1. Background
Cancer is a type of malignancy that causes the change and growth of body cells out of control. Most cancer cells eventually form a growth or mass called a tumor (1). The origin of breast cancer is breast tissue (milk-producing lobules and their ducts). In addition, fat tissue, lymphatic tissue, and connective tissue are also found in breast tissue (2). Breast lumps are usually seen using a mammogram, most of which are benign and do not pose any risk to the individual (3). Breast cancer, the second most common cancer in the world, has the highest prevalence among women. According to the statistics provided by the American Cancer Society, about 13% (one out of eight) of American women have breast cancer (4, 5). In Iran, 76% of common women’s cancers are of the breast type. The number of people with this cancer is 41,000 in Iran (6). Breast cancer is divided into three different groups according to different molecular subtypes, risk factors, response to treatment, and gene expression profiling (7).
Luminal A: The general type of breast cancer that makes up about 40% of all breast cancers is luminal A type, which is ER+ and/or PR+ and HER2-. These types of tumors grow little and are less aggressive. Luminal B: These tumors are about 10 to 20% of breast cancers, and like the luminal A tumor, most of the luminal B tumors are ER+ and/or PR+, but they are HER2+ and have a high proliferation rate. Basal-like: This category also includes about 10 to 20% of breast cancers. Most basal-like cancers are called triple-negative because they are both ER-, PR-, and HER2-. These tumors, along with BRCA1 mutation, are often seen in American and African women and usually have a weaker prognosis and a shorter lifespan than other types of cancer. HER2 enriched: In this category, the level of HER2 expression is very high and includes about 10% of breast cancers. The growth of these tumors is similar to basal-like and aggressive ones (8, 9).
Photodynamic therapy (PDT) is used for clinical use to destroy the tumor to remove the tumor in the early stages and relieve symptoms in patients with late-stage tumors. The treatment includes several stages: (1) a light sensor, (2) light produced by a laser source in the visible range of electromagnetic waves, and (3) molecular oxygen to produce single oxygen or other toxic oxygen species, such as superoxide anion radicals and hydroxyl radicals (10, 11). The PDT destroys the tumor by directly affecting the tumor cells and its stroma. Stroma includes extracellular matrix, vascular system, fibroblast, endothelial cells, and immune system cells. Studies show the effectiveness of PDT in destroying tumor cells and their stroma (12).
Curcumin is one of the main components of turmeric and is known by the chemical name 17-Bis(4-hydroxy-3-methoxyphenyl)-16-heptadiene-35-dione (13). Different pharmacological and biological mechanisms of curcumin include antioxidant effect, inhibition of inflammatory factors, induction of cell death, anti-cancer, and activation or inhibition of intracellular pathways effective in causing disease (14). Various clinical studies have proven the role of curcumin in the prevention and treatment of cancer. Curcumin application studies on cancer, on the one hand, show that the use of curcumin prevents cancer, and people who take curcumin have a lower prevalence of cancer. On the other hand, these studies show that the simultaneous use of curcumin with chemotherapy drugs increases the process of cancer treatment and also increases the complications caused by chemotherapy or radiotherapy (15).
2. Objectives
In this study, given that curcumin has an aggressive effect on breast cancer cells and the laser with a wavelength of 810 nm in the presence of curcumin can destroy more cancer cells, we aimed to determine the percentage of living cells under the influence of curcumin in breast cancer cells and the percentage of living cells under the influence of low power laser in breast cancer cells.
3. Methods
This research was carried out in the year 2021 - 2022 in Afra Biology Laboratory (Tehran). Cell culture: The MDA-MB-231 cell line (an epithelial, human breast cancer cell line) was obtained from the Pasteur Institute cell bank and cultured in a culture medium containing 90% DMEM and 10% fetal bovine serum. The cells were kept in standard conditions of 37°C and 5% of carbon dioxide, and about 80% humidity. The cell culture medium was changed every two days.
Study of curcumin: According to the study of previous studies, logarithmic concentrations of 1, 10, 100, and 500 µg/mL of curcumin (Sigma) were selected. The metabolic activity of cells incubated in the presence of different concentrations of curcumin was evaluated using the MTT test (MELFORD, manufactured in Etgolestan). For this purpose, 5 µL of MTT stock was added to each well. After keeping for four hours in a 37-degree incubator and removing the culture medium, the reaction was stopped by adding 100 microliters of DMSO or 0.04 normal HCl solution in isopropanol. After the dissolution of the purple formazan chromophore in the cells, the absorbance was measured by an ELISA plate reader (BioTeK, USA) at a wavelength of 570 nm. The results were expressed as a percentage of the control values (16). The following formula was used to calculate the viability of cells as a percentage.
Vital cell percentage = (drug - treated photo absorbed cells - absorbed optical blank)/(absorbed optical control - absorbed optical blank) = ×100
Determination of IC-50: IC-50, that is, the concentration of the drug in the presence of which 50% of the growth of the cells was inhibited compared to the control. To determine the IC-50 and find the appropriate dose for the next tests, the cells were treated with different concentrations of the drug for 48 hours in 96-well culture dishes.
Measuring the release of lactate dehydrogenase enzyme into the culture medium: The rate of cell death was evaluated by measuring the release of cytosolic enzyme lactate dehydrogenase (LDH) from the cells. The amount of LDH enzyme activity in supernatant solution and cell lysed sediment (in control) was measured by the spectrophotometric method (LDH diagnostic kit of Pars Azmoun). The percentage of LDH released from the cell to the culture medium was calculated by the following formula:
Percentage of LDH released = (LDH in the culture medium)/ (LDH present in culture medium + LDH present in cell lysate solution) ×100
Low-power laser combined with curcumin: In this step, the cell suspension arranged from Breast cancer cells (MDA) containing 1 × 104 cells was refined in 96-well plates. Twenty-four hours after development, the cells were uncovered to low-power diode laser radiation with a wavelength of 810 nm for 90 seconds. In the next step, the irradiated cells were placed in the incubator with specified amounts of curcumin for 24 hours at 37°C and a 5% CO2 atmosphere. Then, after adding the specified amount of MTT and DMSO according to the protocol, the light absorption rate of formazan was read by an ELISA reader (Stat Fax-2100 model, Spain) in the absorbance spectrum of 570, and the survival rate was calculated according to the relevant formula.
4. Results
The proliferation of breast cancer cells (MDA) was well done, and the morphology of the cells was similar before and after freezing.
4.1. Evaluation of Toxicity Induced
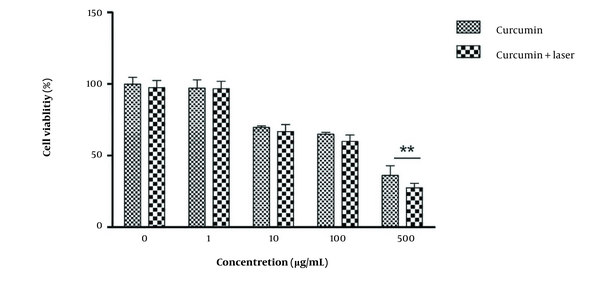
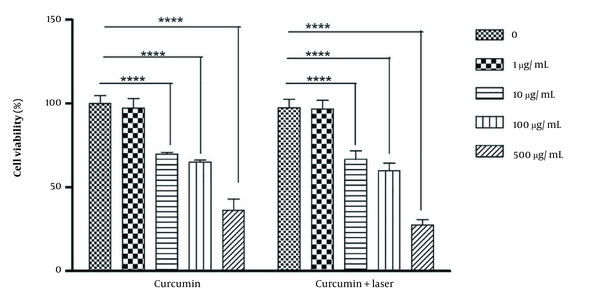
The range of IC-50 was first determined to evaluate the toxicity induced by different concentrations of curcumin alone, the laser alone, and the combination of different concentrations of curcumin with laser. The test was repeated 6 times, and the average of live cells in different groups was obtained based on the control group. Accordingly, the survival of the control group was considered 100%, and the rest of the group was reduced to the same ratio compared to the control group based on the mentioned formula (Figure 1). The survival percentage of MDA cells in the combination of 500 μg/mL concentration with laser was significantly lower than the rest of the groups (Figure 2).
4.2. Determination of IC-50 Concentration
First, the range of IC-50 was determined. The results of IC-50 were calculated from the percentage of live cells based on the MTT test with PRISM software. The results showed that in the groups that received concentrations of 1, 10, 100, and 500 µg/mL of curcumin alone, the IC-50 gel was 195.8 µg/mL, but in the combination of curcumin with laser, the concentration decreased (108.8 µg/mL).
4.3. Determination of Cell Proliferation Rate
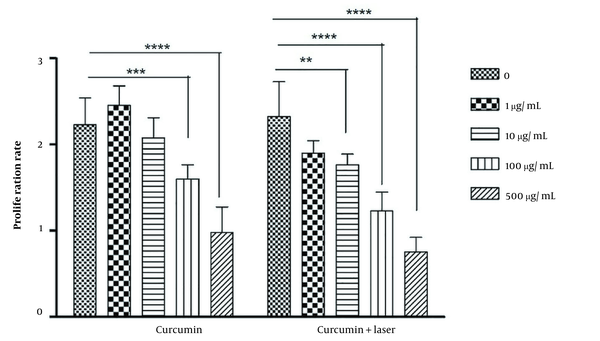
To determine the rate of cell proliferation in curcumin alone, the laser alone, and the combination of different concentrations of curcumin with laser, the rate of multiplication of MDA cells after 48 hours was evaluated 6 times. Based on the obtained results, the rate of cell proliferation of MDA cells in the combination of 500 μg/mL concentration with a laser decreased compared to the other groups (Figure 3).
4.4. Determining the Amount of Peroxidation of Membrane Lipids
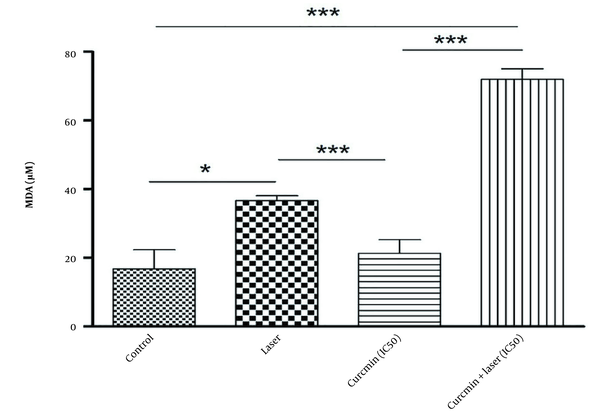
To determine the amount of peroxidation of membrane lipids in curcumin alone, laser alone, and the combination of different concentrations of curcumin with lysis, a line slope was first determined based on the obtained standards and then based on the line slope equation and the light absorption rate of the unknown Malone D sample. Aldehyde peroxidation of membrane lipids of the target sample was determined. Based on the results of Figures 4 and 5, the average concentration of malondialdehyde obtained in micrograms in the curcumin group with laser was higher than in other groups.
4.5. Determining the Release of Lactate Dehydrogenase Enzyme Into the Culture Medium
The percentage of the release of lactate dehydrogenase enzyme into the culture medium in different groups compared to the control group was determined based on the light absorbance of the LDH at a wavelength of 340 nm. Furthermore, the release rate of the LDH in each group was calculated according to the light absorption of each group divided by the light absorption of the control. According to the results, the highest release rate of the LDH was seen in the combination of curcumin combined with the laser group (Figure 6).
5. Discussion
Cancer is one of the main causes of death in the world; as the World Health Organization (WHO) announced in 2020, about 20 million new cases of cancer were registered in the world. Tumors usually arise as a result of an imbalance between cell proliferation and cell death. Most tumor cells that escape from cell death and uncontrolled cell proliferation may eventually cause cancer (17, 18). For more than a hundred years, curcumin has been widely used in traditional medicine as a compound with low side effects and high safety (13). In 1985, Kuttan et al. showed that curcumin has anti-cancer and antimutagenic properties. After that, studies revealed the anti-proliferative, anti-invasive, and apoptotic properties of curcumin. However, curcumin’s current potential for anti-cancer activity has primarily been investigated using in vitro experiments and animal models (19).
Today, studies have confirmed that curcumin combined with a photodynamic laser has effective antibacterial, anti-inflammatory, antioxidant, and anti-cancer activity (20). In the present study, the effects of curcumin and low-power laser therapy on breast cancer cells (MDA) were examined. The results showed that the survival percentage and the cell proliferation rate of MDA cells decreased with increasing the concentration of curcumin and the combination with a laser, so the lowest survival percentage and proliferation rate of MDA cells were observed in the concentrate combination of 500 μg/mL with laser. Furthermore, the study of Xie and colleagues showed that laser therapy could significantly increase the anti-cancer activity of curcumin (21). Bayer et al. also showed that curcumin, at low concentrations (from 0.01 to 1 μg/mL) alone, had almost no effect on the proliferation of human oral squamous cell carcinoma cells. But after treatment with the same amount of curcumin and subsequently, with 5 minutes of visible light irradiation, cell proliferation was significantly inhibited, and the rate of cell proliferation decreased to 30%. Furthermore, curcumin (0.2 μg/mL) combined with visible light significantly increased DNA fragmentation, which was approximately three folds greater than the control group (22).
On the other hand, the study of the effects of different wavelengths of light radiation on the absorption of light energy and the targeted stimulation of curcumin showed that the combination of red light (630 nm) and blue light (405 nm) could effectively increase the autophagy of A375 cancer cells, thereby preventing cell proliferation (23). In total, the results of the present study show that the combination of curcumin with a photodynamic laser can reduce the survival effect and cell proliferation rate of curcumin on MDA cells. Curcumin is one of the main active ingredients in turmeric extract, which has antibacterial, anti-inflammatory, antioxidant, and anti-cancer activities. Although many pieces of evidence show that curcumin has good anti-cancer potential in vitro, curcumin has rapid metabolism in the body and poor oral availability, which limits its application.
The combination of curcumin with photodynamic laser partially overcomes this limitation and can effectively prevent the proliferation of different types of cancer cells, induce apoptosis of cancer cells, and improve the bioavailability of curcumin to a great extent. However, most of the current research on curcumin combined with photodynamic laser focuses on in vitro experiments and animal models, and further clinical research is needed.