1. Background
Abortion is pregnancy termination before 20 weeks of gestation or weight of less than 500 gr for a fetus. The basis of this evidence differs from stillbirth, which is the termination of pregnancy after 20 weeks (1, 2).
Recurrent miscarriage (RM) is defined as the presence of two or more spontaneous abortions, which imposes emotional, physical, and financial complications for the couples. It has been estimated that 1% to 3% of couples experience RM (3). The disease could be considered as a multifactorial disease, affected by factors such as maternal nutrition, hormonal and endocrine disorders, anatomical abnormalities, infections, immune system and metabolic pathway malfunctions, endometriosis, sperm quality, and genetic abnormalities (2, 4-6).
Though the genetic basis of RM is not well understood, many gene mutations and chromosomal abnormalities are known to be related. Other genetic abnormalities involved in RM, include mosaicism in placenta, skewed X-chromosome inactivation, and sperm DNA disorders (7, 8).
Beside these, using association studies, variations in more than 90 genes are known to be related to RM. Polymorphisms in Leiden factor (thrombophilia-associated factor) and MTHFR genes are the most popular variations. The second known gene variations belong to genes involved in inflammation (3). Among genetic factors related to RM, chromosomal abnormalities are the most influential. Karyotype abnormalities have a very high incidence in products of abortion, but low in the couples (9).
The chromosomal rearrangement is the result of rejoining of breaking DNA molecules in DNA repair system, and encompasses many classes, including translocation, inversion, deletion, and duplication. Although it changes the main structure of chromosomes, in the balance form there is no phenotypic manifestation. Most individuals carry balanced reciprocal translocations and inversions are considered as normal carriers. However, it still has the chance of having unbalanced gametes (2, 9).
Balanced abnormality in the structure of chromosomes is considered as a cause of recurrent miscarriage in couples. The incidence of carrier status is different in populations, yet it is estimated around 0.7% in the general population. This amount increases with the number of miscarriage, so that is more than 5% in patients with a history of 3 miscarriages (10, 11). In this study, 30 couples with RM were evaluated for their karyotypes, for the first time in Sistan and Baluchestan province.
2. Methods
2.1. Patient Selection
The subjects of this study included 60 individuals (30 couples) with a history of recurrent miscarriage that had referred to Imam Ali Hospital (3 or more abortions before 20 weeks of pregnancy or baby weighing less than 500 grams). All patients were examined for thrombophilia (factor V Leiden, prothrombin G20210A gene mutation, protein C and S, and antithrombin III), antiphospholipid syndrome, Lupus, polycystic ovarian syndrome, infections, hormonal status (thyroid stimulating hormone (TSH), prolactin, and fasting blood sugar (FBS)), and ultrasonography for anatomic anomalies. According to the Declaration of Helsinki, all participants provided informed consent and accepted codes of the university ethics committee. After the examinations, patients were referred to the genetic center of Ali-Asghar hospital in Zahedan for chromosomal analysis by karyotyping. Two milliliters of peripheral blood was collected from each participant with heparin as an anticoagulant.
For this purpose, lymphocyte cell cultures were incubated at 37°C for 72 hours. Cell culture consisted of RPMI 1640 medium (Gibco, USA) supplemented with L-glutamine and enriched with Fetal Bovine Serum (FBS, Gibco, USA), phytohaemagglutinin (Gibco, USA) and penicillin/streptomycin. The next step was the harvest of cells by adding colcemid (Gibco, USA) for 45 minutes at 37°C, continued with treatment by a hypotonic solution of KCl (Merck, Germany) for 25 minutes at 37°C. The cells were then fixed by fixative solution, containing 3:1 methanol-acetic acid (Merck, Germany). Pancreatin-Giemsa procedure was used for staining the chromosomal spreads. Olympus photomicroscope (CX-30, Japan) was applied for chromosomal analysis at 400 to 550 band resolution. Furthermore, 100 metaphase spreads were evaluated in cases of mosaicism. The results were reported based on the international system for human Cytogenetics nomenclature (ISCN).
3. Results
Thirty couples with a mean age of 29 ± 5 years for females and 33.3 ± 5.6 for males were recruited in this study. All these participants were examined for thrombophilia, antiphospholipid syndrome, Lupus, polycystic ovarian syndrome, infections, hormonal status, and ultrasonography for anatomic anomalies, and these individuals revealed a disorder in all the items listed. The mean body mass index (BMI) for females was 21.7 ± 1.4. About 43.7% of couples had consanguineous marriage. The average number of gestations in each couple was 4.1 ± 1.4 (with a minimum of 3 and a maximum of 8), yet the mean number of children for each couple was 0.8 ± 1.2. Seventeen couples (56.7%) had no children.
The mean number of abortions was 3.3 ± 0.8 (with a minimum of 3 and a maximum of 7). Overall, 20% of the participants had a history of miscarriage in their family.
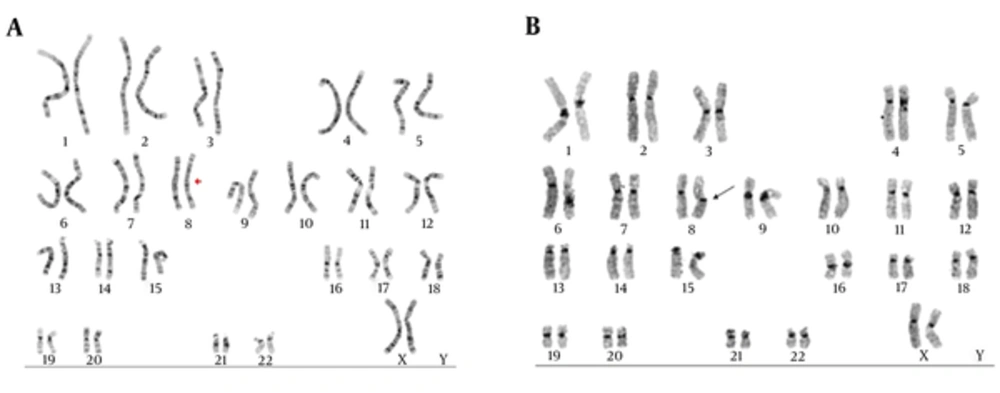
Among all male and female subjects of this study, only 2 couples (6.7%) showed chromosomal aberration: mos45, X(3)/46XX(47) and 46,XX, inv(8)(p12q21). Both of them were females, yet none of the males revealed chromosomal disorders. Both of these females had no children with a record of 3 miscarriages. These females and their partners had no history of miscarriage in their family members. Figure 1A and B show the karyotype of the female with inv (8)(p12q21), by G-banding and C- banding, respectively.
4. Discussion
It has been estimated that 15% to 25% of pregnancies fail, while recurrent spontaneous abortion is considered to be a medical problem by at least 5% of couples (9). The exact cause of miscarriage is identified in only 50% of cases and the rest is mainly unknown (12). Chromosomal disorders are known as one of the main causes of very early miscarriages (13), in 50% of pregnancy losses in the first trimester (14-16). In most cases, the cause of abortion in the second trimester of pregnancy could be due to anatomic abnormalities and other genetic factors in parents (17). In this cross-sectional study, to investigate the role of chromosomal disorders in recurrent miscarriage, 30 couples with abortion-related disorders were recruited. The karyotype of peripheral blood of each participant was prepared, using the standard G-banding technique. There was no chromosomal aberration in males, while 2 of the females carried chromosomal disorders, making up 6.7% of cases. One of them had a mosaicism in the X chromosome and no symptom of Turner’s syndrome. She was 26-year-old, had no children, and had a history of 3 miscarriages. Mosaicism of the X chromosome is a known cause of abortion (18). The other one showed a normal phenotype, yet with a pericentric inversion in chromosome 8. In inversions, 2 breaks occurred in one chromosome.
Then, before repairing these breaks, the area between these points rotated 180 degrees. If breaks did not disturb the function of a gene and as inversions make no changes in the amount of DNA, most carriers showed a normal phenotype. However, during meiosis for gametogenesis 50% of produced gametes carried deletions and insertions. 46,XX inv(8)(p12q21) was the karyotype of a 36-year-old female with no children and a history of 3 miscarriages. This rearrangement was reported in another study as a cause of recurrent miscarriage in a male (19).
Many studies have been conducted to find the causes of recurrent spontaneous abortion in different populations. In most populations balanced structural chromosome abnormality is responsible for only 2% to 6% of RM (20). However, this amount is greater in samples obtained from an aborted mass. In Japan, abnormal embryonic outcomes were found in 41.1% of cases of recurrent abortion (13). In the Netherlands, chromosomal abnormalities in couples with RM was estimated as 0.5% to 10.2% (21), while balanced rearrangement was stated as the cause of 1.9% of RM in the UK (22). Many investigations were conducted in Iran to find the role of chromosomal aberrations in Iran. In a study in Ahvaz, 12.7% of females and 11.3% of males with a history of 2.8 RM per case showed abnormal karyotypes (23). However, this rate was 12.5% in Yazd (24), 9.50% in Tehran (25), and 9.8% and 11.7% in two separate studies in Mashad (21, 26). The number of chromosomal disorders found in the current study was less than other studies in Iran, which may be because of the low number of participants that took part in this investigation. Therefore, it is a necessity to have more evaluations in this regard. In addition, it is essential for this group of patients to be evaluated by more powerful techniques of molecular cytogenetics like quantitative fluorescent polymerase chain reaction (QF-PCR), fluorescence in situ hybridization (FISH), and multiplex ligation-dependent probe amplification (MLPA) to find the chromosomal aberrations that may not be revealed by the standard G-banding technique.