1. Background
Osteoarthritis (OA) of the knee joint is characterized pathologically by loss of articular cartilage and clinically by pain and disability. Its prevalence will increase rapidly over the next 20 years (1). The occurrence of OA is related to age, obesity, trauma, genetics and various pathophysiological events within the joints (2). It is also associated with major cellular and molecular alterations in articular tissue. Inflammatory factors such as cytokines, nitric oxide, prostaglandin E2 (inflammatory pain mediators), neuropeptides and proteolytic enzymes are produced by the inflamed synovium (3). Chondrial changes include cartilage fragmentation (fibrillation), presence of tear, loss of type II collagen, chondrocyte apoptosis and loss of proteoglycans. As one grows older, the ability of chondrocytes to maintain and restore of articular cartilage decreases (4). Articular cartilage is composed of chondrocytes and extracellular matrix (ECM) and the ECM is composed mainly of collagen fibers type II (5). The balance of collagen and Proteoglycan (PG) is reject disrupted in OA, and proteoglycans are disrupted in OA and trauma. Early stages of OA, are characterized by a decrease in proteoglycans and collagen, change in cartilage properties, activation of some degenerative enzymes and reduction of aggrecan size (6). The breakdown of type II collagen appears in later stages of OA and follows the degradation of PG (7). It is clear that both mechanical and biological factors are involved in degeneration of cartilage, decreased synthesis of ECM and formation of chondrocyte clusters (8). In other words, the number of cells remains unchanged yet distribution of their arrangement occurs, and highly cellular clone formation related to the disability of proliferated cells to migrate in damaged areas. Chondrocytes attach to ECM proteins, via integrin receptors and this adhesion is implicated in OA and leads to cartilage fibrillation (2). Growth factors such as IGF-1 or TGF-β contribute to chondrocyte adhesion to fibronectin (9). It has been established by animal models of OA, that the intra-articular injection of Monosodium Iodoacetate (MIA) acts as an inhibitor of glycolysis and promotes loss of articular cartilage similar to OA and produces necrosis with degeneration of chondrocyte in tibial plateaus and femoral condyles (10). Despite the high prevalence of OA, an effective treatment is currently lacking. Difficulties of studying OA in humans have led to highly dynamic animal models (11). There are different pharmacologic and non-pharmacologic methods for treatment of OA; however the application of cell–based therapies has been used for a number of decades (12). Bone marrow is the main source for isolation of Mesenchymal Stem Cells (MSCs), and these cells have the potential to differentiate to a variety of cells of different tissues (13). The application of MSCs for OA repair requires different techniques, although the local injection of MSCs may be the simplest procedure (14).
There are two administration methods of stem cells: direct or local and intravascular administration. Numerous researchers have discussed the use of MSCs to repair a variety of tissues (15). The structural damages of articular cartilage have been demonstrated in a variety of ways such as clefts. Proteoglycan loss and collagen breakdown have been observed by microscopic slides (16). It is clear that MSCs are suited for cartilage repair. These cells facilitate regeneration, by secreting nutrient and bioactive factors (17).
2. Objectives
In the current study, MSCs and their secretions were used to repair articular cartilage defects by assessment of ECM (PG) depletion in a rat model.
3. Materials and Methods
3.1. Animals
Eighteen healthy, 18-week-old, male Sprague-Dawley rats were used as sample rejectcases in this study. Animal experiments were reviewed and approved by the institutional animal care and use committee (IACUC) of our institution.
3.2. Experimental Protocol
The rats were selected randomly and divided to four groups, each consisting of six animals except the control group. Induction was done through intra-articular injection of MIA in right knee joints, while the other six left knee joints were considered as the control group. The MSCs were harvested in a separate procedure and a single dose of 2.5 × 105 cells was injected for the samples of the MSCs group, while the same amount of medium plus cell secretions was injected for the M + CS group. All animals were sacrificed two weeks after therapy.
3.3. Intra-Articular Injection of Monosodium Iodoacetate (MIA)
Osteoarthritis was induced in rats briefly anaesthetized by an intraperitoneal injection of xylazine 10 mg/kg and ketamine 20 mg/kg, by intra-articular injection of MIA (Sigma, UK) into the right knee joint. The knee joint was shaved and flexed at a 90 degree angle. Five microliters of 1 mg/50 µL (MIA) in sterile saline (0.9%) was injected through the infrapatellar ligament into the joint space of the right knee with an insulin syringe. This concentration of MIA has been previously demonstrated to induce OA in mice. Control rats received an intra-articular injection of 50 µL of sterile saline in the left knee joint [R5].
3.4. Cell Isolation and Culture
The bone marrow aspirate was diluted with Dulbecco’s Modified Eagle’s Medium (DMEM) (1:3) and 10% fetal serum bovine (FBS), and was then centrifuged at 1200 g for five minutes. The supernatant was discarded and the cells pellet was washed two times with PBS. The cells pellet was suspended with DMEM (1 mm) and was plated as primary culture in a culture flask at 0.5 mm. The flask was then incubated at 37°C in a humidified atmosphere containing 5% CO2. On day four of culture, the non-adherent cells were removed along with the change of medium every three days. Undifferentiated MSCs were transplanted upon reaching 70% - 80% confluency (12).
3.5. Histological Analysis
At two weeks post treatment, the animals were sacrificed. The knee joints of the three experimental (right knees) and six contra-lateral joints were dissected and fixed in 10% formalin at 4°C and decalcified in formic acid (5%) for three days. The specimens then underwent automated tissue processing for 24 hours. Sections were imbedded in paraffin and cut into 5 µm slices. Each section was stained with toluidine-blue and safranin-O.
3.6. Safranin-O
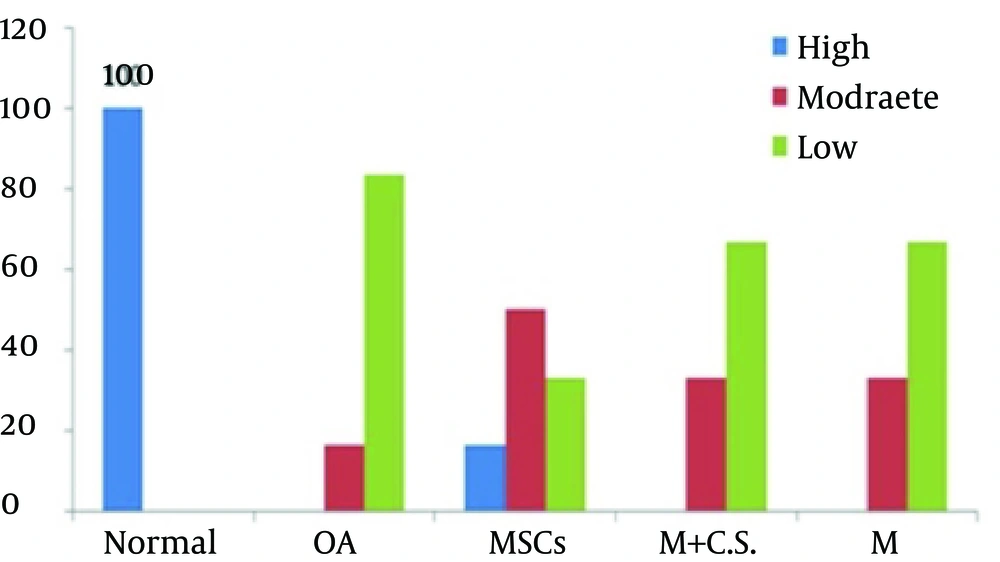
The distal femurs were removed and fixed in 10% formaldehyde, decalcified in 10% formic acid, and embedded in paraffin. Serial sections of 6 µm were cut sagittally. Histological staining was performed with safranin-O histo-chemical stain to analyze proteoglycan distribution. Slides were first placed in Mayer’s hematoxylin for two minutes and rinsed in water. These slides were then placed in fast green fetal calf fluid (FCF) Yellowish for 10 minutes and rinsed in acetic acid (1%) until the glass was free of stain. Next, the slides were placed in safranin-O for 30 minutes, and rinsed in water and 95% EtOH until the glass was free of stain. After air drying, the slides were cover-slipped. The histological preparations were examined by a light microscope and PG depletion of cartilage matrix was graded as high, moderate and low.
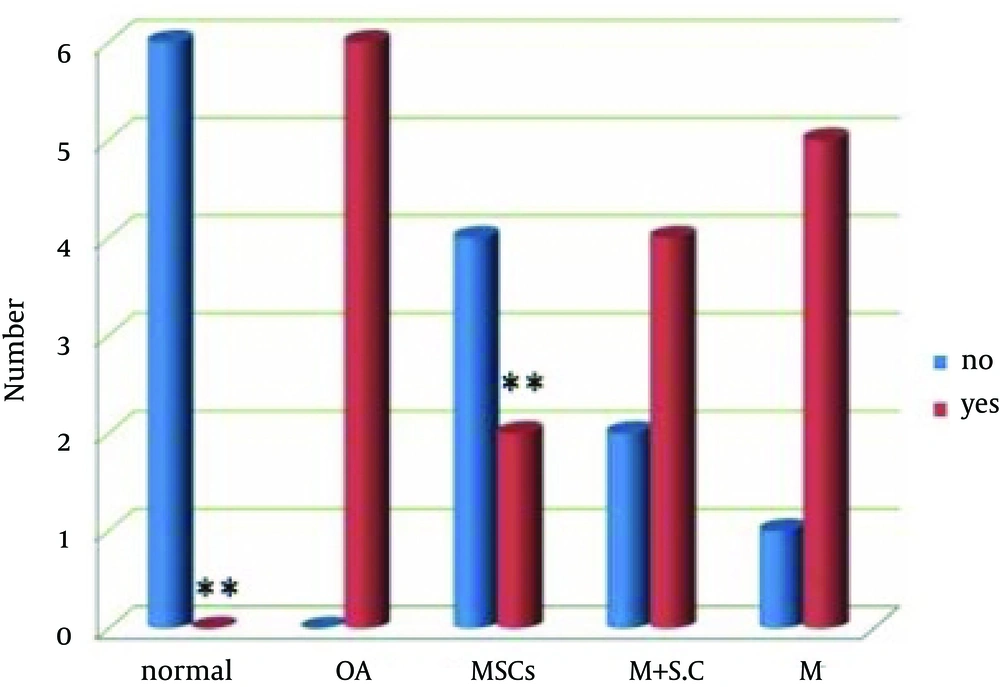
The Mann-Whitney test for nonparametric analysis was used to compare the number of high, moderate and low density PGs. Values of P < 0.05 were considered statistically significant.
4. Results
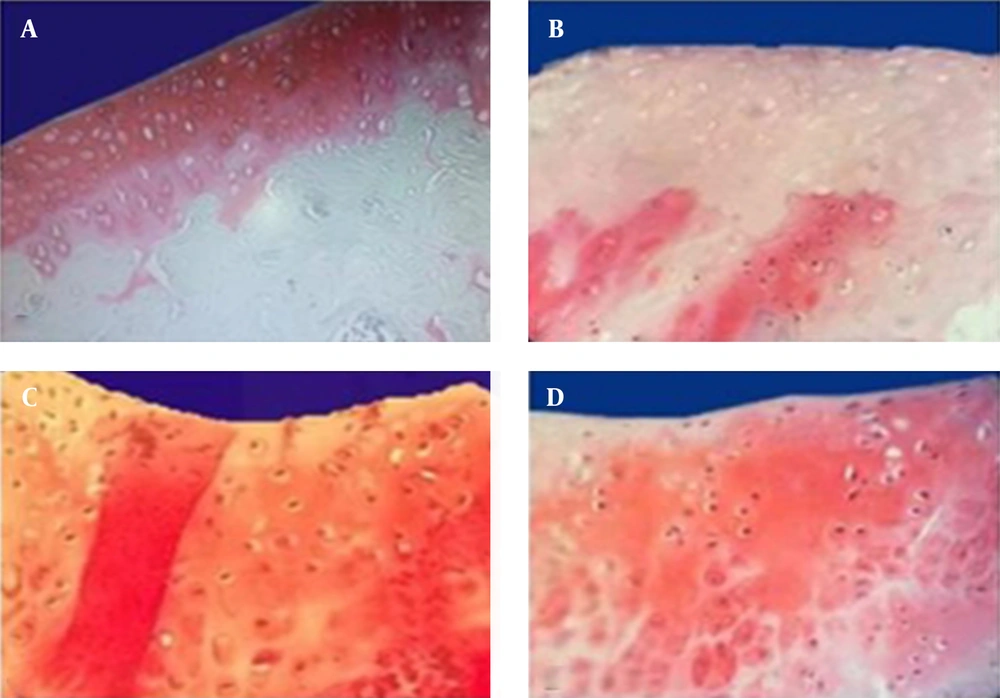
Outcomes were evaluated microscopically with histological staining. Uniform safranin-O staining was seen through all zones in the controls, with dense underlying bony plate, yet it was lighter near the articular surface where the PG content was lower. In the OA group, a progressive loss of PG staining was found at the cartilage surface and progressed to deeper layers. Direct injection of MSCs to articular space caused a subsequent marked synthesis of PG in the MSCs group, and after cell transfer these samples showed uniformly stronger staining in the proliferative zone than in the calcified region (Figure 1). Similar results with low intensity were obtained when the cells were removed and medium plus secretions of cells was used as a second treatment method. Analysis with Mann-Whitney test using the SPSS software (Figure 2) indicated considerable increase of PG synthesis in the MSCs group compared with the control cases (P < 0.05). Presence of fibrillation was identified in all groups at the femoral condyles of the cartilage surface with toluidine blue staining. In fibrillated cartilage (OA), the cluster shape of chondrocytes was formed and some areas of the cartilage presented a cellular formation, which was filled with ECM. Cartilage fibrillation was observed in OA groups, and it deceased significantly in the MSCs group (P < 0.05) after injection of a single dose of 2.5 × 10 cells (Figures 3 and 4).
Intact articular cartilage with intense red (S-O) staining indicates the concentration of PG. A, A uniform red staining was seen in all regions of normal articular cartilage; B, Increases loss of red staining was seen in OA group; C, Significant increase of PG in MSCs and D, slight increase of PG in (M + C.S.).
A, Intact articular cartilage with chondrocytes in all zones of the cartilage; B, Cluster shape of chondrocytes in fibrillated cartilage on OA; C, marked increase of chondrocytes and did not show fibrillation after two weeks from treatment by MSCs; D, moderate increase of chondrocytes in the M + C.S. group.
5. Discussion
Mesenchymal stem cells with inherent chondrogenic potential and anti-inflammatory function are the best candidates for repairing cartilage damage in OA. These cells facilitate regeneration by secreting various factors (12). Osteoarthritis makes changes in ECM and these changes are transmitted to the environment of chondrocytes. This condition, thus promotes the progression of disease. In this disease, many inflammatory factors, such as cytokines and proteases change the quality of ECM (18). The changes in ECM component can prevent the differentiation of MSCs to chondrocytes (19). It has been revealed that PG content is decreased in OA, which is in concordance with our results. Because of the incidence of proliferation of chondrocytes and the formation of cluster shaped cells, this cellular arrangement can change the quantity and composition of ECM (20). Our findings revealed a significant loss of proteoglycan synthesis. Both treated groups (MSCs, M + C.S.) demonstrated potential regeneration of cartilage compared to the control group with a higher degree in the MSCs group. Previous in vitro studies have shown that the induction of bioactive factors, such as TGF-β and IGF-1, promote the synthesis of PG. The present study also reported that fibrillated cartilage was seen significantly in OA cartilage samples from femoral ends and injection of MSCs could significantly decrease the percentage of fibrillated cartilage. The association between both highly cellular clusters and acellular areas is known as a common finding in OA (21). It was observed that some chondrocytes do not have the ability to migrate to damaged areas. Decrease in cell adhesion to fibronectin could be related to presence of apoptotic or necrotic chondrocytes in affected cartilage (22). Mesenchymal stem cells have the potential of differentiating into chondrocytes and can migrate to injured areas (23). In conclusion, MSCs can be used for healing and regeneration of cartilage. One of the most interesting findings of this study was the progression in repair of OA caused by factors that were secreted from MSCs. It has been emphasized that, possibly because of growth factors needed for cartilage healing, the absence of cells cannot stop the regeneration process.