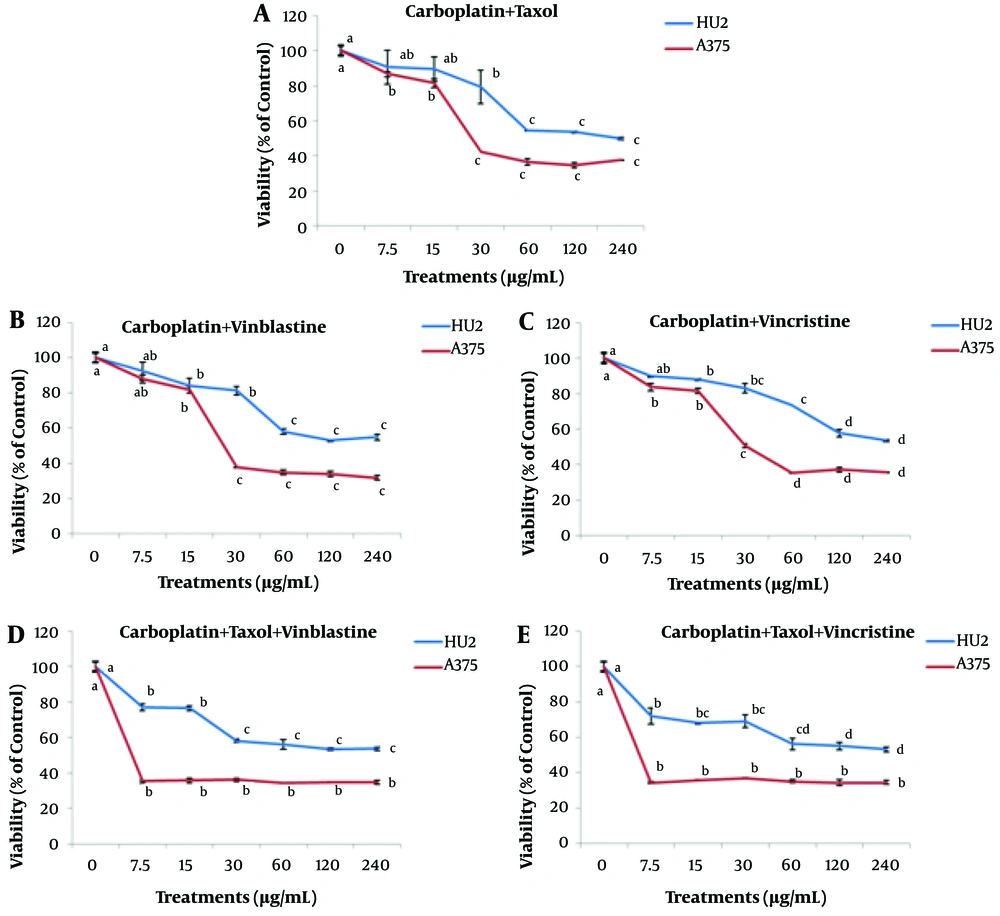
The effect of different concentrations of carboplatin combined with Taxol, vinblastine, and vincristine (0 - 240 μg/mL) is illustrated in Figure 3. The effective concentration for all the three compounds of carboplatin + Taxol, carboplatin + vinblastine, and carboplatin + vincristine in treating the cancer cells was found to be 30 μg/mL. The survivals of cancer and normal cells at this concentration were calculated as 79 ± 9.6 and 42 ± 0, 81 ± 2.5 and 38 ± 0.2, and 83 ± 2.4 and 50 ± 0.4% for carboplatin + Taxol, carboplatin + vinblastine, and carboplatin + vincristine treatments, respectively.
The effects of different concentrations of carboplatin + Taxol + vinblastine and carboplatin + Taxol + vincristine are illustrated in Figure 3. The results showed that a concentration of 7.5 μg/ml of carboplatin + Taxol + vinblastine significantly reduced the survival of cancer and normal cells (35 ± 0.8 and 77 ± 2% for A375 and HU2 cells, respectively). Also, a concentration of 7.5 μg/ml of carboplatin + Taxol + vincristine led to a significant reduction in the survival of cancer and normal cells (34 ± 0.6 and 72 ± 4.5% for A375 and HU2 cells, respectively). By increasing the concentration of these compounds from 7.5 to 240 μg/ml, no significant difference was observed among the treatments.
4.2. The Influence of the Effective Concentrations of Taxol, Vinblastine, Vincristine, and Carboplatin Alone and Their Combinations on the Viability of A375 Cells
Based on the results regarding the effects of various concentrations from 7.5 to 240 μg/mL of Taxol, vinblastine, vincristine, and carboplatin alone and their combinations on A375 and HU2 cells, the effective concentrations of these compounds were identified. The results of comparing the effective concentrations of these compounds are exhibited in Figure 4. Results indicated that the most significant effect was related to 120 μg/mL vincristine treatment with the survival of 24 ± 0.6%. Moreover, the highest effects were associated with Taxol + vincristine (7.5 μg/mL), carboplatin + Taxol + vincristine (7.5 μg/mL), Taxol (60 μg/mL), and carboplatin + Taxol + vinblastine (7.5 μg/mL) treatments with the estimated survivals of 28 ± 0, 34 ± 5.3, 35 ± 0.6, and 35 ± 0.6%, respectively.
The results of 50% inhibitory properties of the examined treatments are listed in Table 1. Accordingly, the best inhibitory effects were related to Taxol + vincristine, vinblastine, Taxol + vinblastine, carboplatin + Taxol + vincristine, and carboplatin + Taxol + vinblastine treatments at the concentrations of 0.04, 2.2, 3.4, 7.5, and 7.5 μg/mL, respectively. However, when calcysyn software received the related data of surviving fractions for calculating combination index (CI) values, the derivation of which was done through the Chou and Talalay method to assess for synergy, addition, or antagonism, there was no recording of synergistic reaction among the different combination of drugs (CI > 1).
| Cell Lines | Treatments | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxol | Vinblastine | Vincristine | Taxol + Vinblastine | Taxol + Vincristine | Carboplatin | Carboplatin + Taxol | Carboplatin + Vinblastine | Carboplatin + Vincristine | Carboplatin + Taxol + Vinblastine | Carboplatin + Taxol + Vincristine | |
| HU2 | 245.387 | 108.161 | 961.949 | 97.507 | 126.773 | 378.661 | 152.719 | 189.210 | 254.103 | 198.032 | 302.943 |
| A375 | 13.907 | 2.206 | 7.913 | 3.46 | 0.038 | 230.070 | 43.308 | 37.929 | 47.678 | <7.5 | <7.5 |
5. Discussion
In vitro toxicity results indicated that the studied carboplatin did not affect A375 human malignant melanoma cells when used alone. Dvorak et al. (2011) found that carboplatin does not cause significant cell damage in seven commercial human cancer cell lines (i.e., A2780, A2780cis, G361, MCF7, A549, HOS, and HeLa). These cell lines were derived from different types of cancer (vide supra) and in two primary cultures of human hepatocytes, because the cytotoxic potential of this substance is restricted by their limited solubility (16).
In contrast, the cytotoxicity of Taxol, vinblastine, vincristine, and their combinations was observed against A375 cells. However, the examined complexes were not cytotoxic against human fibroblast HU2 cells. The most significant effect on A375 cells was observed for vincristine and Taxol + vincristine treatments, respectively. In fact, Vinca alkaloids exert their cytoxicity via arresting mitosis and going into the interphase (19).
Nevertheless, laboratory and animal models reported other mechanisms of activity for vincristine, like cytolytic impacts on nondividing cells, stimulating cell death, affecting blood flow towards tumor tissues, and impacting the production of DNA, RNA, and intracellular proteins. Some 28-30 microfilaments have an important impact on the axoplasmic movement in neurons. They can be used to ‘tract’ axoplasmic movement of proteins or vesicles (20). Because of their function, Vinca alkaloids are schedule-dependent pharmacological interventions; hence, their function depends on exposure time to the drug (21). Taxol (Paclitaxel), as a new ingredient for chemotherapy can attach to the β-subunit of tubulin, which causes the development of stable, non-functional microtubule bundles that can affect mitosis during the G2/M phase of the cell cycle. Apart from its capability to stimulate different signal transduction mechanisms, such as JNK, p38MAPK, and ERK, Taxol was reported to enhance JNK/SAPK stimulation by Ras and ASK1 mechanisms (4).
The most effective concentrations of vincristine and Taxol were reported as 120 and 60 µg/mL, respectively. However, Taxol + vincristine combination was found to be effective at a concentration of 7.5 µg/mL. Vinca alkaloids are a group of anticancer pharmacological interventions developed for chemotherapy for several types of tumor tissues. Nevertheless, their negative consequences have resulted in the limitation of their applications (22).
A great number of studies have been conducted to evaluate the combination of different chemotherapeutic agents against several types of cancer (11, 22). The present achievements revealed that vinblastine can be employed to address various types of tumor tissues combined with other chemotherapy pharmacological interventions. Combined therapy, which is combination with other chemotherapy drugs, is a novel treatment intended to decline the consequences of Vinca alkaloids for increasing the antitumor properties. The main objective of combining pharmacological ingredients is to maximize their therapeutic effects (22).
Comparison of treatments regarding the viability of human fibroblast HU2 cells showed that carboplatin increased normal cell viability, while Taxol, vinblastine, and vincristine respectively reduced viability of normal cells significantly at the concentrations of 120, 15, and 240 µg/mL. As a result, carboplatin did not have any side effects, and vincristine had the least side effect on normal HU2 cells.
Only the MTT assay was used for examining the viability of the studied cells. To overcome this limitation, it is suggested to use the flow cytometry test to gain a deeper understanding of the mechanism of drugs’ action on the apoptosis or necrosis of studied cells.
Exploring novel drugs with increased anticancer activity and less toxicity for the normal cells is of high prominence (15). In this study, attention has been paid to the effect of four anticancer drugs on the viability of melanoma cells. Moreover, the possibility to use a combination of Taxol and carboplatin together and with Vinca alkaloids (vinblastine or vincristine) has been investigated. The results indicated that the most significant effects on A375 cells were associated with vincristine treatment, and no synergistic reaction was recorded among different combinations of drugs based on the calculated CI values by calcysyn software.