Studies show that the expression of
HOTAIR is significantly higher in tumors compared to normal tissues. On the other hand, many important processes in GC are influenced by this regulatory RNA. Identifying the mode of action of this RNA is effective in better understanding of pivotal molecular mechanisms in GC (
27).
The results of this study showed that the
HOTAIR gene may have a significant effect on the growth and proliferation of cancer cells. In fact, increased
HOTAIR expression can be considered as an important sign in the proliferation and spread of cancerous mass. Regulating the growth and proliferation of cancer cells requires influencing cell cycle and its inhibitors. Considering the mechanism of
HOTAIR action on cell cycles and the path of apoptosis can have a significant impact on better understanding the role of
HOTAIR on the proliferation and development of malignant tumors. The molecular pathway of
HATAIR in GC has also been investigated (
28).
Xu et al. stated that the decrease of
HOTAIR expression could be accompanied by the decrease of invasion potential and suppressing the EMT pathway. In this study,
HOTAIR was shown to impact a wide range of markers and transcription factors involved in the EMT pathway, and the decrease of
HOTAIR expression accounted for an important target in suppressing the invasion in GC (
23). Zhang et al. also showed that
HOTAIR could be regarded as an important factor in the EMT pathway and metastasis of GC (
29).
Previous studies demonstrated that lncRNAs are abnormally expressed in cancer and act as oncogenes or tumor suppressors, revealing an important role in cancer prognosis (
30).
HOTAIR is a possible therapeutic target in treating GC, according to current research findings.
HOTAIR has been shown in several investigations to induce EMT by inhibiting target genes (
31,
32). We discovered a potential action mechanism for
HOTAIR in the up-regulation of
ZEB1, one of the most critical markers of EMT.
It was previously revealed that miR-200 acts as a tumor suppressor factor by inhibiting EMT and tumor growth in GC by targeting
ZEB1 and ZEB2 (
28).
Takei et al. showed that a lncRNA (
HOTAIR) is expressed more vastly in 60As6 than HSC-60 cells. Expression of
HOTAIR promotes EMT in 60As6 cells. They discovered that
HOTAIR binds and targets miR-217, which binds to
ZEB1. In the orthotopic tumor mouse model, the knocking down
HOTAIR in 60As6 cells greatly declined invasion activity and peritoneal dispersion, and significantly extended longevity. The
HOTAIR-miR-217-ZEB1 axis, linked to EMT, appears to prevent peritoneal spread. They concluded that EMT is accelerated in scirrhous gastric tumors by at least two distinct mechanisms: (i)
HOTAIR overexpression (60As6 & 44As3 cell lines) and (ii) miR-200 family down-regulation (58As9 cell line). Both methods then work together to increase
ZEB1 expression and give cells mesenchymal characteristics (
33).
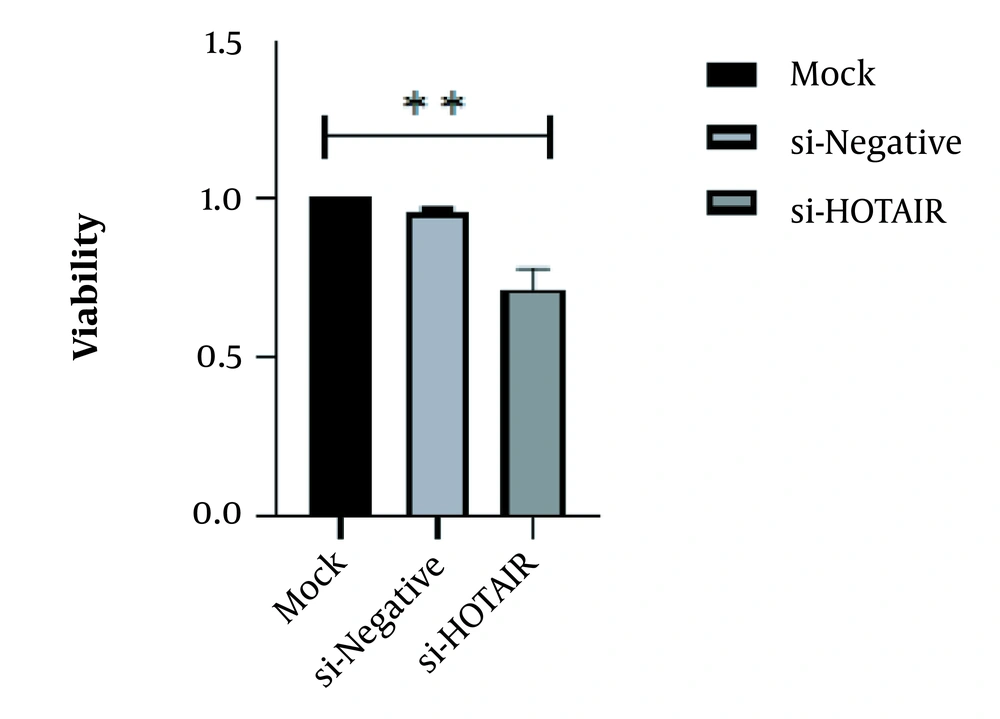
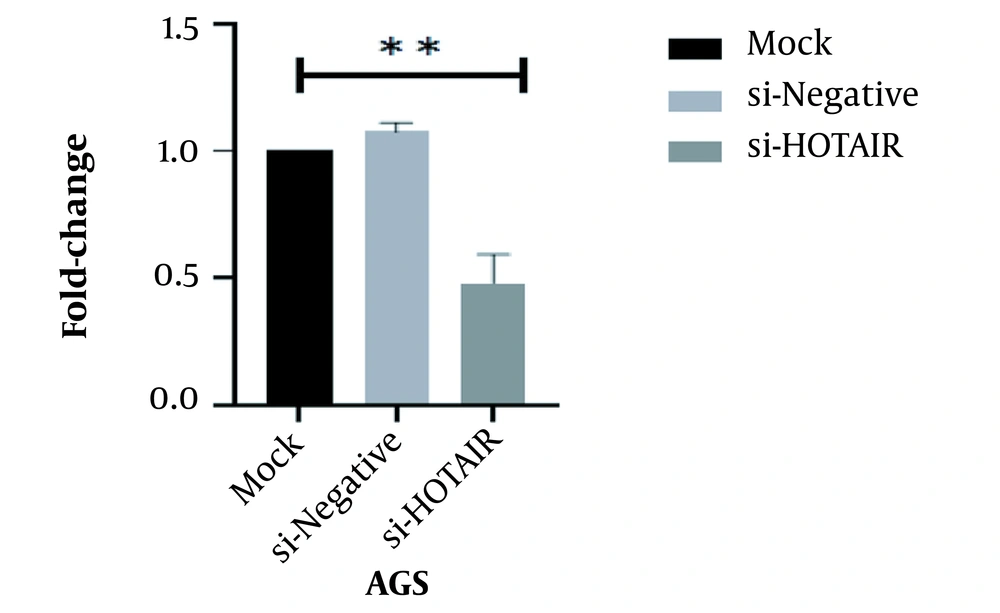
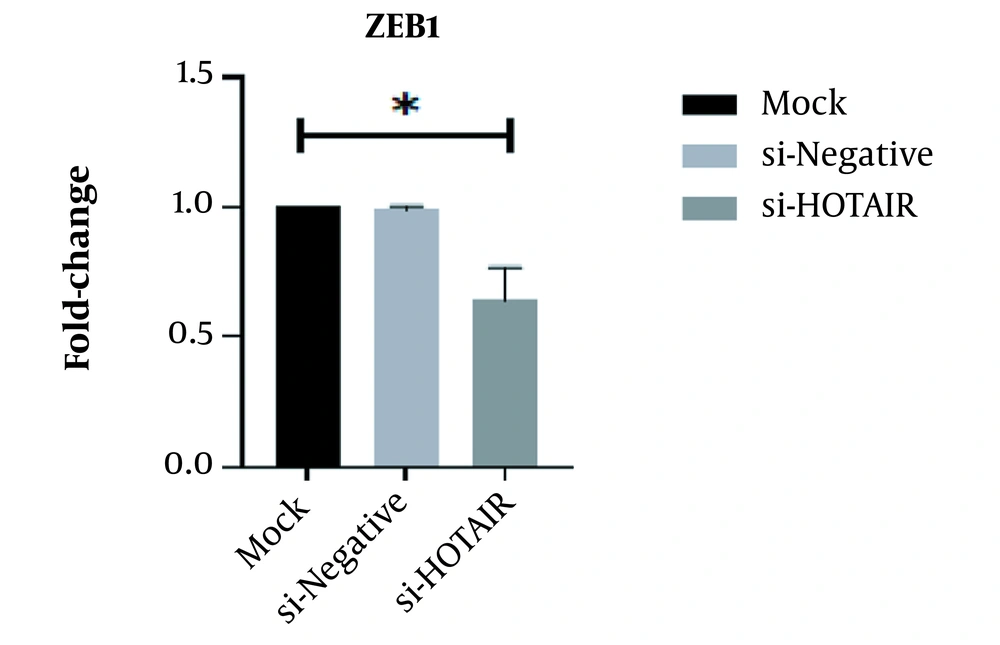
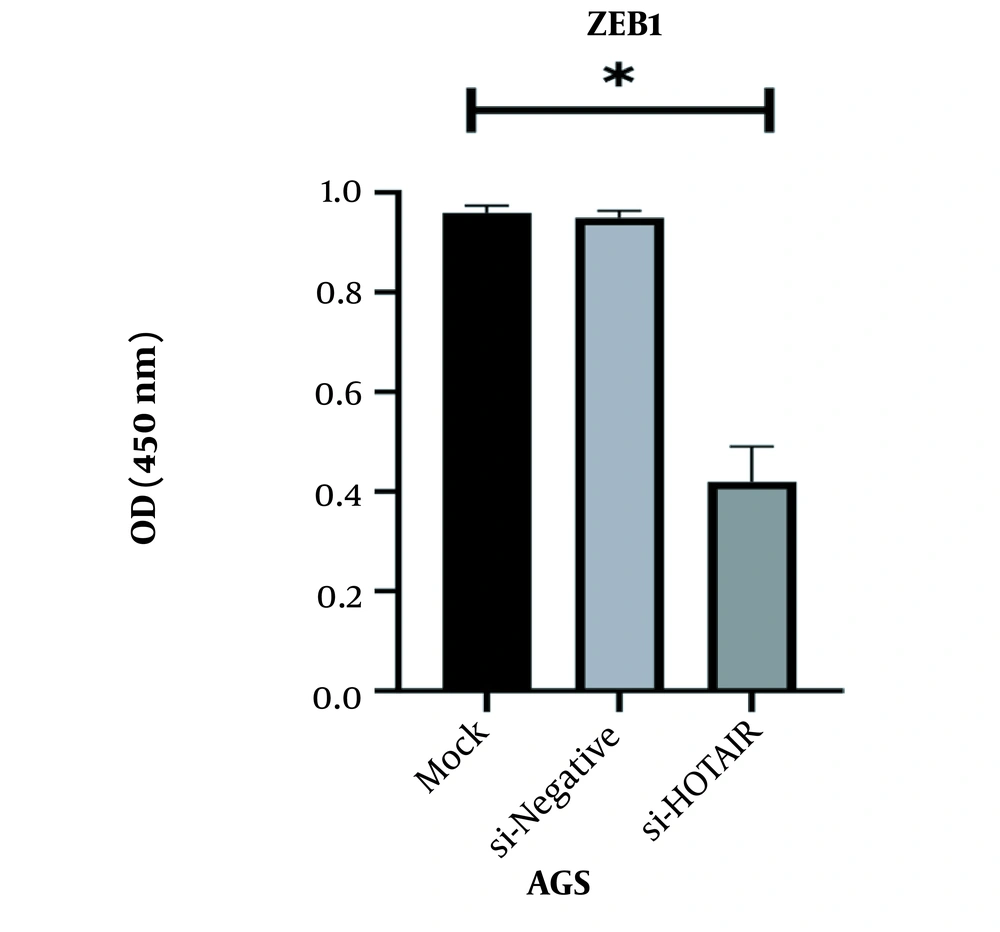
This study revealed that a drop in HOTAIR expression was linked to reducing cancer cell proliferation and growth. The decrease of HOTAIR expression and the ZEB1 gene transcription could happen alongside each other. According to our results, assessing the effect of suppressing HOTAIR expression in the AGS cell line at the level of protein by the ELISA technique showed that suppressing HOTAIR expression could decrease the expression of ZEB1 protein. Considering the key role of the ZEB1 gene as an important marker in progressing the EMT pathway and invasion, it can be concluded that HOTAIR can be a pivotal regulator in the occurrence of metastasis and invasion in GC tumors.
Our results suggested a new mechanism mediated by HOTAIR and ZEB1 that mediates the transitions between EMT and MET programs. As a result of their common molecular properties in the EMT process, ZEB1 and HOTAIR were studied. However, more investigations are needed to define the precise regulation mechanism of HOTAIR on the ZEB1. Further studies are needed to investigate this axis by overexpressing HOTAIR and analyzing interactions between HOTAIR and regulatory elements, evaluation of HOTAIR expression changes on other markers and transcription factors in the EMT pathway, as well as on cell cycle inhibitors and apoptosis for better understanding the effects on cell proliferation and inhibition of apoptosis. Finally, it is recommended to evaluate the effect of HOTAIR knockdown on the expression and translation levels of ZEB1 markers in other cancer cell lines. However, further studies on animal models are necessary.
5.1. Conclusions
The HOTAIR-ZEB1 axis appears to play a key role in human GC, making it a possible therapeutic target for the future. As far as the researchers investigated, this is the first research on the effect of HOTAIR-ZEB1 axis in GC. Nevertheless, more research on the specific mechanism is needed.