1. Background
Cutaneous Leishmaniasis (CL) is now accounted as a major problem in many developed and undeveloped countries around the world (1). The clinical manifestation may be presented as nodules, lymphadenopathy, plaque-like, and ulcerative skin lesions, which are commonly painless and expand gradually as granulomatous with a higher border (2). Duration of CL lesions depends upon the clinical symptoms and varies from 4 to 6 months, and in fact, the prolonged course of the Leihmania infection potentially reflects some molecular defects in immune responses. Previous studies demonstrated that Leishmania immune responses are strongly affected by activation of Th1 cells where the over expression of IL-10 as well as any failure occurred in IL-12/IFN-γ axis can obviously exacerbate the Leishmania infection (3-5). For example, development of monocytes and CD103+ subset of dendritic cells, which have indispensable roles in IL-12 production, are significantly affected by CXCR3 on T cells and BALB/c mice over expressing CXCR3 on their T cells or being deficient in CD103+ dendritic cells are more susceptible to L. major (6, 7).
Burning injuries also produce pro/anti-inflammatory cytokines, which determines the patient outcome. Cytokines are pleotropic and have multifunctional effects. In fact, these cytokines affect the wound in different ways. For example, appropriate levels of tumor necrosis factor alpha (TNF-α) accelerate wound healing procedures since this cytokine is beneficial in host immunity against pathogens. TNF-α modulate production of cytokines, which modulate cell proliferation survival and death (8). IL-1 has been assumed as a crucial regulator in expression of growth factor and matrix metalloproteinases (9). Likewise, other pro-inflammatory cytokines facilitate damage tissue. However, over expression of pro-inflammatory cytokines exacerbate skin destruction (10). Thus, a complicated network of these molecules potentially leads to tissue damage (8, 9). Various studies are aimed to determine pathogenic and protective cytokines and nevertheless, characterization of Leishmania lesions in terms of cytokine expression, cell proliferation, and healing process is not fully understood.
The main objective of this study was to investigate the cytokine expression profile in Leishmania lesions and comparing this profile with that of the burning lesion before and after treatment.
2. Methods
2.1. Animals and Parasites
For 8 weeks BALB/c mice were purchased from the Pasteur institute of Iran and housed in the BU-Ali Sina University animal house according to the standard of ethical agreement for keeping inbred animals. The L. major promastigots strain MHOM/76/IR was obtained from the Pasteur Institute of Iran. The parasites were cultured in Roswell park memorial institute (RPMI) 1640 medium supplemented with 10% fetal calf serum (FCS).
2.2. Leishmania and Burning Lesions
Two groups of 8-week BALB/c mice were anaesthetized in the base of the tail followed by burning the skins with a hot metal instrument at 200 for 5 seconds. The burning injuries were assessed as in grade II. The test group was treated daily with silver sulfadiazine cream and the control group was treated with PBS every day for 3 weeks.
For Leishmania lesions, 2 groups of 8-week BALB/c mice were intra-dermally injected with 1 × 107/100 µL Leishmania promastigotes in front of the tail. The first group was treated with 0.02 mg/kg/day Glucantime® (Glucantime, Paris, France) for 3 weeks. Each dose of the injecting drug was equally divided into 2 parts, which were injected intramuscularly (i.m) and intralesionally. The second group was also injected with PBS as control using the same procedure.
2.3. Size Assessment and Sampling in Leishmania and Burning Lesions
The size of the lesions in both Leishmania and burning experiments were assessed every 2 days using the following formula:
P = (D + d)/2
D = the large diameter, d = the small diameter, P = the average size of the lesion.
2.4. Sample Collection
Samples were collected from the lesions in both burning and Leishmania groups before starting the experiment, day 7, and day 14 of the experiment. In Leishmania experiments, the sampling procedure started when Leishmania lesions appeared. Before the sampling procedure, Lidocaine 2% was subcutaneously injected to the sampling region of the mice. Afterwards, we incised the skin using a No. 10 scalpel blade. The tissue samples were fixed in formaldehyde 10%. Samples were delivered to the pathology laboratory of Bu-Ali Sina University (Hamadan, Iran) for RNA extraction. For neutrophil extraction, heparinized blood samples taken from the heart of the mice were diluted with equal volumes of NaCl 0.85%. Meglumine compound (meglumine diatrizoate 66%, sodium diatrizoate 10%) was diluted in 3 times the volume of NaCl 0.85% and used for isolation of neutrophils. 5 mL of the diluted blood samples were slowly added to 4 mL of the diluted meglumine compound being careful not to mix the two phases of the blood and the meglumine compound. The samples were centrifuged for 15 minutes at 250 g and the supernatant was replaced with 0.5 mL of PBS. The pellet was homogenized by slow pipetting. Two 25 seconds of hypotonic lyses were applied with distilled water and NaCl 2.55%. The cells were then centrifuged for 5 minutes at 200 g, replacing the supernatant with NaCl 0.85%. In case of the mice spleen, the tissue was exposed to ultrasound pulses for 15 minutes at an average intensity of 1 W/cm2, a duration of 10 seconds, and a duty cycle of 50%.s. Afterwards, the homogenized tissue was mixed through a vortex and a pellet was formed through centrifugation (1,800 g 12 minutes). The obtained cells were used for RNA extraction.
2.5. Primers
All samples were examined for expression of CCL4, CCL3, TNF-α, IL-1α, IL-12P35, IL-12P40, CCL5, CCR5, IL-1β, and IFN-γ proinflammatory genes using specific primers. The primers were designed using software tools: Primer-BLAST, OLIGO, and DNASIS. Sequences of the primers used in this study are shown in Table 1.
| Accession | Sequence | Gene |
|---|---|---|
| NM_013652.2 | F: CAG CCC TGA TGC TTC TCA CT | CCL4/MIP-1β |
| R: GGG AGA CAC GCG TCC TAT AAC | ||
| NM_011337.2 | F: TCT GCG CTG ACT CCA AAG AG | CCL3/MIP-1α |
| R: GTG GCT ATC TGG CAG CAA AC | ||
| M38296.1 | F: TAT AAA GCG GCC GTC TGC AC | TNF-α |
| R: TCT TCT GCC AGT TCC ACG TC | ||
| CCDS16725 | F: CAG TTC TGC CAT TGA CCA TC | ll-1α |
| R: TCT CAC TGA AAC TCA GCC GT | ||
| M86672.1 | F: ATG ATG ACC CTG TGC CTT GG | IL-12P35 |
| R: CAC CCT GTT GAT GGT CAC GA | ||
| M86671.1 | F: CTG CTG CTC CAC AAG AAG GA | IL-12P40 |
| R: ACG CCA TTC CAC ATG TCA CT | ||
| NM_013653.3 | F: GTG CTC CAA TCT TGC AGT CG | CCL-5 |
| R: AGA GCA AGC AAT GAC AGG GA | ||
| NM_009917.5 | F: ATT CTC CAC ACC CTG TTT CG | CCR5 |
| R: GAA TTC CTG GAA GGT GGT CA | ||
| CCDS16726.1 | F: TTG ACG GAC CCC AAA AGA TG | IL-1β |
| R: AGA AGG TGC TCA TGT CCT CA | ||
| MN-008337 | F: GCT CTG AGA CAA TGA ACG CT | IFN- γ |
| R: AAA GAG ATA ATC TGG CTC TGC |
2.6. RNA Extraction
According to the manufacturer's instructions, the total mRNA was extracted from samples using the Denazist commercial kit. 100 mg of tissue or cell pellets was sonicated for 20 minutes. 1 mL of the lysing buffer included in the kit was added to the samples vortexing for 15 seconds and incubating at room temperature for 5 minutes. The samples were centrifuged at 12,000 g for 10 minutes at 4°C and the supernatant was transferred into a new tube. 200 μL of chloroform was added to the tubes and vortexed for 15 seconds followed by incubation at room temperature for 3 minutes. The samples were then spined at 12,000 g for 15 minutes at 4°C and the top phase was transferred into a new tube mixing with an equal volume of isopropanol. An equal volume of G2 buffer was added and mixed followed by incubation at room temperature for 10 minutes and centrifugation at 10,000 g for 10 minutes at 4°C. After discarding the supernatant and adding 1 mL of 70% ethanol, the tubes were quickly vortexed and centrifuged at 10,000 g for 5 minutes at 4°C. The supernatant was then discarded and 30 - 100 μL of nuclease-free water was added. The extracted supernatant was treated with DNase I and lack of genomic contamination was confirmed when there was not any band in PCR amplicon.
2.7. RT-PCR
According to the manufacturer’s instructions, RT-PCR was performed using the DENAzist kit. A mixture of 10 μg total RNA-primer, 1 μL oligo dT primer, 1 μL dNTP, and nuclease-free water up to 10 μL was prepared. The mixture was incubated at 65°C for 5 minutes and chilled on ice for 2 minutes. A mixture of cDNA Synthesis (10X Buffer M-MuLV 2 μL, M-MuLV reverse transcriptase 100 unit, nuclease-free water top up to 10 μL) was prepared and 10 μL of the cDNA synthesis mixture was added to each RNA-primer incubating at 42°C for 60 minutes followed by an extra incubation at 85°C for 5 minutes and then chilled on ice. DNA amplification was performed using a PC-708-02 thermal cycler (ASTEC; Fukuoka, Japan). The PCR was programmed as 40 cycles of denaturation at 95°C for 30 seconds, annealing at 4°C less than the melting temperature of the primers for 30 seconds and an extension step at 72°C for 20 seconds. An initial denaturation step of 95°C for 10 minutes before the cycles and a final extension step at 72°C for 10 minutes after the cycles was also applied. Distilled water was used as negative control.
3. Results
3.1. Healing of Burning Lesions
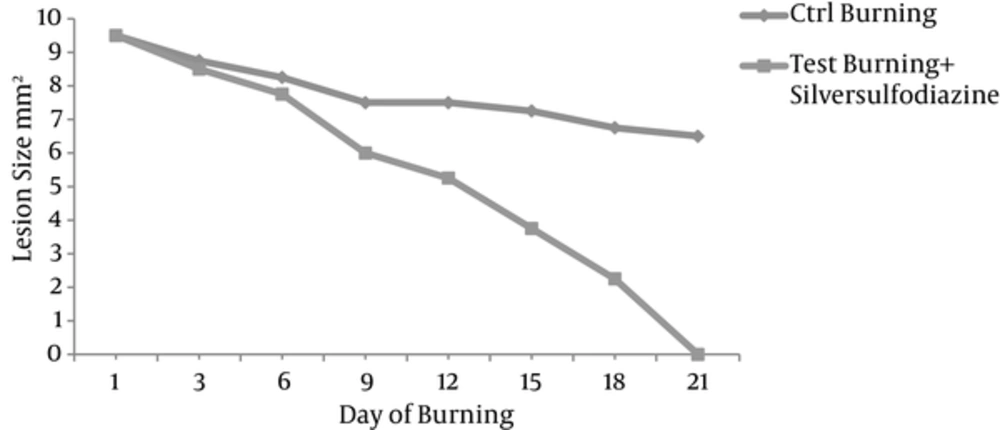
The healing process of the lesions caused by burning was monitored and the area of the lesions was measured and compared every 2 days (Figure 1).
The results showed that in the first week, the size of lesions in the group test receiving Silver sulfadiazine treatment was not significantly different compared to those of the group control, however, the interventional groups produced less fibrin deposition. Healing of the lesion ameliorated during the second week. This procedure was more progressive in the interventional group and at the end of the third week. More healing in the intervention group resulted in re-epithelialization.
3.2. Healing of Leishmania Lesions
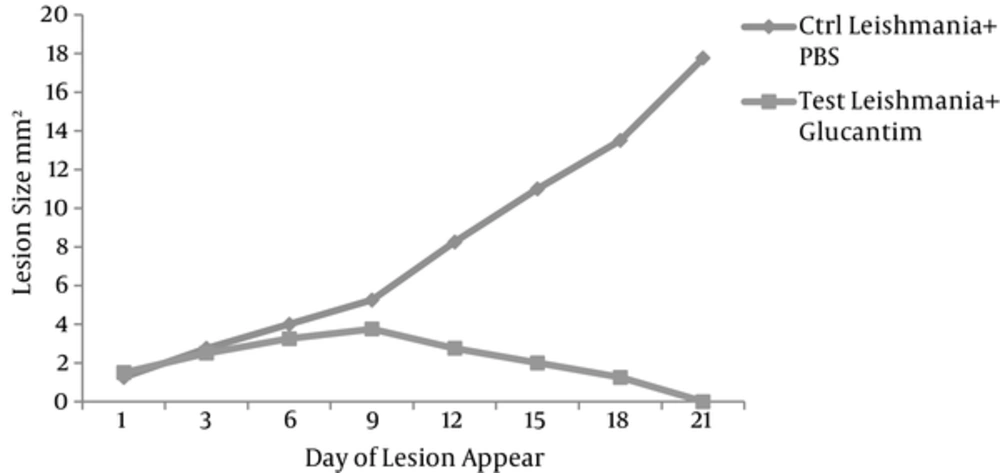
After a week of infection, Leishmania lesions were developed. The size of Leishmania lesions were measured and compared in every 3 days for at least 3 weeks until all lesions cured. The interventional group achieved clinical amelioration (within 3 weeks) as a result of Glucantime treatment and all lesions completely disappeared. In contrary, all of the mice in the control group died within 2 weeks (Figure 2).
3.3. Expression of Pro-Inflammatory Genes in Burning and Leishmania Lesions
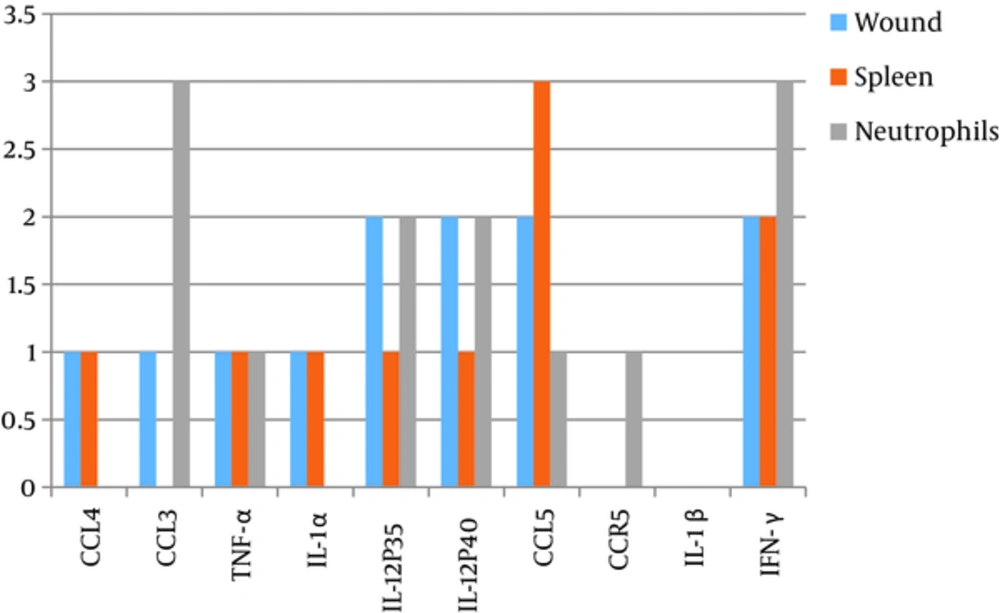
The expression of 10 pro-inflammatory genes including CCL4/MIP-1β, CCL3/MIP-1α, and TNF-α, IL-1α, IL-12p35, IL-12p40, CCL-5, CCR5, IL-1β, and IFN-γ was evaluated and semi-quantitatively appraised in all of the trials. The results showed that none of the pro-inflammatory genes were expressed in the healthy tissue. In Leishmania infected mice receiving only PBS, after Leishmania lesions became visible, all genes except IFN-γ were switched off and the expression of IFN-γ continued till the end of the second week. In Leishmania infected mice, which were treated with Glucantime, the figure of expression of the pro-inflamatory genes changed by the end of the first week and some of the genes including CCL4, CCL3, TNF-α, IL-12P35, IL-12P40, CCL5, and IFN- γ started to express. By end of the second week, the lesions cured and nearly disappeared, the expression the pro-inflammatory genes in the lesions even enhanced, and levels of IFN-γ, CCL-5, IL12p-40, IL12p-35, IL-1α, TNF-α, CCL-3 (MIP-1α), and CCL-4(MIP-1β) were detectable (Table 2, Figures 3 and 4).
| Sampling Time | Sampling Site | CCL4 | CCL3 | TNF-α | IL-1α | IL-12P35 | IL-12P40 | CCL5 | CCR5 | IL-1β | IFN- γ | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Healthy Tissue | All week | Wound | - | - | - | - | - | - | - | - | - | - |
| Spleen | - | - | - | - | - | - | - | - | - | - | ||
| Neutrophils | - | - | - | - | - | - | - | - | - | - | ||
| Group control (treated with PBS) | Week 1 | Wound | - | - | - | - | ++ | + | - | ++ | ++ | +++ |
| Spleen | - | - | + | - | - | - | ++ | ++ | + | ++ | ||
| Neutrophils | - | - | - | - | - | - | + | + | + | +++ | ||
| Week 2 | Wound | - | - | ++ | + | + | + | - | - | + | + | |
| Spleen | - | + | ++ | + | + | + | - | - | + | + | ||
| Neutrophils | - | - | ++ | ++ | + | + | - | - | + | + | ||
| Group test (treated with Silver sulfadiazine) | week 1 | wound | - | - | ++ | - | ++ | ++ | - | - | + | - |
| Spleen | + | - | - | + | + | + | - | + | - | + | ||
| Neutrophils | - | - | - | - | - | - | - | - | - | - | ||
| Week 2 | Wound | - | - | - | - | + | + | - | - | - | ++ | |
| Spleen | - | - | - | - | + | + | - | - | - | ++ | ||
| Neutrophils | - | - | - | - | - | - | - | - | - | - |
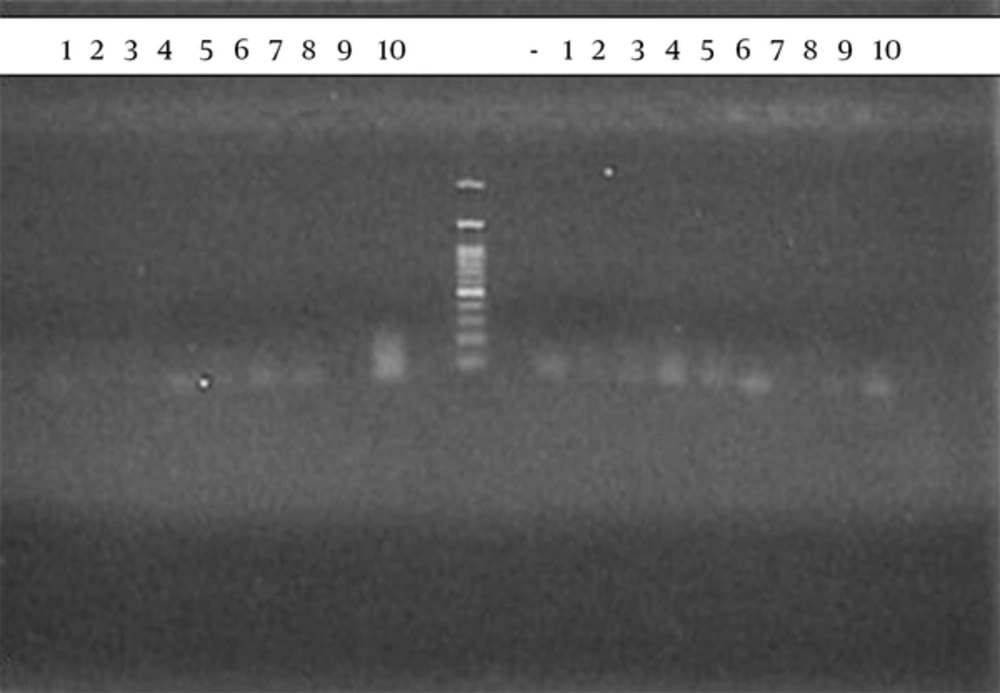
aThe thickness of bands shown in the gel electrophoresis was marked from 1 to 3 plus.
Expression of 1- CCL4, 2- CCL3, 3- TNF-α, 4- Il-1α, 5- IL-12p35, 6- IL-12p40, 7- CCL-5, 8- CCR5, 9- IL-1β, and 10-IFN- γ proinflammatory genes were evaluated in burning or Leishmania lesions. Bands 1-10 proinflammatory genes in burning lesions of group control (treated with PBS) in week 1, 11 negative control, 12 standard DNA and 13 - 23 proinflammatory genes in lesions caused by Leishmania in group test (treated with Glucantime).
On the other hand, the expressions of the same genes in burning lesions were also evaluated. In the control group, which received no treatment, the expression of IFN-γ, IL-1β, CCR5, IL-12p35, and IL-12p40 genes was detectable during the first week of the burning. In the second week, TNF-α, IL-1α, IL-12P35, IL-12P40, IL-1β, and IFN-γ genes were significantly expressed (Table 3, Figures 3 and 5).
| Sampling Time | Sampling Site | CCL4 | CCL3 | TNF-α | IL-1α | IL-12P35 | IL-12P40 | CCL5 | CCR5 | IL-1β | IFN- γ | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Healthy Tissue | All week | Wound | - | - | - | - | - | - | - | - | - | - |
| Spleen | - | - | - | - | - | - | - | - | - | - | ||
| Neutrophils | - | - | - | - | - | - | - | - | - | - | ||
| Group control (treated with PBS) | Week 1 | Wound | - | - | - | - | - | - | - | - | - | +++ |
| Spleen | - | - | - | - | - | - | - | - | - | +++ | ||
| Neutrophils | - | + | - | - | ++ | ++ | ++ | ++ | + | +++ | ||
| Week 2 | Wound | - | - | - | - | - | - | - | - | - | - | |
| Spleen | - | - | - | - | - | - | - | - | - | - | ||
| Neutrophils | - | + | ++ | + | + | + | + | - | + | + | ||
| Group test (treated with glucantime) | Week 1 | Wound | + | + | + | - | ++ | ++ | +++ | - | - | ++ |
| Spleen | + | - | ++ | ++ | ++ | ++ | ++ | + | - | - | ||
| Neutrophils | - | + | + | + | ++ | ++ | - | - | - | ++ | ||
| Week 2 | Wound | + | + | + | + | ++ | ++ | ++ | - | - | ++ | |
| Spleen | + | + | + | + | +++ | - | - | ++ | ||||
| Neutrophils | - | +++ | + | - | ++ | ++ | + | + | - | +++ |
aThe thickness of bands shown in the gel electrophoresis was marked from 1 to 3 plus.
On the contrary, when in the other group, the lesions treated with silver sulfadiazine, the expression of the pro-inflammatory genes altered in the first week and TNF-α, IL-12P35, IL-12P40, and IL-1β were expressed. However, only IL-12 (p35 and p40) and IFN-γ genes were detectable in the lesions by the end of the second week (Figures 3 and 5).
4. Discussion
This study was designed for better understanding of Leishmania and burning lesions based on their pro-inflammatory gene expression profiles and to make a better understanding on different characteristics of Leishmania lesions in comparison with burning injuries.
Leishmania infection in humans and wild rodents is mostly harnessed yielding localized cutaneous lesions and long immunity. There are many reasons that claim BALB/c mice as laboratory models in clinical presentations and immunological responses resemble human Leishmaniasis (11). These mice fail to constrain the Leishmaniasis developing progressive lesions, splenomegaly, hepatomegaly, and systematic symptoms releasing levels of cytokines, which suppress the immune system and cause the death (12, 13). Similarly, severe burn injuries are also induced production of immunological cytokines, which potentially yield shock and animal death (14). Therefore, cytokine profiles play the most prominent role in the outcome of all lesions including those caused by Leishmania infections and burn injuries (15). The results clearly showed that the expression pattern for proinflammatory genes in Glucantime treated and untreated lesions caused by Leishmania is different than those caused by burning injuries. In Leishmania lesions, the expression of pro-inflammatory genes in untreated lesions is highly down regulated and except for IFN-γ all other examined genes were switched off, where after treatment and during the healing period, most of the genes were strongly expressed in the lesions. In burning injuries, most of the proinflammatory genes are highly expressed in the lesions before treatment and also started down regulation when the healing process progressed in the lesions.
TNF-α is shown to play an important role in inducing the lesion in Leishmaniasis. Oliveira et al. in a cohort study, demonstrated that Leishmania braziliensis derived lesions were strongly correlated with amounts of TNF-α levels released in supernatants of peripheral blood mononuclear cell culture. They suggested that TNF-α inhibitors associated with standard therapy could be exploited for treatment of severe Leishmaniasis (16). Furthermore, The negative effect of TNF-α expression in Leishmania lesions was approved when anti-TNF drugs appeared as a promising candidate for Leishmania treatment (17). In burning injuries, transcription of NF-κB and AP-1 genes are augmented leading to up regulation of pro-inflammatary cytokines such as IL-6, TNF-α, and keratinocyte-derived chemokine (KC). TNF-α, in turn induced systemic inflammatory reactions potentially resulted in multiple organ failure (18).
In the present study, TNF-α was only expressed in Leishmania lesions treated with Anti-Leishmanial drugs. In untreated lesions, no sign of TNF-α expression was observed. These findings, which verified association between the lacks of expression of TNF-α and the severity of Leishmaniasis, was not in-line with those of the previous reports. In burning lesions, TNF-α was switched off in control groups (untreated wounds) until the end stages of the lesions where in silver sulfadiazine treated groups at first days of the lesions, the gene was expressed but in the healed lesions the expressing of the gene was ceased. These results also confirmed the association of TNF-α in healing of burning injuries and did not agree with those of the previous reports.
CCR5 is a protein, which is detectable on some of the hematopoietic and non-hematopoietic cells. The natural ligand of CCR5 comprising CCL3 (MIP-1α) and CCL4 (MIP-1β) can negatively affect the level of IFN-γ, which is expressed in Leishmania lesions. Indeed, CD4+, CD25+ natural T regulatory cells expressing CCR5 establish Leishmania survival due to modulation of IFN-γ production. Notably, CCR5-/- mice were resistant to L. major and a high expression of CCR5 resulted in lesion deterioration, arose in further stages (19, 20). Our results were in contrary with those of previous ones where CCR5 was never expressed in Leishmania lesions either in controls or in the groups treated with anti-Leishmanial drugs in Leishmania susceptible BALB/c mice. However, in both groups, CCL3 (MIP-1α) and CCL4 (MIP-1β) expressed only in Leishmania lesions after treatment.
It is known that T Lymphocytes, NK cells, inflammatory monocyte and NKT cells are perceived as the main source of IFN-γ production (21). Recent studies demonstrated that neutrophils could be assumed as a crucial source of IFN-γ production through toll like recepetor-2 and Dectine contributing towards immunity against intracellular pathogens (21-23). According to the results, IFN-γ was not expressed in healthy tissues but Leishmania lesions were expressing IFN-γ either before or after treatment with anti-Leishmanial drugs. The importance of IFN-γ production would be brought to notice when Toll Like receptor 11−/− infected with mice Toxoplasma gondii achieved significant levels of IFN-γ production. Notably, neutrophils are capable of producing IFN-γ through the TLR-independent pathway, which differ from the conventional pathway (myeloid-differentiation factor 88) exploited by T and NK cells (22). Additionally, different cytokines such as IL-17a, IL-12, and IL-15 or their combination yield IFN-γ transcription (23, 24). In this context, in vitro studies demonstrated that IL-12 in combination with IL-15 could spur neutrophils on IFN-γ production (23). Constitution of IL-12 cytokine needs production of both IL-12p35 and IL-12p40. IL12p35 is shown to be produced by a wide array of cells including lymphocytes whilst IL-12p40 is just produced by activated phagocytes (25). We detected IL-12p40 in neutrophils (data not shown) as activated phagocytes, which coincides with other recent studies showing that neutrophils produce IL-12 cytokine when they are activated through TLR-2 (26). Another study demonstrated that IFN-γ knock-out mice produced high levels of IL-12 when they were intraperitoneally infected by Toxoplasma gondii (27). The results indicated a high expression of both IL-12p35 and IL-12p40 in Leishmania lesions only after treatment with the anti-Leishmanial drug while in burning lesions the expression of the IL-12 genes was at a medium level in both the test and control groups with no significant difference. It seems the production of IL-12 is implemented through the IFN-γ independent pathway and strongly suppressed by the parasite but when the lesion is treated with anti-Leishmanial drugs the expression of IL-12 was switched on. On the other hand, the expression of IFN-γ alone was not able to eliminate the parasite and the expression of IL-12 contributed in removing the parasite through the activation of IL-12 and IFN-γ axis as indicated by other researchers (25, 28-30).
It has been shown by other researchers that the expression of IL-1β is conversely associated with the healing of Leishmania infections. It seems IL-1β not only does not efficiently alleviate Leishmania lesions, but this cytokine assists the infected neutrophils, which are presumed as Trojan horses, to disseminate Leishmania infections (31, 32).
The detrimental effects of IL-1β cytokine, however, are controversial when some researches believed that this cytokine promotes leishmanicidal impacts through augmentation of nitric oxide production (33). Furthermore, production of IL-1 skews the T cell response toward TH2.
The results showed that IL-1β is only expressed in burning lesions not in the Leishmania lesion even after treatment confirming some previous studies, which pointed out the Leishmania infection affected levels of gene expression constituting failure in IL-1β production (34). Interestingly, some cytokines such as IL-28A (IFN-λ2), IL-28B (IFN-λ3) in mice and humans, and IL-29 (IFN-λ3) in humans could harness the production of IL-1β. Hence, this cytokine could not be detected in Leishmania lesions, which were either in the shrinkage or healing periods (35, 36).
Similarly, no IL-1α expression was observed in the lesions of Leishmania infected mice in both the test and control groups. However, in Glucantime treated mice, IL-1α was expressed in the end stages of the disease. Conversely, in burning lesions, IL-1α was only expressed in the group treated with PBS but not in the mice treated with silver sulfadiazine.
4.1. Conclusions
The expression figure of pro-inflammatory genes in Leishmania lesions is totally different than those of burning lesions. It seems Leishmania intimately down-regulates the expression of proinflammatory genes in the lesion and therefore, in the lesions that received no treatment, most of the proinflammatory genes are switched off or down regulated. In the lesions that received anti-Leishmanial treatment, the pro-inflammatory genes started to be expressed or up regulated. Instead, in the burning injuries, the proinflammatory genes were mostly expressed during the healing process and until the lesions were cured the expression of the genes were downregulated or stopped.