1. Background
In broiler chickens, optimal growth performance requires a crude protein (CP) diet and a proper combination of amino acids (AAs). The priciest feed ingredients for chicken diets are sources of dietary protein. Consequently, the absence of AAs causes a delay in the growth of the intestines and the proliferation of muscle cells linked to the main source of protein, making it one of the major obstacles to the efficient production of broilers (1). According to Norouzian et al. (2), there is a direct correlation between the broiler chickens’ final growth rate and the early growth rate of their skeletal muscles, particularly their intestines. However, there is a trend towards reducing CP content in broiler diets (3). Additionally, it is anticipated that a low-CP diet will reduce excess water consumption in birds, which frequently results in damp bedding and anomalies in the legs by excreting more nitrogen (4). Reducing CP in diets, however, should only be done after taking into account the minimal requirements for vital AAs. It should be highlighted that non-essential AAs are also significant, and inadequate synthesis and low levels of these AAs can impact physiological function and performance (5).
Branched-chain amino acids (BCAAs) — leucine, isoleucine, and valine — are included in the category of essential AAs. All three BCAAs are structurally similar to branched-chain fatty acids and have a hydrophobic side chain. Leucine is 2-amino-4-methyl-pentanoic acid, isoleucine is 2-amino-3-methyl-pentanoic acid, and valine is 2-amino-3-methyl-butanoic acid (6). Nutrients can control muscle growth directly or indirectly through effects on regulatory factors. The nervous and endocrine systems, as coordinators of body metabolism, play an important role in regulating animal growth. Growth hormones, insulin-like growth factor 1 (IGF-1), thyroid hormones (T3 and T4), and insulin play important and different roles in animal growth. The IGF hormones are important regulators in growth stimulation, AA elongation stimulation, glucose metabolism, DNA synthesis, protein synthesis, and the proliferation and differentiation of different cell types (7). The primary role of BCAA is to participate in the building of proteins in the body, and IGF-1 is one of the stimulants of protein synthesis (8). The IGF-1 is a potential key regulator of chick growth and body composition (9). There are specific pathways mediated by the mTOR protein that favor protein synthesis and cell proliferation. These pathways are stimulated by mitogens such as insulin and BCAA (10).
2. Objectives
The present experiment was conducted to determine whether the effects of low-CP diets manifest themselves and can be attenuated or exacerbated by manipulation of BCAA levels. Therefore, the purpose of this research is to evaluate the effect of different levels of BCAA in low-CP diets on performance, intestinal morphology, liver enzymes, blood parameters, and IGF-1 gene expression in broilers.
3. Methods
3.1. Animals, Diets, and Experimental Design
To conduct this research, 480 one-day-old male and female broiler chickens of the Ross 308 strain were used, with an average one-day weight of 42.75 ± 0.47 g. This experiment was carried out as a 3 × 2 factorial in the form of a completely randomized design with 6 treatments. Four repetitions were considered for each treatment, and 20 chickens (10 males and 10 females) were placed randomly in each repetition. Experimental treatments were based on a corn-soybean meal and included: (1) Diet with standard protein and BCAA (SP: BCAA-S), (2) diet with standard CP and BCAA 10% higher than the standard (SP: BCAA-10), (3) diet with standard CP and BCAA 20% higher than the standard (SP: BCAA-20), (4) diet with CP 10% less than standard and BCAA standard (LP: BCAA-S), (5) diet with CP 10% less than standard and BCAA 10% higher than standard (LP: BCAA-10), (6) diet with CP 10% lower than the standard and BCAA 20% higher than the standard (LP: BCAA-20).
The diets were adjusted based on the 2019 Ross 308 nutritional requirements catalog and formulated using the UFFDA ration software. The energy-to-CP ratio in all diets was considered according to the standard of the Ross 308 strain. The adjusted diets used in the starter (1 - 10 days), grower (11 - 24 days), and finisher (25 - 42 days) periods are presented in Table 1 (11). During the entire rearing period, feed and water were freely provided to the birds. Breeding management programs, including light, ventilation, temperature, density, and substrate, were the same for all treatments and were carried out according to the recommended standard.
| Ingredients (%) | Starter (1 - 10 d) | Grower (11 - 24 d) | Finisher (25 - 42 d) | |||
|---|---|---|---|---|---|---|
| Standard | Low-CP | Standard | Low-CP | Standard | Low-CP | |
| Maize | 60.62 | 63.85 | 55.15 | 60.02 | 60.33 | 65.37 |
| Soybean meal (44% CP) | 27.58 | 26.81 | 36.73 | 32.68 | 31.26 | 27.04 |
| Corn gluten | 6.89 | 4.33 | 0.0 | 0.0 | 0.0 | 0.0 |
| Soybean oil | 0.0 | 0.0 | 4.01 | 3.17 | 4.54 | 3.67 |
| Calcium carbonate | 1.70 | 1.70 | 1.53 | 1.54 | 1.40 | 1.42 |
| Dicalcium phosphate | 1.51 | 1.52 | 1.29 | 1.32 | 1.16 | 1.19 |
| Salt | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| Vitamin-mineral mix a | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 |
| Sodium bicarbonate | 0.07 | 0.07 | 0.08 | 0.07 | 0.07 | 0.07 |
| Lysine-HCL | 0.46 | 0.38 | 0.20 | 0.12 | 0.21 | 0.14 |
| DL-methionine | 0.17 | 0.23 | 0.18 | 0.22 | 0.17 | 0.20 |
| L-threonine | 0.07 | 0.11 | 0.0 | 0.02 | 0.0 | 0.02 |
| L-valine | 0.0 | 0.0 | 0.002 | 0.0 | 0.0 | 0.0 |
| L-isoleucine | 0.1 | 0.1 | 0.0 | 0.01 | 0.01 | 0.03 |
| L-arginine | 0.10 | 0.16 | 0.0 | 0.0 | 0.0 | 0.0 |
| Total | 100 | 100 | 100 | 100 | 100 | 100 |
| Nutrient levels | ||||||
| Metabolizable energy (Kcal/kg) | 2950 | 2950 | 3050 | 3050 | 3150 | 3150 |
| CP | 22.23 | 20.01 | 21.15 | 19.75 | 19.20 | 17.75 |
| Calcium | 0.93 | 0.93 | 0.85 | 0.85 | 0.86 | 0.86 |
| Available phosphorus | 0.46 | 0.46 | 0.43 | 0.43 | 0.39 | 0.39 |
| Dig. lysine | 1.34 | 1.24 | 1.29 | 1.13 | 1.17 | 1.01 |
| Dig. methionine | 0.50 | 0.58 | 0.51 | 0.53 | 0.47 | 0.49 |
| Dig. met + cyst | 0.92 | 0.92 | 0.86 | 0.86 | 0.79 | 0.79 |
| Dig. threonine | 0.83 | 0.83 | 0.79 | 0.76 | 0.71 | 0.68 |
| Dig. arginine | 1.32 | 1.32 | 1.36 | 1.25 | 1.21 | 1.10 |
| Dig. valine | 1.01 | 0.93 | 0.98 | 0.92 | 0.89 | 0.82 |
| Dig. leucine | 2.23 | 1.18 | 1.80 | 1.71 | 1.66 | 1.57 |
| Dig. isoleucine | 0.90 | 0.83 | 0.88 | 0.83 | 0.81 | 0.75 |
Abbreviation: CP, crude protein.
a Supplied per kilogram of diet: Vitamin A, 9000 U; vitamin D3, 2000 U; vitamin E, 18 U; vitamin B12, 0.15 mg; riboflavin, 6.6 mg; calcium pantothenate, 10 mg; niacin, 30 mg; choline, 500 mg; biotin, 0.1 mg; thiamine, 1.8 mg; pyridoxin, 3 mg; folic acid, 1 mg; vitamin K3., 2 mg; antioxidant (ethoxyquin), 100 mg; zinc, 50 mg; manganese oxide, 100 mg; copper, 10 mg; Fe, 50 mg; I, 1 mg; Se, 0.2 mg.
3.2. Intestinal Morphology
At 42 days of age, 2 birds from each replicate (1 male and 1 female) were selected for small intestinal morphology evaluation. The segments of the duodenum, jejunum, and ileum were gathered and examined in accordance with Liu et al. (12). After being cleaned with 0.1 M phosphate-buffered saline, 1 cm sections of the middle part of the duodenum, jejunum, and ileum tissues were fixed in 10% formaldehyde phosphate buffer. The fixed sections were then processed, dehydrated, and embedded in paraffin wax. They were sectioned at 5 μm and stained with hematoxylin-eosin. Using a digital camera microscope (BA400 Digital) and the Motic Advanced 3.2 digital image analysis system, histological sections were analyzed for villus height (VH), villus width (VW), crypt depth (CD), and muscular thickness. Ten well-oriented villi were selected, and ten muscular thicknesses were measured from each segment. The VH to CD ratio (VH/CD) was then computed.
3.3. Insulin-Like Growth Factor 1 Gene Expression in Liver Tissue
After euthanizing the birds at the age of 42 days, liver tissue samples were taken 4 times from each treatment to measure the level of IGF-1 gene expression. For this purpose, the real-time polymerase chain reaction (PCR) method was used, considering the GAPDH gene as a reference gene. RNA extraction was performed using Synaclon kits according to the manufacturer's instructions. The quality of extracted RNA was checked using 1% agarose gel electrophoresis. The quantity of extracted RNA was also evaluated with a nanodrop device. The extracted RNAs were immediately used to carry out the reverse transcription step and produce cDNA using the Synaclone kit. The samples were kept at -20°C for further processing.
To amplify fragments of the IGF-1 and GAPDH genes by the real-time PCR method, reverse primers were designed for each gene (Table 2). Polymerase chain reaction was performed to amplify parts of the IGF-1 and GAPDH genes from cDNA samples obtained from chicken liver tissue mRNAs using primers designed by the ABI7300 device (Applied Biosystems, Warrington, UK) and the Cybergreen method. The program required for this reaction included initial denaturation (95°C for 10 min) and 40 three-step cycles: Denaturation (95°C for 15 s), primer annealing (60°C for 30 s), and sequencing (72°C for 30 s) using an ABI7300 (Applied Biosystems, Warrington, UK). The melting curve was drawn by measuring fluorescence changes at different times using a real-time PCR machine.
| Genes | Gene Bank ID | Primer Sequence, Sense/Antisense | Product Size (bp) |
| IGF-1 | NM_001004384.3 | 5’TACCTTGGCCTGTGTTTGCT3’ | 214 |
| 5’GCCTCCTCAGGTCACAACTC3’ | |||
| GAPDH | NM_204305.2 | 5’AGGACCAGGTTGTCTCCTGT3’ | 228 |
| 5’CTCCAACAAAGGGTCCTGCT3’ |
Abbreviation: IGF-1, insulin-like growth factor 1.
After carrying out the amplification reaction using the relative quantitative real-time PCR method, the raw data in the form of the cycle threshold (CT) of each of the two target and reference genes were extracted from the device for each repetition and entered into Excel software. The CT averages of each treatment were calculated for both genes, then the difference in CT values of target genes (IGF-1) and reference (GAPDH) was obtained as ΔCT. In the next step, the difference of ΔCT of each treatment with the control treatment was calculated as ΔΔCT. Then the relative changes of the target mRNA level were calculated using the 2-ΔΔCT relationship (13).
3.4. Statistical Analysis
Data analysis was performed using SAS 9.4 statistical software (SAS Institute Inc., Cary, NC) with the mixed procedure in the form of a completely randomized design. The significance of the differences between the mean data was evaluated using Duncan's multiple range test at the 5% significance level. The statistical model used in the experiment was as follows:
Yijk = value of each observation, μ = mean of observations, Ai = protein levels, Bj = AA effect, ABij = interaction between protein and AA, and eijk = residual effect (experimental error).
4. Results
4.1. Intestinal Morphology
Table 3 presents the effect of different levels of CP and BCAA in the diet on intestinal morphology. The results indicated that the VH/CD ratio in the duodenum, jejunum, and ileum is influenced by CP and BCAA levels. Using 20% BCAAs in the diet increased the VH/CD ratio in the duodenum, jejunum, and ileum compared to the standard level of BCAA (P < 0.05). Conversely, a 10% decrease in CP resulted in a lower VH/CD ratio in the duodenum, jejunum, and ileum compared to the standard level (P < 0.05). There was no significant effect of BCAA inclusion level and CP level on VH, VW, and CD (P > 0.05).
| Treatments and Items | Duodenum (μm) | Jejunum (μm) | Ileum (μm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VH | VW | CD | VH/CD | VH | VW | CD | VH/CD | VH | VW | CD | VH/CD | |
| CP | ||||||||||||
| Standard | 2060.42 | 240.97 | 401.37 | 5.13 A | 1180.75 | 193.70 | 220.74 | 5.34 A | 1074.22 | 187.92 | 207.18 | 5.18 A |
| Low | 2034.94 | 239.56 | 408.16 | 4.99 B | 1109.21 | 191.52 | 219.73 | 5.04 B | 1054.45 | 184.99 | 207.83 | 5.07 B |
| SEM | 50.91 | 6.96 | 9.64 | 0.03 | 41.29 | 5.58 | 6.40 | 0.05 | 30.3 | 09.27 | 5.82 | 0.01 |
| P-value | 0.72 | 0.84 | 0.62 | 0.04 | 0.23 | 0.78 | 0.91 | 0.02 | 0.64 | 0.18 | 0.92 | 0.03 |
| BCAA | ||||||||||||
| 0% | 2035.50 | 242.53 | 409.17 | 4.98 B | 1134.56 | 195.62 | 224.43 | 5.05 B | 1046.94 | 188.68 | 208.52 | 5.02 B |
| 10% | 2027.48 | 237.36 | 400.53 | 5.06 A, B | 1124.52 | 190.14 | 215.57 | 5.21 A | 1043.20 | 184.52 | 203.09 | 5.14 A, B |
| 20% | 2080.01 | 240.02 | 404.57 | 5.14 A | 1175.94 | 192.27 | 220.91 | 5.32 A | 1102.97 | 186.48 | 210.75 | 5.23 A |
| SEM | 62.47 | 8.55 | 11.76 | 0.03 | 50.24 | 6.79 | 7.96 | 0.06 | 37.02 | 11.08 | 7.18 | 0.02 |
| P-value | 0.81 | 0.91 | 0.87 | 0.02 | 0.74 | 0.84 | 0.72 | 0.02 | 0.46 | 0.39 | 0.73 | 0.01 |
| CP | ||||||||||||
| Standard (BCAA 0%) | 2030.03 | 237.45 | 400.16 | 5.07 | 1161.53 | 192.72 | 220.71 | 5.24 | 1047.52 | 183.39 | 204.53 | 5.12 |
| Standard (BCAA 10%) | 2029.44 | 237.69 | 394.51 | 5.14 | 1143.26 | 190.29 | 213.57 | 5.35 | 1051.53 | 187.56 | 203.94 | 5.18 |
| Standard (BCAA 20%) | 2121.76 | 247.73 | 409.57 | 5.18 | 1237.24 | 198.24 | 228.05 | 5.43 | 1123.74 | 192.83 | 213.57 | 5.27 |
| Low (BCAA 0%) | 2041.08 | 247.77 | 418.23 | 4.88 | 1107.36 | 198.47 | 228.11 | 4.85 | 1046.29 | 193.98 | 212.52 | 4.92 |
| Low (BCAA 10%) | 2025.57 | 237.19 | 406.59 | 4.99 | 1105.69 | 190.16 | 217.54 | 5.07 | 1035.79 | 181.17 | 203.94 | 5.10 |
| Low (BCAA 20%) | 2038.29 | 232.26 | 399.57 | 5.10 | 1114.55 | 186.22 | 213.73 | 5.21 | 1082.94 | 180.14 | 208.47 | 5.20 |
| SEM | 68.36 | 7.18 | 10.74 | 0.06 | 51.08 | 6.63 | 8.26 | 0.08 | 32.51 | 10.87 | 8.15 | 0.03 |
| P-value | 0.84 | 0.57 | 0.69 | 0.08 | 0.81 | 0.65 | 0.59 | 0.07 | 0.92 | 0.57 | 0.80 | 0.08 |
Abbreviations: VH, villus height; VW, villus width; CD, crypt depth; VH/CD, villus height to crypt depth; CP, crude protein; BCAA, branched-chain amino acid; SEM, standard error of means.
a Different capital letters show a significant difference between means within a variable.
4.2. Insulin-Like Growth Factor 1 Gene Expression
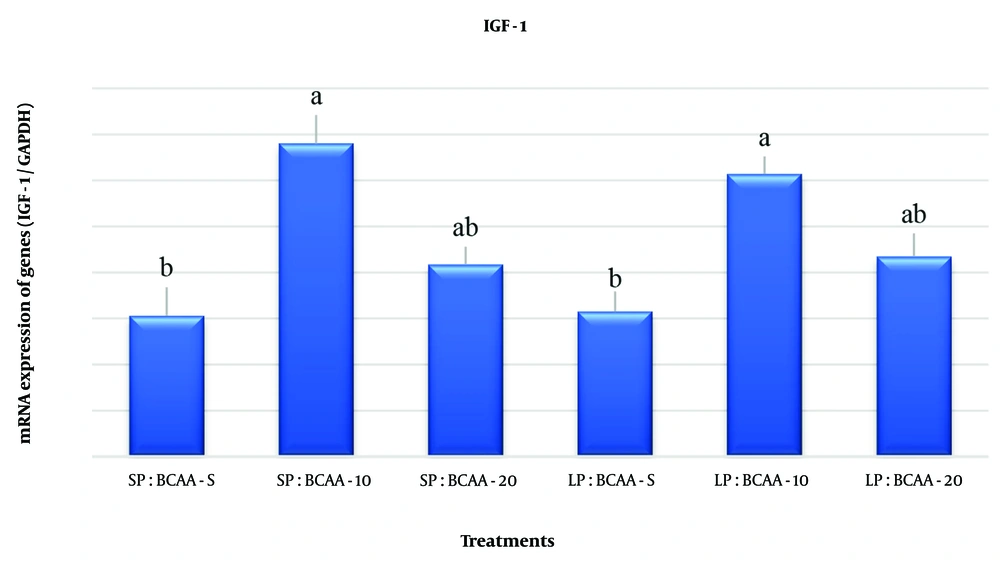
As shown in Figure 1, the expression of IGF-1 mRNA in the liver tissue was higher in the groups containing 10% BCAA than in the standard BCAA groups, regardless of the CP level.
The effect of different levels of crude protein (CP) and branched-chain amino acids (BCAAs) in the diet on relative mRNA gene expression related to the insulin-like growth factor 1 (IGF-1) signaling pathway in the liver tissue of broiler chickens at the age of 42 days. Experimental treatments including: Diet with standard protein and BCAA (SP: BCAA-S); diet with standard CP and BCAA 10% higher than the standard (SP: BCAA-10); diet with standard CP and BCAA 20% higher than standard (SP: BCAA-20); diet with CP 10% less than standard and BCAA standard (LP: BCAA-S); diet with CP 10% less than standard and BCAA 10% higher than standard (LP: BCAA-10); and the diet with CP was 10% lower than the standard and BCAA was 20% higher than the standard (LP: BCAA-20) (n = 24).
5. Discussion
The healthy development and operation of the gastrointestinal organs connected to immune cells, as well as a balanced intestinal microbiota, are essential for improved growth (14, 15). Although the importance of BCAAs for gut health has been acknowledged, the literature has not yet identified a precise mechanism for how they interact. Additionally, research on the use of BCAAs for general intestinal health in chickens has been minimal. It is anticipated that the effects of BCAA supplementation will elicit responses in poultry that are comparable to those shown in mice or pigs (16). However, as the immunity and microbiota of poultry, pigs, and other monogastric species differ naturally, more research is necessary to determine the effects of BCAAs on the gut-health parameters of poultry.
In the current investigation, the rate of VH/CD was higher at a 20% BCAAs level than at the standard level. Longer villi are necessary for all types of chickens to increase surface area and improve nutrient absorption. The constant shedding of villus epithelial cells, which can be significant during specific illness states, is caused by senescence, contact with food, and gut bacteria (14). The VH/CD ratio is a reliable measure of intestinal health because the villus’s lost cells are restored by actively dividing crypts, and the CD rises as villus atrophy occurs (17, 18). The importance of leucine in intestinal development was validated when broiler diets supplemented with leucine (1.37 - 2.2% of food) significantly raised the VH in the jejunum and ileum as well as the VH/CD ratio in the duodenum, jejunum, and ileum (19). According to a study by Allameh and Toghyani (20) on broiler chickens, adding valine to low-CP diets raised the number of goblet cells in the ileum and jejunum by 12% and 9%, respectively, and the VH of the ileum and jejunum by 29% and 17%. The scientists discovered that whereas valine supplementation enhanced protein accretion and improved intestinal architecture in broilers, it had no effect on the immunological response.
The BCAAs may also provide the amino groups needed for the synthesis of other AAs during the transamination process, particularly glutamate and aspartate, which are known to be important sources of energy for the small intestinal mucosa during intracellular protein turnover and nutrient transport (21). If the ratio of leucine to isoleucine and valine is unbalanced, intestinal cell growth and multiplication may not always occur when leucine is added (22, 23).
The idea that BCAA supplementation may stimulate the IGF-1 signaling pathway in broiler liver makes up another aspect of our research. According to a number of authors, the IGF-1 signaling pathway may function as an intrinsic mediator of skeletal muscle adaptation and repair in an autocrine/paracrine mode, enhancing muscle regeneration, regulating protein synthesis, and ultimately increasing muscle mass by promoting satellite cell proliferation and differentiation (24, 25). Previous studies reported that valine supplementation could increase serum, liver, and muscle IGF-1 concentrations in broilers (26). In our study, IGF-1 mRNA expression in the liver was higher in the 10% BCAA groups than in the standard BCAA groups, regardless of the CP level. Referring to the growth function, our data showed that BCAAs act as a positive stimulus for the activation of the IGF-1 signaling pathway in promoting muscle growth.
The IGF hormones are important regulators in growth stimulation, AA elongation stimulation, glucose metabolism, DNA synthesis, protein synthesis, proliferation, and differentiation of various cell types (27). The liver is the main source of production and secretion of IGF-1 in blood serum. The activity of IGF-1 in the body is endocrine, paracrine, and autocrine (28). Paracrine IGF-1 is more important for muscle growth than endocrine IGF-1 (29). The study of IGF-1 as a mediator of growth hormone activity is important (29). The IGF-1 stimulates and increases glucose, AA uptake, protein and DNA synthesis, and inhibits protein breakdown by satellite cell-derived muscle fibers (30). The IGF-1 also stimulates the proliferation of various cell types (31).
The primary role of BCAAs is to participate in the building of body proteins. The process of protein synthesis in cells is regulated by the mTOR complex. The mTOR receptors receive messages from extracellular compounds that activate or inhibit protein synthesis. The TIGF-1 is probably one of the stimulators of protein synthesis (32). Amino acid signaling is initiated by mTOR and activates S6k1 and 4EBP1 (33). Consumption of a high-protein diet clearly activated S6k1 and increased its activity in pectoral muscle (34). The S6k1 activation is sensitive to the availability of dietary AAs during the first days of broiler feeding, which can increase mRNA translation in the skeletal muscle of chickens (35). In the present study, a 20% BCAA level had no significant effect on IGF-1 gene expression compared to the control treatment, while a 10% level significantly increased IGF-1 gene expression, which probably indicates excessive BCAA levels. The BCAAs are taken up by the liver in small amounts as nutrient signals in most animal species except fish (35). Imbalance or excess of BCAAs affects the transfer of their co-rows. In addition, the excess of one BCAA stimulates the oxidation of other BCAAs because the BCKD enzyme has a high affinity for all of them (36).
5.1. Conclusions
In general, feeding low CP diets decreased the VH/CD ratio, but BCAA supplementation in low CP diets could increase the VH/CD ratio and improve IGF-1 gene expression. Therefore, BCAA supplementation could be beneficial to improve IGF-1 gene expression and intestinal morphology in low-CP diets, and in the formulation of poultry diets, different feed ingredients must be carefully selected to ensure a suitable proportion of BCAAs.