1. Background
Breast cancer (BC) is one of the most common cancers and is the main cause of mortality from cancer among women worldwide (1). BC constitutes about 33% of malignancies among women (2). This malignancy is a multifactorial disease in which several genetic and environmental factors are involved. The role of genetic and heritable factors in BC has been proven. For example, BC risk in women with a mother or sister suffering from BC is four times higher than in other women. So far, the effects of various genes on BC, which can be referred to as oncogenes, have been studied (3). The product of oncogenes controls cellular growth through various mechanisms and is involved in cell transformation or induction of cancer (4).
Today, BC prognosis and diagnosis are performed through various variables such as histology, tumor size, lymph node, estrogen receptor (ER), progesterone receptor and overexpression of HER-2 (5, 6). Estrogen and progesterone play an important role in the growth and normal functioning of breast glands. Disrupting the function of these hormones also causes the development of BC. These hormones act through connecting to their receptors, which are known as ER and PR. These receptors function as signaling triggers and ligand-activated transcription factors (7).
BC is classified as ER+/PR+ when ER and PR proteins are overexpressed. ER+/PR+ tumors are very different in comparison to ER+/PR- in responding to hormone therapy (8). About 55% - 60% and 8% of women with ER+ and ER-BC respond to hormone therapy, respectively. Also, BC tumors with better differentiation are mostly ER+/PR+ and have better prognosis (9).
The human epidermal growth factor receptor 2 (HER-2) is one of the most important biomarkers and is currently used for the diagnosis and differentiation of different types of BCs. The overexpression of HER-2 is observed in approximately 15% of BCs (10). Studies showed that the overexpression of HER-2 is associated with resistance to treatment with endocrine agents in BC patients (11). In BC patients with HER-2 overexpression (HER-2+), chemotherapy and treatment with trastuzumab improve survival (12). The multi-drug resistance associated with protein 9 (MRP9) is a member of the superfamily of ATP-binding cassette (ABC) transporters and is encoded by ABCC12 gene in humans. Studies showed that MRP9 gene in mice is expressed in various tissues such as the ovaries, brain, breasts, prostate and testes (13, 14). In a study by Bera et al. it was shown that ABCC12 gene is an unusual truncated member of the ABC transporter superfamily and is highly expressed in BC (15). So far, there is no other study on the physiological role and involvement of ABCC12 gene in BC or other cancers.
2. Objectives
Therefore, the aim of this study was to investigate the expression of ABCC12 gene in patients with ductal breast carcinoma and its association with other biomarkers such as ER, PR and HER-2.
3. Methods
3.1. Study Subjects
In this study 14 women were recruited from hospitals of Zanjan city, Iran, during October to December 2017. Nine women with breast invasive ductal carcinoma (age range: 29 - 61 years) and the mean age of 45 years, who had not received any treatments such as chemotherapy or radiotherapy, were evaluated. BC was diagnosed in the subjects by clinical examination and gross pathology. Also, five healthy women (age matched) were selected as healthy controls. These subjects had presented for routine physical examination. BC tissue from patients and normal breast tissue from healthy controls (0.01 - 0.005 g) were taken by a surgeon using the core needle biopsy (CNB) method. A portion of the obtained samples was blocked by paraffin and another portion was kept in a nitrogen tank. All the studied women signed a consent form in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The sample collection procedure was approved by the Ethics Committee of Islamic Azad University, Zanjan Branch (IR.IAU.Z.REC.1396,72).
3.2. Immunohistochemical (IHC) Procedure
The paraffin sections were cut at 1 - 2 µm thickness and kept in an incubator for 24 hours at 37°C. In order to remove paraffin from the tissues, the slides were placed in an oven at 60°C for 60 minutes. The slides were placed in a jar containing xylene for 7 minutes. Subsequently, the slides were placed in absolute alcohol (5 minutes), 85% alcohol (5 minutes), 70% alcohol (5 minutes) and phosphate buffered saline (PBS; 5 minutes), respectively. The slides were placed inside a target retrieval solution and transferred to a steamer for 15 minutes. The slides were then placed in dihydrogen dioxide at a concentration of 3.5% - 5% for 7 - 10 minutes. After washing, the slides were placed in the PBS solution with 7.2 - 7.4 pH. The surrounding tissue was marked with Dako marker, so that after the addition of the primary antibodies, the solution remained in the target area. Primary antibodies of ER (flex monoclonal mouse anti human estrogen receptor α; Dako), PR (rabbit monoclonal antibody, Diagnostic biosystem) and HER-2 (rabbit anti C-erbB2 monoclonal, diagnostic biosystem) were added to each of the slides and placed in the incubator at 37°C for 1 hour. After washing with IHC washing buffer, the slides were placed in PBS solution for 5 minutes. Then, secondary antibodies (envision detection systems/peroxidase/DAB; rabbit-mouse; diagnostic biosystem) were added to each of the slides and placed in the incubator at 37°C for 30 minutes, and after washing with IHC washing buffer, they were placed in PBS for 5 minutes. 3,3’-Diaminobenzidine (DAB) solution was added to the slides and washed with distilled water after 5 - 7 minutes. The slides were placed in a jar containing hematoxylin for 20 - 30 minutes and washed with distilled water. The slides were placed in 70% alcohol (5 minutes), 80% alcohol (5 minutes), 100% alcohol (5 minutes) and xylene (5 minutes), and then they were dried at room temperature. Finally, mounting glue was poured onto the slides, the cover slip was put in place, and then examined after drying.
3.3. HER-2 Gene Amplification by Chromogenic In Situ Hybridization (CISH)
The paraffin sections were cut at 0.2-μm thickness and placed on the prepared slides with a silane adhesive, and then the slides were placed in oven at 60°C for 20 - 24 hours. In order to remove paraffin, the slides were placed in 100% alcohol (5 minutes), 80% alcohol (5 minutes), 70% alcohol (5 minutes) and distilled water (10 minutes), respectively. The slides were placed in PT EDTA solution (95°C for 20 minutes) and then washed with Tris-buffered saline (TBS) and then dried. The pepsin solution (100 λ) was added on the slides and incubated at 37°C for 7 minutes and then washed with TBS-Tween 20. In order for dewatering, the slides were placed in 70% alcohol (3 minutes), 80% alcohol (3 minutes) and 100% alcohol (3 minutes), respectively. The HER-2/CEN17 probes (10 λ) were added to the slides. The slides were placed at 80°C for 5 minutes for denaturation and then placed at 37°C for 16 hours for hybridization. In order to separate the cover slip from the tissue, the slides were placed in post-hybridization wash buffer at room temperature for 5 minutes and then washed with post-hybridization wash buffer (75°C) and subsequently placed in TBS solution. The peroxidase blocking solution (10 λ) was added on the slides and then washed with TBS-Tween 20. The mix of secondary Ab (100 λ) was added on the slides and then washed with TBS-Tween 20. Also, the mix of polymers (100 λ) was added on the slides and then washed with TBS-Tween 20. In the following, red chromogen (100 λ) was added on the slides and incubated at room temperature for 10 minutes. Then, green chromogen (100 λ) was added on the slides and incubated at room temperature for 10 minutes. After washing the slides with distilled water, the hematoxylin solution was added on the slides for 30 seconds and then washed with distilled water. Finally, after dewatering, the slides were placed in xylene for 30 seconds and the cover slip was placed on it.
3.4. ABCC12 Gene Expression by Real Time PCR
RNA extraction was performed using the RNX kit (Sinagene, Iran) according to the manufacturer’s instructions. The concentration of the extracted RNA was determined using a spectrophotometer. cDNA was synthesized using cDNA synthesis kit (TaKaRa, Japan) according to the manufacturer’s instructions. The expression rates of ABCC12 and GAPDH (housekeeping genes) were evaluated using specifically-designed primers (Table 1). Real Time polymerase chain reaction (PCR) reaction was performed using SYBR Green (Ampliqon, Denmark) in triplicate. PCR was performed using rotor-gene Q PCR system at 95°C for 15 minutes and 40 cycles at 95°C for 15 seconds, 60°C for 30 seconds and 72°C for 30 seconds. Also, electrophoresis was performed on agarose gel to ensure the correct PCR reaction. The comparison of ABCC12 expression between the cases and controls was performed using Rest software. t-test was used for the assessment of difference between the groups. P value less than 0.05 was considered statistically significant. Also, the obtained threshold cycles (TCs) from the cases and controls were compared using the 2-ΔΔCt and the Pfaffl method.
| Gene | Sequence | Melting Temperature, °C | Production Size, bp |
|---|---|---|---|
| ABCC12 | 1049 | ||
| GAGGTGCTCAATATACTGTCAAGTGA | 56.4 | ||
| GAAACTCATTCATTGTCTGAACTCGC | 56.4 | ||
| GAPDH | 257 | ||
| AGAAGGCTGGGGCTCATTTG | 53.8 | ||
| AGGGGCCATCCACAGTCTTC | 55.9 |
4. Results
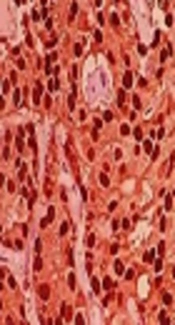
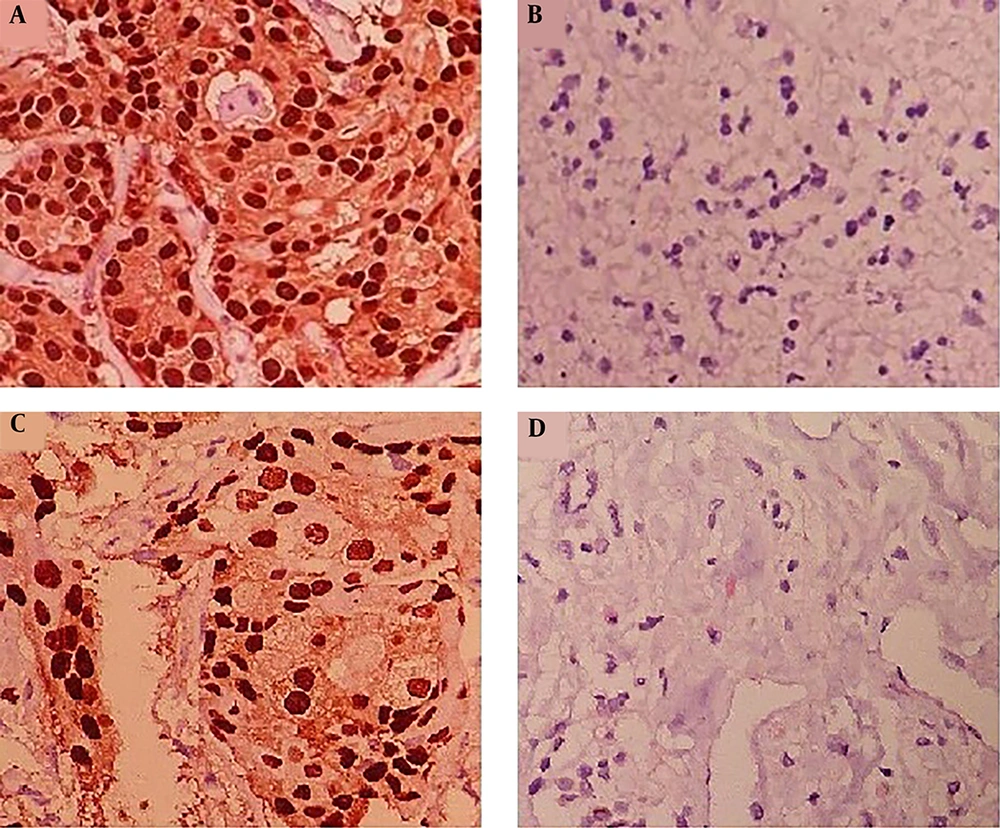
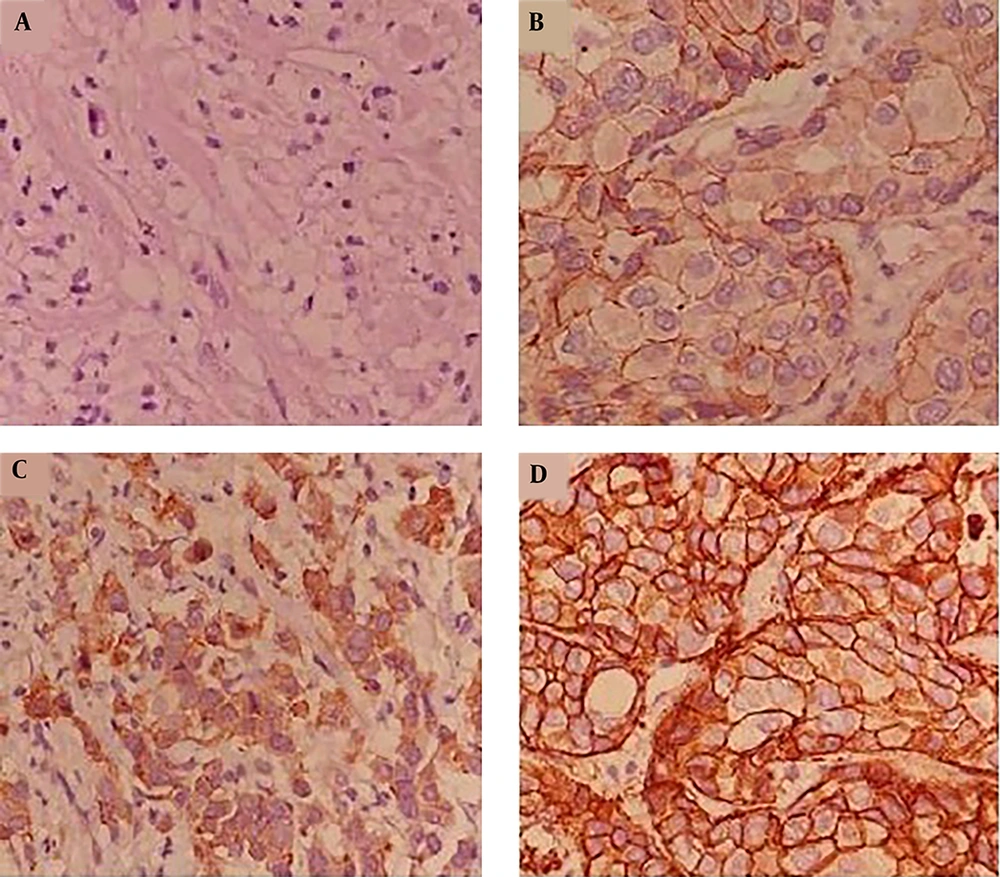
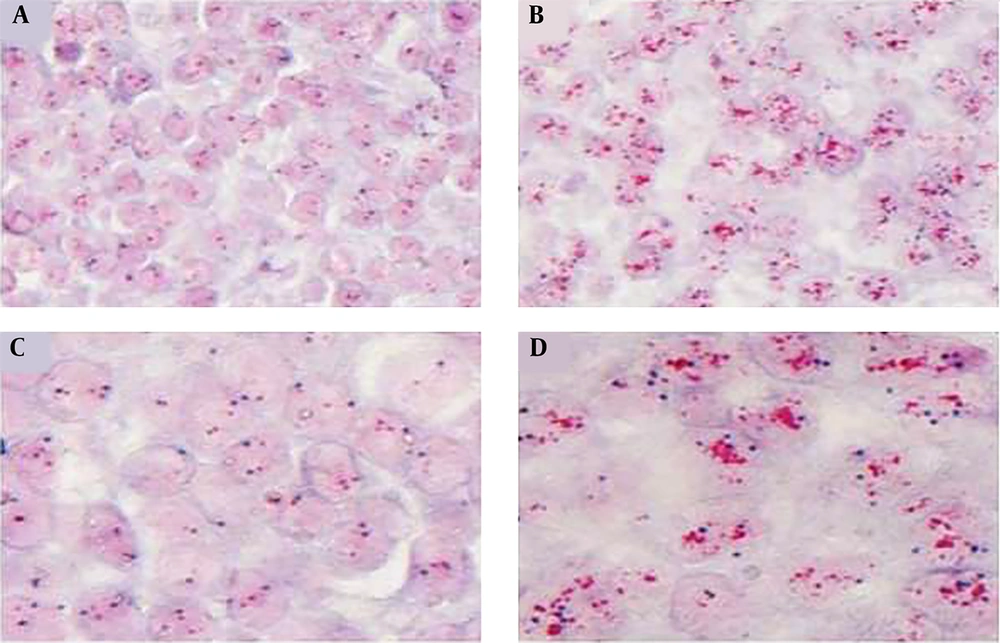
The results of ER and PR antigen assessment were considered as - and + (Figure 1). The obtained results from immunohistochemistry (IHC) analysis are presented in Table 2. Also, the HER-2 antigen was considered as 0 to 3+ based on the intensity of cancer cells nucleus staining (Figure 2). According to chromogenic in situ hybridization (CISH) analysis, two patients showed positive amplification for HER-2 antigen (Table 2 and Figure 3).
| Patient Number | HER-2 Antigen | PR Antigen | ER Antigen | HER-2 Amplification | Subtype |
|---|---|---|---|---|---|
| 1 | - | - | - | - | Triple negative |
| 2 | 3+ | - | - | + | HER-2 enrich |
| 3 | - | - | + | - | Luminal B |
| 4 | 1+ | + | + | - | Luminal B |
| 5 | 1+ | + | + | - | Luminal B |
| 6 | 1+ | + | + | - | Luminal B |
| 7 | 3+ | + | + | + | Luminal B |
| 8 | 1+ | + | + | - | Luminal B |
| 9 | - | + | + | - | Luminal A |
The obtained results from real time PCR showed that the expression of ABCC12 gene significantly increased in tumor tissues. The mean expression of this gene in patients was 3.74 times higher than in healthy controls (P = 0.007). The expression of ABCC12 gene was evaluated in luminal B subtype (patient numbers are 3 to 8). The results showed that the mean expression of ABCC12 gene in luminal B subtype was 3.94 times higher than in healthy controls (P = 0.002). The average expression of ABCC12 gene in HER-2 (+1) subtype (patient numbers are 4, 5, 6 and 8) was 3.54 times higher than in healthy controls (P = 0.0006). Also, the mean expression rates of ABCC gene in ER (+) (patient numbers are 3 to 9) and PR (+) (patient numbers are 4 to 9) subtypes were 3.86 (P = 0.002) and 3.63 (P = 0.0005) times higher than in healthy controls, respectively.
In the present study, HER-2 gene amplification was evaluated in patients over and under 45 years old. Of six patients aged over 45 years old, one patient showed HER-2 gene amplification. Of the three patients aged under 45 years old, one patient showed HER-2 gene amplification. Also, there was no difference in the expression of HER-2 gene between patients aged over and under 45 years old. The statistical analyses showed no significant relationship between amplification and expression of HER-2 gene and age of the patients (P > 0.05). However, by decreasing patients’ age, ER expression was significantly increased (P < 0.05).
5. Discussion
BCs are heterogeneous at morphologic, immunohistochemical and molecular levels. This heterogeneity persists even within apparently homogeneous groups of ER+ and ER- tumors. In the past few years, ER+ tumors have been subjected to multigene prognostic/predictive assays to identify more aggressive types of ER+ tumors that are expected to benefit from additional chemotherapy and define the risk of recurrence in an individual patient (16, 17).
The evaluation of ER, PR and HER-2 genes is very important to determine BC subtype and guide the therapeutic strategy of patients with BC (18). ABC proteins are known as the carriers of lipids, carbohydrates, amino acids, steroids and chemical compounds such as chemotherapy drugs and products derived from metabolism (19). Considering that the failure of chemotherapy is associated with increased expression of ABC proteins (20), it is possible to use better treatment methods and drugs in treating patients with BC by examining these proteins. Therefore, in this study we evaluated the association of ABCC12 gene expression with ER, PR and HER-2 biomarkers in patients with breast ductal carcinoma.
Our results showed that ABCC12 gene expression significantly increased (3.74 times) in patients with breast ductal carcinoma compared to healthy controls. Therefore, maybe increasing the expression of this gene can disrupt the treatment of patients and performance of the used drugs. In agreement with our study, Hlavac et al. showed that ABCC12 gene expression significantly increased in post-treatment breast tumors (21).
Kubelka-Sabit et al. evaluated HER-2 status in BC patients using CISH and IHC methods and showed HER-2 gene amplification in all 3+ patients, whereas 1+ patients did not show HER-2 gene amplification (22). Our findings are in agreement with this result. In a similar study by Musa et al. it was reported that HER-2 gene was amplified in 36.5% of +2 patients (23), whereas in our study there were no +2 patients.
Madrid and Lo demonstrated that HER-2 was amplified in all +3 patients and 45% of +2 patients. Also, in patients under 50 years of age, the amplification rate of HER-2 gene was higher than in patients over 50 years of age (24). However, our study showed no significant association between HER-2 gene amplification and age of patients. In a study, Anders et al. evaluated the expression of PR, ER and HER-2 genes in patients with BC (under 45 years old and over 65 years old). Their results showed a significant increase in tumor size, tumor grade, lymph node metastasis and HER-2 expression in subjects under 45 years of age, whereas ER expression was decreased significantly in this group (25). However, our study showed that the expression of ER increased significantly in patients under 45 years of age, but the expression of HER-2 remained unchanged and no significant association was found between age of patients and expression of this gene. The difference in sample size could explain the difference in results. Brase et al. and Paik et al. also showed continuous expression of HER-2 RNA according to IHC/FISH categories. It is notable that although a major overlap exists between the groups, the HER-2 IHC 1+ category shows significantly higher RNA levels than the IHC 0 category, suggesting that some biologic meaning is associated with the separation of these IHC staining groups, rather than being merely a staining artifact (26, 27).
In another study, Collins et al. showed that luminal B subtype frequency (35%) was higher than other subtypes in BC patients under 40 years of age and ER and PR expression significantly increased in these patients (28). Also, our study showed that the frequency of luminal B subtype was higher than others in patients under 45 years old and expression of ER was significantly increased.
Considering that the expression of ABCC12 gene was significantly increased in patients with breast ductal carcinoma, it can be stated that this increase disrupts patient treatment and the performance of used drugs. The expression of ABCC12 gene can be a useful prognostic factor to identify the stages of BC and its therapeutic approaches. Evaluation of ABCC12 gene expression character in BC patients with other diagnostic profiles was performed for the first time in this study in Iranian women. Although this study added to our understanding of BC, sample size in our study was not large enough; thus, further studies using a larger cohort of patients are recommended.